Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
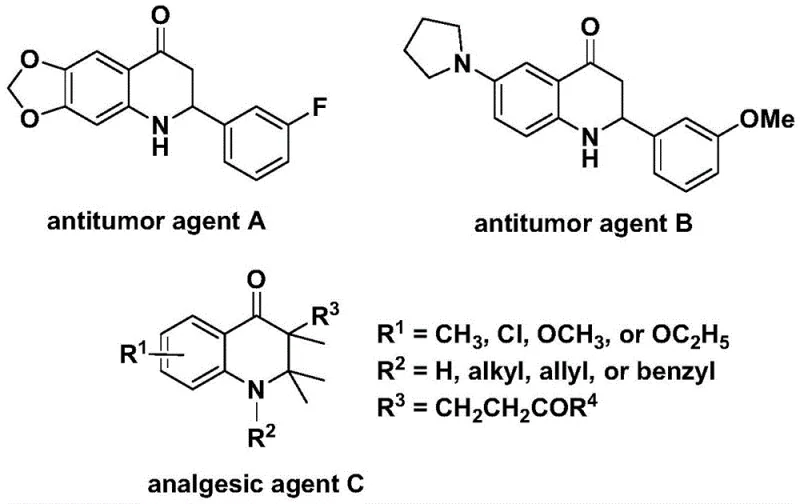
The synthesis of nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities such as antitumor and analgesic properties. As illustrated in the structural diversity of bioactive molecules like antitumor agent A and analgesic agent C, the 2,3-dihydroquinolone core is a privileged structure in drug discovery. However, accessing these complex skeletons efficiently has historically posed significant challenges regarding regioselectivity and functional group tolerance. Patent CN112239456B introduces a transformative approach utilizing transition metal palladium catalysis to construct these valuable frameworks via a carbonylation reaction. This methodology not only streamlines the synthetic route but also enhances the feasibility of producing high-purity pharmaceutical intermediates on a commercial scale, addressing critical needs for reliable pharmaceutical intermediate suppliers globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the 2,3-dihydroquinolone skeleton often rely on multi-step sequences involving harsh cyclization conditions or the use of unstable acylating agents. These conventional methods frequently suffer from poor atom economy and limited substrate scope, particularly when introducing diverse substituents at the 2- and 3-positions of the quinolone ring. Furthermore, many existing protocols require stoichiometric amounts of toxic reagents or extreme temperatures that degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purge. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times, while R&D teams face bottlenecks in generating diverse analog libraries for structure-activity relationship studies due to the rigidity of older synthetic designs.
The Novel Approach
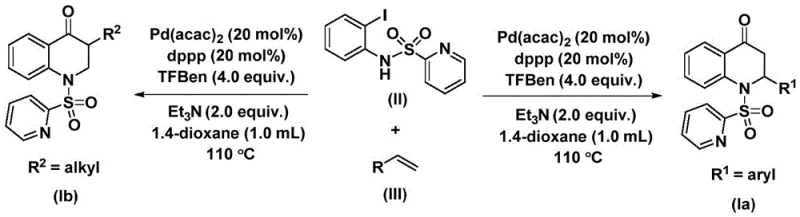
The innovative strategy disclosed in the patent leverages a palladium-catalyzed carbonylation reaction between N-pyridine sulfonyl-o-iodoaniline and various olefins. This direct assembly method bypasses the need for pre-functionalized carbonyl sources by utilizing a solid carbon monoxide surrogate, specifically 1,3,5-trimesic acid phenol ester, which releases CO in situ under mild thermal conditions. By employing a robust catalyst system comprising Pd(acac)2 and dppp ligand, the reaction achieves high conversion rates at moderate temperatures ranging from 100 °C to 120 °C. This approach significantly simplifies the operational complexity, allowing for the direct synthesis of both 2-aryl and 3-alkyl substituted derivatives with exceptional efficiency, thereby offering a versatile platform for cost reduction in API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent transformations while maintaining the integrity of the sulfonamide protecting group. Following this activation, the carbon monoxide molecule, released thermally from the phenol ester surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This insertion step is highly exothermic and drives the formation of the carbonyl functionality essential for the lactam ring closure, ensuring that the carbonyl group is positioned precisely for the subsequent cyclization event without requiring external gas handling equipment.
