Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for nitrogen-containing heterocycles, particularly those incorporating trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses critical bottlenecks in current manufacturing processes. This technology leverages a non-metallic iodine-promoted strategy to construct the triazole core efficiently, bypassing the need for toxic heavy metal catalysts often required in traditional cross-coupling reactions. The significance of this scaffold is underscored by its presence in blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, where the triazole ring serves as a pivotal pharmacophore. By utilizing readily available aryl ethyl ketones and trifluoroethylimide hydrazides, this innovation offers a streamlined route to high-value intermediates essential for modern drug discovery and development pipelines.
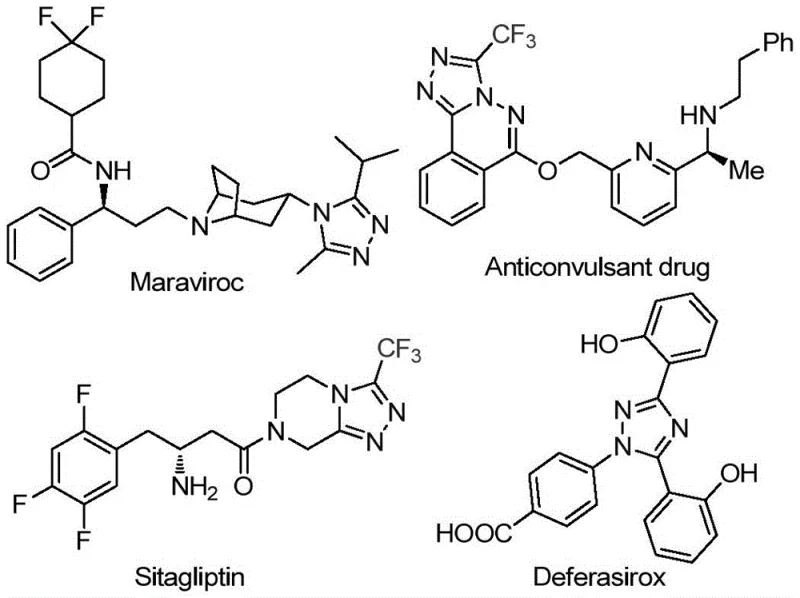
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazoles, especially those bearing both acyl and trifluoromethyl functionalities, has relied on complex multistep sequences or transition metal catalysis. Conventional protocols often necessitate the use of expensive palladium or copper catalysts, which introduce significant challenges regarding residual metal limits in final Active Pharmaceutical Ingredients (APIs). Furthermore, many existing methods require stringent anhydrous and oxygen-free environments, demanding specialized equipment and increasing operational expenditures. The reliance on harsh conditions not only elevates safety risks but also limits the functional group tolerance, preventing the synthesis of diverse analogues required for Structure-Activity Relationship (SAR) studies. These factors collectively contribute to prolonged lead times and inflated production costs, creating a substantial barrier for reliable pharmaceutical intermediate suppliers aiming to deliver cost-effective solutions to global markets.
The Novel Approach
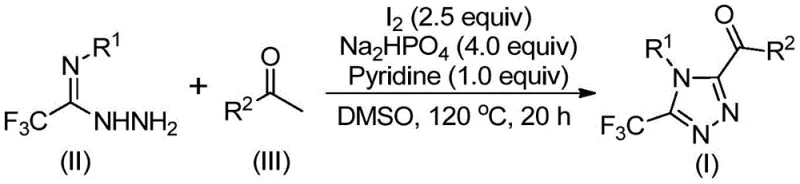
In stark contrast, the methodology outlined in CN113105402B introduces a remarkably simple yet powerful iodine-mediated cyclization strategy. This novel approach utilizes elemental iodine and dimethyl sulfoxide (DMSO) to facilitate an iodination and Kornblum oxidation sequence in situ, generating reactive intermediates that spontaneously cyclize with hydrazides. The process operates under relatively mild thermal conditions without the need for inert atmosphere protection, drastically simplifying the operational workflow. By eliminating transition metals entirely, the method inherently produces cleaner crude products, reducing the burden on downstream purification processes. This metal-free paradigm shift not only aligns with green chemistry principles but also ensures that the resulting 3,4,5-trisubstituted 1,2,4-triazole compounds meet the rigorous purity specifications demanded by regulatory bodies for clinical applications.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation steps driven by the unique properties of the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes alpha-iodination followed by Kornblum oxidation to form an aryl diketone species within the reaction matrix. This highly electrophilic intermediate then engages in a dehydration condensation with the trifluoroethylimide hydrazide to generate a hydrazone intermediate. Subsequently, under the synergistic promotion of iodine and the added base system (sodium dihydrogen phosphate and pyridine), an intramolecular cyclization occurs to close the triazole ring. This cascade reaction effectively installs the trifluoromethyl group at the 3-position and the acyl group at the 5-position simultaneously, a structural motif that is notoriously difficult to access via other means. The precise control over regioselectivity ensures that the desired 3,4,5-substitution pattern is achieved with high fidelity, minimizing the formation of regioisomeric impurities that could complicate isolation.
