Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
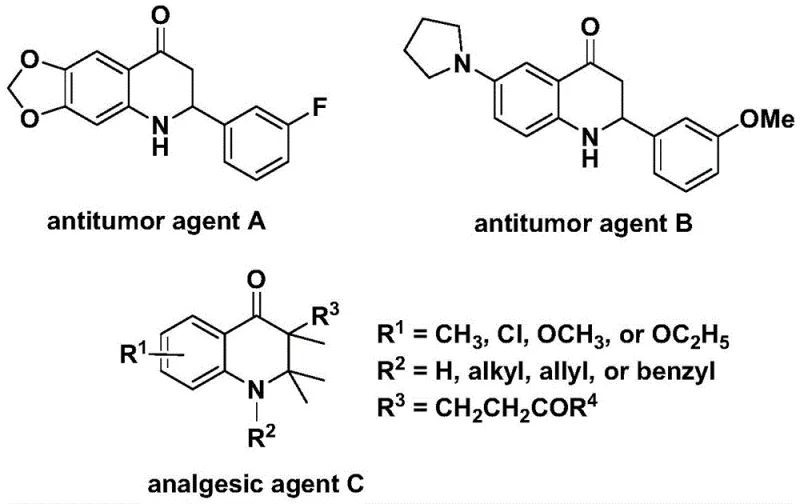
The pharmaceutical industry constantly seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B introduces a transformative preparation method for substituted 2,3-dihydroquinolone compounds, a scaffold critical for developing next-generation therapeutics. These structures are not merely academic curiosities; they form the core backbone of potent antitumor agents and analgesics, as evidenced by historical precedents in medicinal chemistry literature. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary building blocks. This approach represents a significant leap forward in synthetic efficiency, replacing hazardous gaseous carbon monoxide sources with a stable, solid surrogate. For R&D directors and procurement specialists alike, this innovation promises a streamlined pathway to high-value intermediates, reducing both safety risks and operational complexity in the manufacturing of complex heterocyclic systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the 2,3-dihydroquinolone skeleton has relied heavily on classical cyclization strategies that often suffer from severe limitations regarding atom economy and safety. Conventional carbonylation reactions typically require the direct use of carbon monoxide gas, which poses substantial handling hazards due to its high toxicity and flammability, necessitating specialized high-pressure reactors and rigorous safety protocols that drive up capital expenditure. Furthermore, many existing synthetic routes exhibit poor functional group tolerance, leading to side reactions that complicate downstream purification and reduce overall yield. The reliance on harsh conditions often restricts the substrate scope, preventing the incorporation of sensitive moieties required for modern drug design. Consequently, manufacturers face bottlenecks in scaling these processes, as the transition from milligram-scale discovery to kilogram-scale production becomes fraught with engineering challenges and regulatory hurdles associated with hazardous reagents.
The Novel Approach
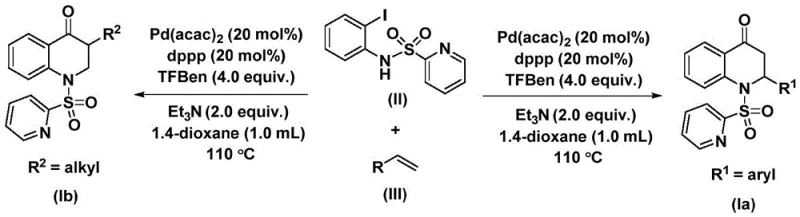
In stark contrast, the methodology outlined in patent CN112239456B offers a sophisticated solution by employing 1,3,5-trimesic acid phenol ester as an effective solid carbon monoxide surrogate. This strategic substitution allows the reaction to proceed under much milder conditions, specifically at atmospheric pressure in standard glassware, thereby eliminating the need for expensive high-pressure infrastructure. The process utilizes a palladium bis(acetylacetonate) catalyst system paired with a dppp ligand, which facilitates a highly selective oxidative addition and subsequent carbonyl insertion. As illustrated in the general reaction scheme below, this novel approach enables the direct coupling of N-pyridine sulfonyl-o-iodoaniline with a diverse range of olefins. The result is a versatile platform capable of generating both 2-aryl and 3-alkyl substituted derivatives with remarkable efficiency. This shift from gas-phase to solid-phase carbonylation sources fundamentally alters the risk profile of the synthesis, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production lines.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The success of this synthetic route lies in the intricate dance of the palladium catalytic cycle, which orchestrates the formation of multiple bonds in a single operation. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide molecule, released in situ from the thermal decomposition of the 1,3,5-trimesic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes coordination and migratory insertion with the incoming olefin substrate. The regioselectivity of this insertion is governed by the steric and electronic properties of the olefin, allowing for the precise installation of diverse side chains. Finally, an intramolecular nucleophilic attack by the nitrogen atom followed by reductive elimination releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst, closing the loop for continuous turnover.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The well-defined organometallic pathway minimizes the formation of polymeric byproducts and random coupling artifacts that often plague less controlled reactions. The use of triethylamine as an additive further assists in neutralizing acidic byproducts and maintaining the catalytic cycle's integrity. Experimental data from the patent indicates that this system tolerates a wide array of substituents on the olefin, including electron-withdrawing halogens and electron-donating alkyl groups, without significant loss in yield. For instance, substrates bearing para-methyl, para-chloro, or even bulky cyclohexyl groups successfully convert to the desired products. This broad substrate compatibility ensures that the impurity profile remains manageable, simplifying the purification process and ensuring the delivery of high-purity heterocyclic compounds essential for downstream pharmaceutical applications.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The execution of this synthesis is designed for practicality, utilizing reagents that are readily accessible from global chemical suppliers. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in a solvent like 1,4-dioxane. The mixture is then heated to 110°C for a duration of 24 to 48 hours, allowing the carbonylation and cyclization to reach completion. Post-reaction workup is straightforward, involving filtration to remove insoluble palladium residues and silica gel treatment, followed by standard column chromatography. This simplicity underscores the method's potential for rapid adoption in process chemistry labs. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized guide below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and olefin in an organic solvent such as dioxane.
