Advanced One-Pot Synthesis of 3-Acyl Pyridines for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, which serve as the backbone for countless active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN107602452B, which discloses a highly efficient synthetic method for 3-acyl pyridine compounds. This technology represents a paradigm shift from traditional, labor-intensive routes to a streamlined, one-pot multi-step series reaction. By leveraging a copper-catalyzed oxidative cyclization strategy, this process transforms readily available enaminone compounds and α,β-saturated ketones or aldehydes into valuable 3-acyl pyridine derivatives. The significance of this innovation lies not only in its chemical elegance but also in its potential to redefine the supply chain dynamics for reliable pharmaceutical intermediate suppliers. As global demand for complex heterocyclic scaffolds increases, the ability to produce high-purity 3-acyl pyridines with minimal environmental impact becomes a critical competitive advantage for downstream drug manufacturers.

The limitations of conventional methods for synthesizing 3-acyl pyridines have long plagued process chemists and procurement teams alike. Traditional approaches often rely on multi-step sequences involving the preparation of sensitive intermediates, which require rigorous purification and handling under inert atmospheres. These legacy methods frequently suffer from poor atom economy, generating substantial amounts of chemical waste and driving up the environmental factor (E-factor) of the process. Furthermore, the reliance on expensive or difficult-to-source starting materials can create bottlenecks in the supply chain, leading to unpredictable lead times and inflated costs. In many cases, the harsh reaction conditions necessary to drive these transformations result in the formation of complex impurity profiles, necessitating costly downstream processing to meet the stringent purity specifications required for high-purity pharmaceutical intermediates. These cumulative inefficiencies make traditional routes less viable for modern, sustainability-focused manufacturing environments.
In stark contrast, the novel approach detailed in the patent offers a transformative solution by utilizing a direct, one-pot cascade reaction. This method employs a catalytic system comprising a copper salt, a nitrogen-based ligand, and a stable nitroxyl radical oxidant such as TEMPO. The reaction proceeds smoothly in common polar aprotic solvents like N,N-dimethylformamide (DMF) at moderate temperatures ranging from 110°C to 130°C. Crucially, the process tolerates a wide array of functional groups, including halogens, methoxy, and trifluoromethyl substituents, without the need for protecting group strategies. This versatility allows for the rapid generation of diverse chemical libraries, accelerating the drug discovery phase. Moreover, the use of air as a co-oxidant support alongside TEMPO enhances the green chemistry profile of the synthesis, aligning with global regulatory trends towards sustainable cost reduction in API manufacturing. The simplicity of the operation—essentially mixing reagents and heating—drastically reduces the operational complexity typically associated with heterocycle synthesis.
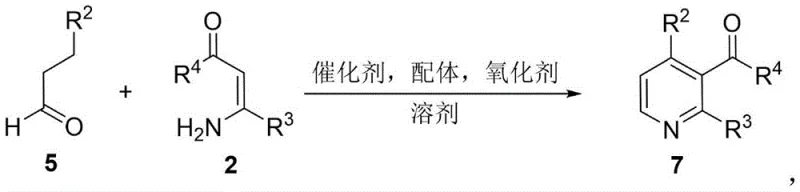
Mechanistically, this transformation is a sophisticated example of copper-catalyzed oxidative C-H functionalization and cyclization. The reaction initiates with the activation of the α,β-saturated ketone or aldehyde by the copper catalyst, likely forming an enolate or radical species that attacks the electron-deficient enaminone. The presence of the TEMPO oxidant is pivotal, facilitating the dehydrogenative aromatization required to form the stable pyridine ring. This oxidative step is carefully balanced to ensure complete conversion while minimizing over-oxidation of sensitive side chains. From an impurity control perspective, the one-pot nature of the reaction is advantageous because reactive intermediates are consumed in situ, preventing their accumulation and subsequent side reactions that often lead to hard-to-remove byproducts. The mild thermal conditions (110-130°C) further mitigate thermal degradation pathways, ensuring a cleaner crude reaction profile. For R&D directors, understanding this mechanism highlights the robustness of the process; the catalytic cycle is resilient enough to handle sterically hindered substrates, as evidenced by the successful synthesis of products with bulky cyclohexyl or substituted phenyl groups, ensuring consistent quality across different batches.
