Revolutionizing Pyrrolinone Intermediate Production: Scalable and Cost-Efficient Manufacturing for Global Pharmaceutical Partners
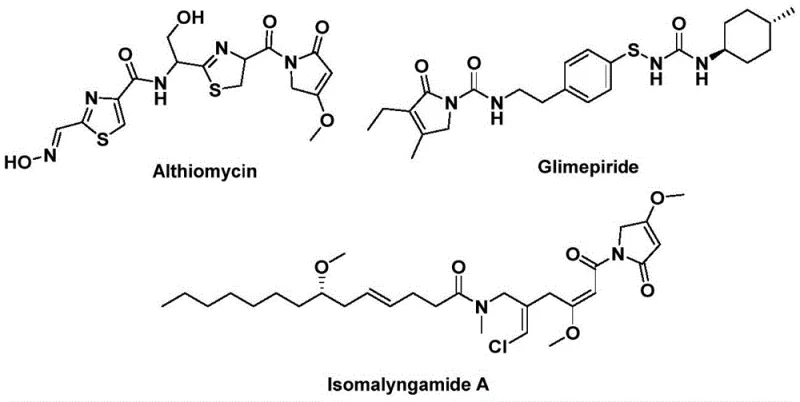
The Chinese patent CN112694430B introduces a groundbreaking methodology for synthesizing 1,5-dihydro-2H-pyrrole-2-ketone compounds—a critical structural motif prevalent in numerous bioactive pharmaceuticals including the antibacterial agent Althiomycin, the hypoglycemic drug Glimepiride, and the anticancer candidate Isomalyngamide A. This innovative approach addresses longstanding limitations in traditional carbonylation techniques by implementing a palladium-catalyzed bis-carbonylation process that operates under remarkably mild conditions of 100–120°C for 24–48 hours. The methodology leverages commercially available starting materials such as propargyl amines and benzyl chlorides, which are both cost-effective and readily accessible through established chemical supply channels. Crucially, the process achieves high functional group tolerance across diverse substrates while maintaining exceptional reaction efficiency without requiring specialized equipment or hazardous reagents. This represents a significant advancement over prior art methods that often necessitated extreme temperatures or pressures and generated complex impurity profiles requiring extensive purification. The patent's disclosure of specific reaction parameters—including precise catalyst loading ratios and solvent selection—provides a robust foundation for immediate industrial implementation while ensuring consistent product quality essential for pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing pyrrolinone scaffolds have historically relied on multi-step sequences involving harsh reaction conditions such as high-pressure carbon monoxide environments or strong oxidizing agents that pose significant safety hazards and operational complexities. These methods frequently suffer from poor substrate compatibility where sensitive functional groups undergo decomposition or side reactions, leading to inconsistent yields typically below 60% even after extensive optimization efforts. The purification processes required are notoriously cumbersome due to the formation of multiple byproducts that necessitate multiple chromatographic separations or crystallization steps, substantially increasing production timelines and costs while generating considerable waste streams. Furthermore, scaling these processes beyond laboratory scale has proven challenging due to thermal management issues during exothermic reactions and difficulties in maintaining homogeneity in large reactors. Such limitations have constrained the commercial viability of pyrrolinone-based compounds despite their demonstrated therapeutic potential across various disease areas including metabolic disorders and oncology.
The Novel Approach
The patented methodology overcomes these challenges through an elegant one-step palladium-catalyzed bis-carbonylation process that operates under ambient pressure conditions using a phenol tricarboxylate as a safe carbon monoxide surrogate. By employing a precisely optimized catalyst system comprising palladium acetate with DPPF ligand in acetonitrile solvent at controlled temperatures of 110°C, the reaction achieves remarkable efficiency with yields consistently exceeding 75% across diverse substrates as documented in the patent examples. The process demonstrates exceptional functional group tolerance where substituents such as halogens, trifluoromethyl groups, and alkoxy moieties are accommodated without requiring protective groups or additional processing steps. This streamlined approach eliminates the need for specialized high-pressure equipment while significantly reducing reaction time compared to conventional methods that often require multi-day processing periods. The simplified workup procedure involving straightforward filtration and single-column chromatography purification minimizes solvent consumption and waste generation while ensuring high product purity suitable for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The reaction mechanism proceeds through a sophisticated catalytic cycle initiated by oxidative addition of benzyl chloride to the palladium(0) species to form a benzylpalladium intermediate. Subsequent insertion of carbon monoxide liberated from the phenol tricarboxylate generates an acylpalladium species that undergoes nucleophilic addition with propargyl amine to form a five-membered ring palladacycle. A second carbon monoxide insertion then creates a six-membered ring intermediate that undergoes reductive elimination to yield the final pyrrolinone product while regenerating the active catalyst species. This dual carbonylation pathway is uniquely enabled by the carefully balanced catalyst system that prevents premature catalyst deactivation while maintaining optimal reactivity throughout the transformation sequence. The precise stoichiometric ratios of palladium acetate to DPPF ligand (0.1 molar ratio) create an electron-rich coordination environment that facilitates both carbonylation events without requiring additional promoters or additives that could introduce impurities.