From an impurity control perspective, the choice of reagents plays a critical role in maintaining product quality. The use of sodium dihydrogen phosphate acts as a mild buffer, preventing the degradation of sensitive functional groups while facilitating the elimination of hydrogen iodide byproducts. Pyridine serves as an auxiliary base to drive the equilibrium towards product formation. Because the reaction does not involve radical initiators or strong oxidants beyond the controlled iodine-DMSO system, side reactions such as over-oxidation or polymerization are significantly suppressed. This inherent selectivity translates to a cleaner reaction profile, allowing for straightforward workup procedures involving simple filtration and silica gel chromatography. For R&D teams, understanding this mechanism provides confidence in the robustness of the process when scaling up to pilot plant quantities, ensuring consistent batch-to-batch reproducibility.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The execution of this synthesis protocol is designed for maximum operational simplicity, making it accessible for both laboratory research and industrial manufacturing settings. The procedure begins with the activation of the ketone substrate in DMSO, followed by the sequential addition of the hydrazide component and promoters. Detailed standard operating procedures regarding stoichiometry, temperature ramping, and quenching strategies are critical for optimizing yield and purity. The following guide outlines the standardized synthesis steps derived from the patent data to ensure successful replication of this high-value transformation.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate iodination and oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-promoted synthesis route offers tangible strategic advantages that directly impact the bottom line and operational resilience. The primary benefit lies in the drastic simplification of the raw material portfolio; aryl ethyl ketones and elemental iodine are commodity chemicals available from multiple global vendors, mitigating the risk of single-source supply disruptions. Unlike processes dependent on scarce noble metals, this method insulates the supply chain from the volatile pricing fluctuations associated with precious metal catalysts. Furthermore, the elimination of heavy metals removes the need for expensive scavenging resins and specialized analytical testing for metal residues, resulting in substantial cost savings in the quality control and purification stages. These efficiencies collectively enhance the overall economic viability of producing complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of expensive catalytic systems with inexpensive stoichiometric reagents. By avoiding palladium or copper catalysts, manufacturers eliminate a major cost center associated with both the initial purchase of the catalyst and the subsequent removal steps required to meet ppm-level specifications. Additionally, the use of DMSO as a solvent, which acts as both the reaction medium and the oxidant, reduces the total volume of solvents required compared to multi-solvent systems. This consolidation of reagent functions leads to a leaner material bill and reduced waste disposal costs, driving down the overall cost of goods sold (COGS) for the final triazole intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of universally available starting materials that do not require long lead times or import licenses typically associated with controlled precursors. The robustness of the reaction conditions, which do not demand strictly anhydrous or oxygen-free environments, allows for production in a wider range of facilities without the need for specialized glovebox infrastructure. This flexibility enables decentralized manufacturing strategies, reducing logistics complexity and transportation risks. Consequently, partners can maintain higher inventory turnover rates and respond more agilely to fluctuating market demands for API precursors without being bottlenecked by fragile process requirements.
- Scalability and Environmental Compliance: The scalability of this method is evidenced by its successful demonstration at the gram level with a clear path to kilogram and tonne scales. The absence of toxic heavy metals simplifies environmental compliance, as wastewater treatment does not require complex heavy metal precipitation protocols. This aligns with increasingly stringent global environmental regulations, reducing the regulatory burden on manufacturing sites. The high atom economy of the cyclization step, combined with the ability to recycle solvent streams, supports sustainable manufacturing practices. This environmental stewardship not only protects the corporate reputation but also future-proofs the supply chain against tightening ecological legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production workflows.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a promoter instead of costly heavy metals like palladium or copper, significantly reducing raw material costs and simplifying downstream purification.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) solvent. It involves a two-stage heating process: initially at 90-110°C for ketone activation, followed by 110-130°C for the cyclization step with hydrazide derivatives.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method avoids strict anhydrous or oxygen-free conditions and uses cheap, commercially available starting materials, making it highly amenable to commercial scale-up from gram to multi-kilogram levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
As the demand for fluorinated heterocycles continues to surge in the development of next-generation therapeutics, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale supply. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest industry standards. We understand the critical nature of timeline adherence in drug development and are committed to delivering consistent quality that accelerates your path to clinical trials.
We invite you to collaborate with us to leverage this advanced iodine-promoted technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a stable, cost-effective supply of high-purity pharmaceutical intermediates that drive your innovation forward.
