Advanced Catalytic Route for High-Purity 3-Trifluoromethyl-1,2,4-Triazole Pharmaceutical Intermediates
Introduction to Next-Generation Triazole Synthesis
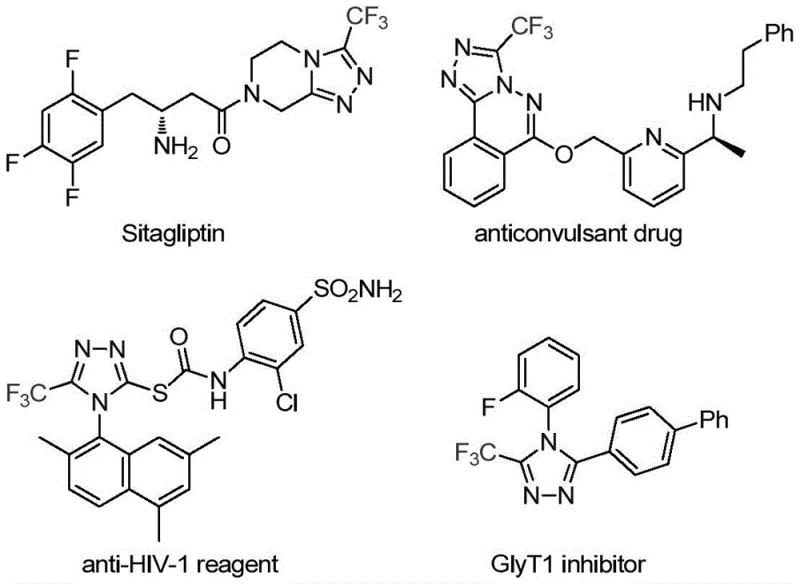
The pharmaceutical industry continuously seeks robust and efficient pathways to construct nitrogen-containing heterocycles, particularly those incorporating fluorine atoms which are pivotal for enhancing metabolic stability and bioavailability. Patent CN113307778A introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic methodologies. This technology leverages a synergistic molybdenum and copper co-catalytic system to facilitate the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. The significance of this advancement cannot be overstated, as the 1,2,4-triazole scaffold is a privileged structure found in numerous blockbuster drugs, including Sitagliptin and various antifungal agents. By enabling the direct installation of the trifluoromethyl group under mild conditions, this process offers a streamlined route to high-value pharmaceutical intermediates that are essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that present significant safety and operational challenges for large-scale manufacturing. Traditional routes often involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinone, processes that frequently require harsh reaction conditions and generate substantial chemical waste. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane pose severe safety risks due to the explosive nature of diazo compounds, making them unsuitable for industrial scale-up. These legacy methods often suffer from limited substrate tolerance, poor atom economy, and the necessity for expensive or difficult-to-remove catalysts, which collectively drive up the cost of goods and complicate the supply chain for critical API intermediates.
The Novel Approach
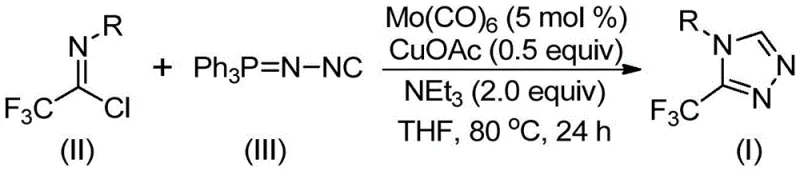
In stark contrast, the novel approach detailed in the patent utilizes a transition metal co-catalysis strategy that operates under remarkably mild and controlled conditions. By employing molybdenum hexacarbonyl and cuprous acetate in the presence of triethylamine, the reaction proceeds efficiently at temperatures between 70°C and 90°C, eliminating the need for hazardous high-energy reagents. This method utilizes trifluoroethylimidoyl chloride and functionalized isonitriles as stable, commercially available starting materials, thereby enhancing the safety profile and reliability of the supply chain. The reaction mechanism involves a copper-promoted [3+2] cycloaddition followed by the elimination of triphenylphosphine oxide, resulting in high yields and exceptional purity profiles. This shift from dangerous diazo chemistry to a stable imidoyl chloride-based protocol represents a paradigm shift in how these valuable heterocycles are manufactured.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The core innovation of this technology lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate the formation of the triazole ring with high precision. Mechanistically, the molybdenum hexacarbonyl serves as a crucial activator for the functionalized isonitrile, forming a reactive metal-isocyanide complex that primes the molecule for nucleophilic attack. Simultaneously, the cuprous acetate facilitates the activation of the trifluoroethylimidoyl chloride, lowering the energy barrier for the subsequent cycloaddition step. This dual-catalyst system ensures that the reaction proceeds through a concerted [3+2] pathway to form a five-membered ring intermediate, which then undergoes hydrolysis to release the final triazole product. The presence of molecular sieves in the reaction mixture further drives the equilibrium forward by sequestering water generated during the process, thereby maximizing conversion rates and minimizing hydrolytic side products.
