Advanced Palladium-Catalyzed Synthesis of Pyrrolinone Intermediates for High-Purity Pharmaceutical Manufacturing at Commercial Scale
The patent CN112694430B discloses a groundbreaking methodology for the preparation of structurally significant pyrrolinone intermediates through an innovative palladium-catalyzed carbonylation process that addresses longstanding challenges in heterocyclic synthesis within the pharmaceutical sector This novel approach represents a substantial advancement over existing techniques by enabling direct access to these critical molecular scaffolds which are prevalent in numerous bioactive compounds including antibiotics such as althiomycin and antidiabetic agents like glimepiride The methodology leverages readily available starting materials and operates under mild reaction conditions that significantly enhance operational safety while maintaining exceptional substrate tolerance across diverse functional groups Furthermore the one-step nature of this transformation eliminates multiple purification stages required by conventional routes thereby reducing both time and resource expenditure throughout the manufacturing workflow This patent therefore establishes a robust foundation for scalable production of high-value intermediates that are indispensable in modern drug discovery and development pipelines The technical elegance of this process lies in its strategic use of carbon monoxide surrogates which circumvent handling hazards associated with gaseous CO while preserving catalytic efficiency through carefully engineered ligand systems that stabilize the active palladium species throughout the reaction cycle
The Limitations of Conventional Methods vs The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing pyrrolinone frameworks typically involve multi-step sequences requiring harsh reaction conditions such as high-pressure carbon monoxide environments which introduce significant safety hazards and necessitate specialized equipment that increases capital expenditure These conventional methods often suffer from poor functional group tolerance particularly with halogenated or sterically hindered substrates leading to reduced yields and complex purification challenges that compromise product purity Furthermore extended reaction times exceeding seventy-two hours are frequently required to achieve acceptable conversion rates resulting in inefficient resource utilization and elevated operational costs The reliance on stoichiometric reagents rather than catalytic systems generates substantial waste streams that complicate environmental compliance and increase disposal costs while limited scalability hinders transition from laboratory discovery to commercial manufacturing volumes These inherent drawbacks collectively restrict the practical application of existing methodologies despite the critical importance of pyrrolinone scaffolds in pharmaceutical development
The Novel Approach
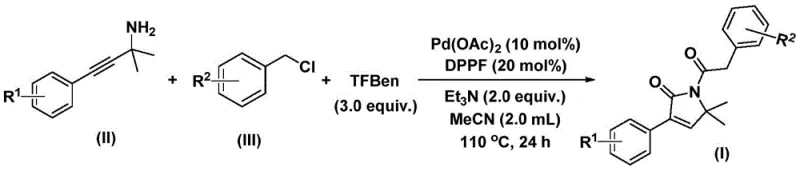
The patented methodology overcomes these limitations through a strategically designed palladium-catalyzed bis-carbonylation process that operates under mild conditions using carbon monoxide surrogates instead of hazardous gaseous CO thereby eliminating high-pressure requirements while maintaining excellent reaction efficiency This innovative approach utilizes commercially available starting materials including propargyl amines and benzyl chlorides which demonstrate broad substrate compatibility across diverse functional groups such as halogens alkyls and aryls without requiring protective group manipulations The one-step transformation achieves complete conversion within twenty-four hours at one hundred ten degrees Celsius significantly reducing processing time compared to conventional multi-step sequences while generating minimal byproducts through precise catalytic control The simplified workup procedure involving filtration through silica gel followed by standard column chromatography ensures high product purity without complex separation techniques making this method exceptionally suitable for scalable pharmaceutical manufacturing operations
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of benzyl chloride into the palladium zero species forming a benzylpalladium intermediate which subsequently undergoes insertion of carbon monoxide liberated from triethylbenzene tricarboxylate to generate an acylpalladium complex This key intermediate then adds across the alkyne functionality of propargyl amine initiating cyclization through nucleophilic attack by the amine group forming a five-membered ring palladacycle A second carbon monoxide insertion event expands this structure into a six-membered ring intermediate before reductive elimination releases the final pyrrolinone product while regenerating the active palladium catalyst The precise coordination geometry enforced by the DPPF ligand prevents undesired β-hydride elimination pathways ensuring high regioselectivity toward the desired five-membered ring structure Critical to this mechanism is the controlled release of carbon monoxide from the phenol ester surrogate which maintains optimal CO concentration throughout the reaction preventing catalyst deactivation while enabling efficient carbonyl incorporation
Impurity control is achieved through multiple mechanistic safeguards including the selective formation of benzylpalladium species that minimizes homocoupling side reactions while the ligand-controlled insertion steps prevent regioisomer formation The use of triethylamine base effectively scavenges hydrochloric acid byproducts preventing acid-mediated decomposition pathways that could generate dehydrogenated impurities The optimized solvent system comprising acetonitrile provides ideal polarity for stabilizing charged intermediates without promoting solvolysis side reactions while maintaining excellent solubility for all reaction components throughout the process The absence of transition metal residues in final products is ensured by thorough silica gel filtration which captures palladium species before chromatographic purification further enhancing product purity profiles required for pharmaceutical applications
How to Synthesize Pyrrolinone Intermediate Efficiently
This patented methodology provides a streamlined pathway for producing high-purity pyrrolinone intermediates through a single-reaction sequence that eliminates multiple isolation steps required by conventional approaches The process leverages commercially accessible starting materials including propargyl amines benzyl chlorides and carbon monoxide surrogates which can be readily sourced from established chemical suppliers ensuring consistent raw material availability The carefully optimized reaction conditions operate within a narrow temperature window that maximizes conversion while minimizing decomposition pathways making this method particularly suitable for implementation in standard pharmaceutical manufacturing facilities Detailed standardized synthesis procedures including precise reagent quantities temperature profiles and purification protocols are provided below to facilitate seamless technology transfer from laboratory scale to commercial production environments