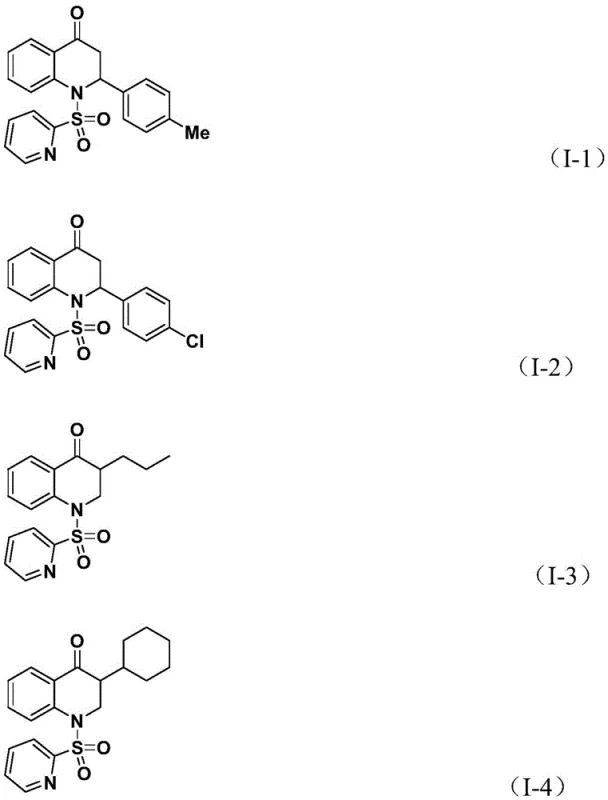
Subsequently, the coordinated olefin substrate undergoes migratory insertion into the acyl-palladium bond, creating a new carbon-carbon bond and extending the molecular framework to form a palladium-alkyl intermediate. The final step involves an intramolecular nucleophilic attack or reductive elimination that closes the six-membered heterocyclic ring, releasing the substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst. This mechanistic pathway explains the broad substrate compatibility observed in the experimental data, as the steric and electronic properties of the olefin can be tuned without disrupting the fundamental catalytic cycle, allowing for the synthesis of complex structures like compounds I-1 through I-5 with high fidelity.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The experimental procedure outlined in the patent provides a robust protocol for executing this transformation with high reproducibility. The process begins by combining the palladium catalyst, ligand, base, and CO surrogate in an aprotic organic solvent such as 1,4-dioxane, which effectively dissolves all reactants and promotes the catalytic turnover. The reaction mixture is then heated to 110 °C for a duration of 48 hours to ensure complete consumption of the starting materials, after which standard workup procedures involving filtration and silica gel chromatography yield the pure product. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis steps provided below.
- Charge a reaction vessel with bis(acetylacetone)palladium, dppp ligand, triethylamine, and the carbon monoxide surrogate (1,3,5-trimesic acid phenol ester) in an organic solvent like dioxane.
- Add the key starting materials: N-pyridine sulfonyl-o-iodoaniline and the selected olefin substrate to the reaction mixture under inert atmosphere.
- Heat the mixture to 110 °C for 48 hours, then filter and purify the crude product via silica gel column chromatography to isolate the target dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this technology offers substantial benefits by utilizing commercially available starting materials such as simple olefins and substituted anilines, which are abundant in the global chemical market. The reliance on a solid CO surrogate eliminates the safety hazards and logistical complexities associated with handling high-pressure carbon monoxide gas, thereby reducing facility requirements and insurance costs for manufacturing sites. Additionally, the use of a heterogeneous-like workup involving simple filtration followed by chromatography minimizes the generation of aqueous waste streams, aligning with increasingly stringent environmental regulations and reducing the burden on waste treatment infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the use of a catalytic amount of palladium significantly lower the raw material cost per kilogram of product. By avoiding specialized high-pressure equipment for gas reactions, capital expenditure for plant setup is drastically reduced, while the high yields reported (up to 88%) minimize material loss and maximize overall process efficiency.
- Enhanced Supply Chain Reliability: Since the key building blocks like 4-methylstyrene or cyclohexene are commodity chemicals produced at massive scales worldwide, the risk of raw material shortage is negligible. This ensures a stable and continuous supply of the intermediate, allowing procurement teams to negotiate better long-term contracts and avoid price volatility associated with niche specialty reagents.
- Scalability and Environmental Compliance: The reaction conditions are mild enough to be safely scaled from gram to multi-kilogram batches without significant exotherms or safety incidents. The simplified post-treatment process reduces solvent consumption and energy usage compared to traditional multi-step syntheses, facilitating easier regulatory approval for commercial production and supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. Understanding these details is essential for R&D directors evaluating the feasibility of integrating this chemistry into their existing pipelines. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation to ensure accuracy and relevance for technical decision-making.
Q: What are the primary advantages of this carbonylation method over traditional cyclization routes?
A: This method utilizes readily available olefins and avoids harsh conditions often required for traditional ring closures. It offers excellent functional group tolerance and high atom economy by directly inserting the carbonyl group.
Q: Can this process be scaled for industrial manufacturing of API intermediates?
A: Yes, the patent explicitly demonstrates gram-level scalability with simple post-treatment procedures like filtration and chromatography, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substituents are compatible with this palladium-catalyzed system?
A: The system shows broad substrate compatibility, successfully accommodating aryl groups with methyl, chloro, or fluoro substituents, as well as linear and branched alkyl chains and silyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,3-dihydroquinolone intermediate meets the exacting standards required for downstream API synthesis, providing our partners with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain objectives and drive innovation in your pharmaceutical portfolio.