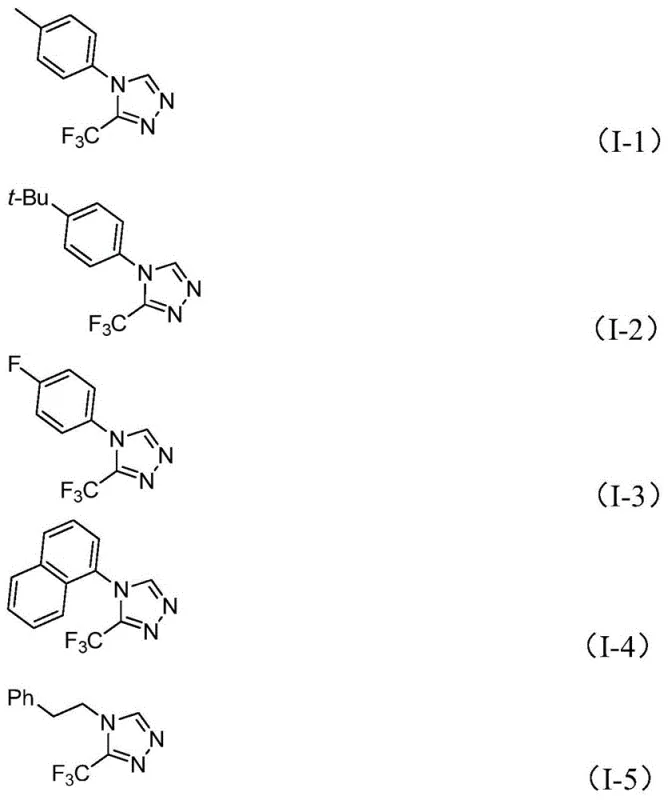
From an impurity control perspective, the mildness of this catalytic system is a distinct advantage for producing high-purity pharmaceutical intermediates. Unlike harsh acidic or basic conditions that can lead to decomposition of sensitive functional groups, this neutral to slightly basic environment preserves the integrity of diverse substituents on the aromatic ring. The patent data demonstrates excellent tolerance for electron-withdrawing groups such as nitro and halogens, as well as electron-donating alkyl and alkoxy groups, without significant formation of by-products. This broad functional group compatibility allows medicinal chemists to access a wide library of analogues for structure-activity relationship (SAR) studies using a single, robust platform. The ability to synthesize compounds like I-1 through I-5 with high fidelity ensures that downstream purification is simplified, reducing the overall processing time and solvent consumption.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and reaction parameters to achieve optimal results. The process begins with the careful charging of molybdenum hexacarbonyl, cuprous acetate, triethylamine, and molecular sieves into a dry reaction vessel under an inert atmosphere. Following the addition of the organic solvent, preferably THF for its superior solvation properties, the trifluoroethylimidoyl chloride and functionalized isonitrile are introduced. The detailed standardized synthesis steps, including specific mixing sequences, temperature ramping profiles, and quenching protocols, are outlined in the guide below to ensure reproducibility and safety.
- Combine molybdenum hexacarbonyl, cuprous acetate, triethylamine, molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The transition to this method addresses several pain points associated with the sourcing and manufacturing of complex heterocyclic intermediates, particularly regarding cost stability and vendor reliability. By shifting away from proprietary or hazardous reagents towards commodity chemicals, manufacturers can mitigate the risk of supply disruptions and price volatility that often plague the fine chemical sector.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive noble metal catalysts with abundant and inexpensive transition metals like copper and molybdenum. This fundamental change in the catalyst system drastically reduces the raw material cost per kilogram of product. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure or cryogenic equipment, allowing production to occur in standard glass-lined reactors, which lowers capital expenditure and operational overheads. The simplified workup procedure, which avoids complex extraction sequences, also contributes to significant savings in labor and solvent recovery costs.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically trifluoroethylimidoyl chloride and functionalized isonitriles, are derived from widely available bulk chemicals, ensuring a stable and continuous supply stream. Unlike methods relying on unstable diazo compounds that require on-site generation and immediate consumption, the reagents in this protocol are shelf-stable and can be sourced from multiple global suppliers. This diversification of the supply base reduces dependency on single-source vendors and enhances the resilience of the manufacturing schedule against external market shocks or logistical delays.
- Scalability and Environmental Compliance: The protocol has been validated for gram-scale synthesis with high efficiency, indicating a clear path toward commercial scale-up without the need for extensive re-optimization. The use of THF as a primary solvent aligns with standard industry practices for solvent recovery and recycling, facilitating compliance with environmental regulations regarding volatile organic compound (VOC) emissions. Additionally, the absence of heavy metal contaminants in the final product simplifies the purification process, reducing the burden on wastewater treatment facilities and ensuring that the final API intermediate meets stringent regulatory specifications for residual metals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: This method utilizes mild reaction conditions (70-90°C) and avoids hazardous reagents like diazonium salts often used in traditional routes. It employs cheap and readily available catalysts (Mo(CO)6 and CuOAc) instead of expensive noble metals, significantly reducing raw material costs and simplifying post-reaction metal removal processes.
Q: What is the substrate scope for the R group in this trifluoromethyl triazole synthesis?
A: The process demonstrates excellent tolerance for various substituents on the aryl ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, and nitro. It also accommodates phenethyl groups, allowing for the synthesis of diverse derivatives suitable for different medicinal chemistry campaigns.
Q: Is this synthetic route scalable for commercial production of API intermediates?
A: Yes, the patent explicitly states that the method can be expanded to gram-level reactions with high efficiency. The use of standard organic solvents like THF and simple workup procedures involving filtration and column chromatography indicates strong potential for scale-up to kilogram or tonne levels in a GMP environment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the marketplace. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with our technical team to evaluate how this innovative synthetic route can optimize your specific project requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic method. We encourage you to contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis needs, securing a competitive advantage in your supply chain.