- Combine palladium acetate catalyst at ten mol percent loading with DPPF ligand at twenty mol percent along with triethylamine base at two equivalents and TFBen carbon monoxide substitute at three equivalents in acetonitrile solvent under inert atmosphere before introducing propargyl amine and benzyl chloride substrates
- Heat the homogeneous reaction mixture to one hundred ten degrees Celsius and maintain this temperature for twenty four hours under controlled conditions to facilitate complete bis-carbonylation through sequential palladium insertion and carbon monoxide transfer steps
- Execute post-reaction processing by filtration through silica gel followed by column chromatography purification using standard elution protocols to isolate high-purity pyrrolinone intermediate while removing residual catalysts and byproducts
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals through its strategic design that prioritizes operational simplicity material accessibility and scalability The elimination of hazardous reagents and specialized equipment requirements significantly reduces both capital investment needs and regulatory compliance burdens associated with traditional multi-step syntheses while enhancing overall process safety profiles The utilization of commercially abundant starting materials ensures stable supply chains with minimal risk of raw material shortages or price volatility commonly encountered with specialized reagents required by conventional routes Furthermore the streamlined single-reaction protocol enables faster production cycles that directly translate to improved responsiveness to fluctuating market demands while maintaining stringent quality standards essential for pharmaceutical applications
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and hazardous reagents inherent in traditional synthetic pathways substantially reduces raw material expenditures while simplifying waste treatment protocols through inherently cleaner reaction profiles that minimize byproduct formation during the carbonylation sequence The one-step nature of this transformation also reduces labor costs associated with multiple intermediate isolations and purifications while decreasing solvent consumption through optimized reaction stoichiometry that enhances overall process efficiency without requiring expensive specialized equipment
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials including standard palladium catalysts propargyl amines and benzyl chlorides ensures consistent sourcing from multiple global suppliers thereby mitigating single-source dependency risks Commonly used solvents like acetonitrile maintain stable supply chains with minimal price fluctuations while simplified logistics requirements reduce lead times associated with specialized reagent procurement The robust nature of this methodology accommodates minor variations in raw material quality without compromising final product specifications further strengthening supply chain resilience
- Scalability and Environmental Compliance: The straightforward scale-up potential from laboratory to commercial production is demonstrated through consistent yields across different batch sizes enabled by mild reaction conditions that do not require specialized high-pressure equipment The process generates minimal hazardous waste streams due to its catalytic nature and selective transformation pathways reducing environmental impact while simplifying regulatory compliance procedures Standard purification techniques using silica gel filtration and column chromatography are readily adaptable to large-scale manufacturing without requiring substantial process re-engineering thus facilitating rapid technology transfer from development to commercial production
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding this patented methodology based on detailed analysis of its implementation parameters performance characteristics and practical applications within pharmaceutical manufacturing environments These answers derive directly from experimental data presented in patent CN112694430B ensuring accurate representation of both technical capabilities and commercial advantages associated with this innovative synthesis approach
Q: What limitations do conventional methods face in synthesizing pyrrolinone intermediates?
A: Conventional synthetic approaches typically require multi-step sequences with harsh reaction conditions including high-pressure carbon monoxide handling which increases operational hazards while exhibiting poor substrate tolerance that limits applicability across diverse functional groups essential for pharmaceutical applications
Q: How does this novel method achieve superior substrate compatibility?
A: The palladium-catalyzed bis-carbonylation process demonstrates exceptional functional group tolerance through carefully optimized ligand systems that stabilize reactive intermediates enabling successful conversion across halogenated alkyl and aryl substituents without requiring protective group strategies
Q: What commercial advantages does this one-step synthesis offer supply chain teams?
A: The streamlined single-reaction protocol significantly reduces manufacturing complexity by eliminating intermediate isolation steps while utilizing commercially available starting materials that enhance supply chain resilience through simplified sourcing and reduced lead times
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolinone Intermediate Supplier
Our patented methodology represents a significant advancement in pyrrolinone intermediate synthesis that combines cutting-edge catalytic chemistry with practical manufacturing considerations essential for modern pharmaceutical production NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through our state-of-the-art facilities equipped with rigorous QC labs capable of meeting global regulatory standards Our technical team specializes in adapting complex synthetic routes like this palladium-catalyzed carbonylation process to industrial-scale operations ensuring seamless technology transfer from laboratory discovery to full commercial implementation without compromising product quality or yield consistency
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements by contacting our technical procurement team who can provide detailed COA data route feasibility assessments and scalability projections based on your target production volumes This collaborative approach enables us to develop optimized solutions that maximize both technical performance and economic efficiency while ensuring reliable supply chain continuity for your critical pharmaceutical intermediates