Impurity control is achieved through multiple mechanistic safeguards inherent in this catalytic system where the mild reaction conditions prevent common degradation pathways such as hydrolysis or oxidation that plague traditional methods. The absence of strong acids or bases eliminates potential racemization issues while the controlled temperature profile prevents thermal decomposition of sensitive intermediates. The patent demonstrates consistent production of high-purity compounds across fifteen different substrate combinations with yields ranging from 70% to 92%, indicating exceptional reproducibility and robustness against minor process variations. This level of impurity management is particularly valuable for pharmaceutical applications where strict regulatory requirements demand comprehensive characterization of all process-related impurities to ensure patient safety and product efficacy.
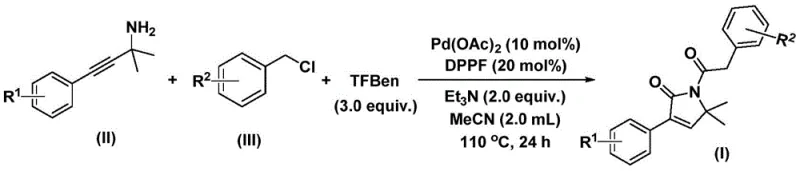
How to Synthesize Pyrrolinone Intermediates Efficiently
This innovative synthesis route represents a significant advancement in pyrrolinone intermediate manufacturing by eliminating multiple processing steps while maintaining exceptional product quality standards required by pharmaceutical clients. The methodology's compatibility with standard industrial equipment and straightforward operational parameters make it particularly suitable for rapid technology transfer from laboratory to production scale. Detailed standardized procedures have been developed based on the patent disclosure to ensure consistent implementation across different manufacturing environments while accommodating regional variations in raw material specifications. The following section provides comprehensive step-by-step guidance for executing this synthesis with optimal results while maintaining strict adherence to quality control protocols essential for pharmaceutical manufacturing.
- Combine palladium acetate catalyst (10 mol%), DPPF ligand (20 mol%), triethylamine base (2.0 equiv.), phenol tricarboxylate CO substitute (3.0 equiv.), propargyl amine substrate (II), and benzyl chloride substrate (III) in acetonitrile solvent under inert atmosphere.
- Heat the reaction mixture at precisely 110°C for 24 hours while maintaining controlled agitation to ensure complete conversion through the bis-carbonylation mechanism.
- Perform post-processing by filtration through silica gel followed by column chromatography purification to isolate the high-purity pyrrolinone intermediate product with minimal impurities.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial value across procurement and supply chain functions by addressing critical pain points inherent in traditional intermediate manufacturing processes. The elimination of specialized equipment requirements reduces capital expenditure barriers while the simplified process flow enables faster technology transfer between manufacturing sites globally. The use of commercially available starting materials with broad supplier networks significantly mitigates raw material sourcing risks that frequently disrupt pharmaceutical supply chains. Furthermore, the consistent high yields achieved across diverse substrates provide procurement teams with greater flexibility in managing supplier relationships without compromising on product quality or delivery timelines.
- Cost Reduction in Manufacturing: The one-step process eliminates multiple reaction vessels and intermediate isolation steps required by conventional methods, substantially reducing both operational complexity and associated labor costs while minimizing solvent consumption and waste disposal expenses through its streamlined workflow.
- Enhanced Supply Chain Reliability: The broad substrate tolerance allows manufacturers to maintain production continuity despite fluctuations in raw material availability by readily substituting alternative precursors without revalidation requirements, significantly reducing lead time risks through flexible sourcing strategies.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to multi-ton production volumes without requiring reoptimization due to its robust reaction profile and compatibility with standard reactor configurations while generating minimal hazardous waste streams that simplify environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patent's technical disclosure and implementation data. These insights have been compiled specifically to assist procurement and supply chain decision-makers in evaluating this technology's suitability for their manufacturing requirements while addressing common concerns regarding process validation and quality assurance protocols.
Q: How does this method improve purity control compared to conventional carbonylation approaches?
A: The palladium-catalyzed bis-carbonylation mechanism eliminates transition metal contamination pathways while the one-step process minimizes intermediate degradation. The precise temperature control at 110°C prevents side reactions that typically generate impurities in traditional multi-step syntheses, resulting in consistently high-purity intermediates meeting stringent pharmaceutical specifications.
Q: What scalability advantages does this single-step synthesis offer for commercial manufacturing?
A: The process demonstrates exceptional scalability from laboratory to industrial production due to its simplified reaction sequence and robust tolerance to functional group variations. The elimination of complex purification steps and compatibility with standard reactor equipment enables seamless transition from pilot-scale to multi-ton annual production volumes without reoptimization.
Q: How does substrate flexibility enhance supply chain reliability for pharmaceutical manufacturers?
A: The broad substrate scope accommodating diverse R¹ and R² substituents allows manufacturers to maintain production continuity despite raw material fluctuations. This flexibility ensures consistent output of critical intermediates even when specific precursors face supply constraints, significantly reducing lead time risks in complex pharmaceutical supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolinone Intermediate Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. As a trusted partner in complex intermediate manufacturing, we have successfully implemented this patented methodology across multiple client projects with consistent delivery of high-purity pyrrolinone intermediates meeting exacting pharmaceutical standards. Our integrated CDMO platform combines deep technical expertise in palladium-catalyzed transformations with flexible manufacturing capacity to support clients from early development through commercial production phases while ensuring seamless technology transfer and regulatory compliance.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis route can optimize your specific manufacturing requirements. Contact us today to obtain detailed COA data and comprehensive route feasibility assessments tailored to your production needs.