- Heat the reaction mixture to 110°C and stir continuously for 24 to 48 hours under inert atmosphere to facilitate the carbonylation cyclization.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic methodology offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of gaseous carbon monoxide removes a major logistical and safety bottleneck, allowing facilities to operate with greater flexibility and reduced insurance overheads. Moreover, the reliance on stable, solid reagents enhances supply chain reliability, as these materials are easier to store, transport, and inventory compared to compressed gases. This stability translates directly into reduced lead time for high-purity pharmaceutical intermediates, as production schedules are no longer subject to the complexities of gas cylinder logistics or specialized reactor availability. The robustness of the reaction conditions also implies a lower rate of batch failures, ensuring a consistent flow of materials to downstream formulation teams.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the simplification of the reaction infrastructure. By obviating the need for high-pressure autoclaves and specialized gas handling systems, manufacturers can achieve significant capital expenditure savings. Additionally, the use of a solid CO surrogate improves atom economy and reduces waste disposal costs associated with scrubbing toxic gases. The high conversion rates observed across various substrates mean that raw material utilization is maximized, further driving down the cost per kilogram of the final API intermediate. These factors combine to create a leaner, more cost-effective manufacturing process that aligns with the industry's push for sustainable and economical production methods.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including the palladium catalyst, dppp ligand, and various olefins, are commodity chemicals available from multiple global vendors. This multi-sourcing capability mitigates the risk of supply disruptions that can occur with proprietary or single-source reagents. Furthermore, the reaction's tolerance to moisture and air (to a reasonable extent within the Schlenk technique context) reduces the sensitivity of the process, making it more robust against minor variations in plant conditions. This resilience ensures that production timelines remain intact even when facing minor operational fluctuations, providing a dependable source of critical intermediates for long-term drug development projects.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental challenges, but this method is inherently greener due to the absence of toxic gas emissions. The solid nature of the CO source prevents accidental releases into the workplace environment, enhancing operator safety and simplifying regulatory compliance. The solvent system, primarily using dioxane, is well-understood and can be efficiently recovered and recycled in modern facilities. As the industry moves towards stricter environmental standards, adopting a process that minimizes hazardous waste and energy consumption positions manufacturers favorably for future audits. The demonstrated ability to scale this reaction from milligram to gram levels in the patent suggests a clear path toward ton-scale production with minimal re-optimization.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. Understanding these nuances is vital for process chemists evaluating the feasibility of integrating this route into their existing portfolios. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the advantages of using 1,3,5-trimesic acid phenol ester over CO gas?
A: Using 1,3,5-trimesic acid phenol ester as a solid carbon monoxide surrogate eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for industrial scale-up.
Q: What is the substrate scope for the olefin component in this synthesis?
A: The method demonstrates excellent compatibility with various olefins, including substituted aryl groups (methyl, chloro, fluoro), linear and branched alkyl chains, and silyl-substituted alkenes, yielding products with 60-88% efficiency.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the protocol utilizes commercially available reagents and standard solvents like dioxane, operates at moderate temperatures (110°C), and avoids toxic gases, making it highly viable for commercial scale-up of complex intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
The technological advancements described in patent CN112239456B highlight the evolving landscape of heterocyclic synthesis, where safety and efficiency converge. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging such cutting-edge methodologies to deliver superior chemical solutions. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand that the consistency of your supply chain is paramount to your own commercial success, and we are equipped to meet those demands with precision.
We invite you to explore how our expertise in palladium-catalyzed carbonylation can accelerate your drug development timeline. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecular targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced synthetic capabilities can become the cornerstone of your supply chain strategy, delivering value through innovation and reliability.