How to Synthesize 3-Acyl Pyridine Compounds Efficiently
To implement this synthesis effectively, process engineers should focus on optimizing the stoichiometric ratios of the catalyst system to balance cost and conversion efficiency. The patent suggests a molar ratio of substrate to catalyst to oxidant that maximizes yield while minimizing metal loading. Detailed standardized synthesis steps see the guide below.
- Dissolve the enaminone compound in a suitable polar aprotic solvent such as N,N-dimethylformamide (DMF).
- Sequentially add the alpha,beta-saturated ketone or aldehyde substrate, copper catalyst (e.g., Cu(OAc)2), ligand (e.g., bipyridine), and TEMPO oxidant.
- Heat the reaction mixture to 110-130°C under air atmosphere for approximately 20 hours, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology presents compelling economic and logistical benefits. The primary driver for cost reduction in manufacturing is the elimination of intermediate isolation steps. In traditional multi-step syntheses, each isolation involves solvent consumption, filtration, drying, and quality control testing, all of which add significant cost and time. By consolidating these steps into a single reactor vessel, the new method drastically reduces solvent usage and labor hours. Furthermore, the catalyst system utilizes copper salts, which are substantially cheaper than precious metals like palladium or platinum often used in cross-coupling reactions. This switch to base metal catalysis lowers the raw material cost baseline and simplifies the removal of residual metals from the final product, a critical requirement for pharmaceutical compliance.
- Cost Reduction in Manufacturing: The economic impact of this process extends beyond just reagent costs. The high atom economy means that a larger proportion of the starting mass ends up in the final product, reducing the cost of goods sold (COGS). Additionally, the use of air as a terminal oxidant support reduces the dependency on stoichiometric amounts of expensive chemical oxidants. The simplified workup procedure, involving basic aqueous quenching and extraction, minimizes the need for complex chromatography on a large scale, allowing for more cost-effective crystallization or distillation purification methods. This efficiency translates directly into improved margins for the final API producer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as starting materials. Enaminones and saturated ketones or aldehydes are widely available from bulk chemical suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous production schedules and meeting the strict delivery timelines demanded by multinational pharmaceutical clients. The ability to source raw materials locally in various regions further de-risks the supply chain against geopolitical disruptions.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by its homogeneous nature and moderate thermal requirements. The absence of hazardous reagents or extreme pressures simplifies the engineering controls needed for large-scale reactors. From an environmental standpoint, the reduced waste generation and lower E-factor align with increasingly strict environmental regulations globally. The process generates fewer hazardous byproducts, simplifying waste treatment and disposal protocols. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a key criterion for vendor selection in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for process evaluation.
Q: What are the primary advantages of this copper-catalyzed method over traditional pyridine synthesis?
A: This method utilizes a one-pot multi-step series reaction that avoids the isolation of unstable intermediates, significantly improving atom economy and reducing waste compared to traditional multi-step protocols requiring harsh conditions.
Q: Is this process suitable for large-scale commercial production of API intermediates?
A: Yes, the process employs commercially available raw materials like enaminones and saturated ketones, operates under relatively mild oxidative conditions (110-130°C), and uses air as a co-oxidant support, making it highly scalable for industrial applications.
Q: What is the substrate scope for the R1 and R4 groups in this synthesis?
A: The method demonstrates broad substrate tolerance, accommodating phenyl, substituted phenyl (with methyl, methoxy, halogens, trifluoromethyl), thienyl, cyclohexyl, and pyridyl groups, allowing for diverse functionalization of the final pyridine scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acyl Pyridine Supplier
The technological potential of this copper-catalyzed synthesis positions 3-acyl pyridine compounds as a cornerstone for next-generation therapeutic agents. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-acyl pyridine intermediate adheres to the highest industry standards. We understand that consistency is key in pharmaceutical supply, and our advanced process control systems allow us to maintain tight tolerances on critical quality attributes throughout the production lifecycle.
We invite you to collaborate with our technical team to explore how this innovative route can optimize your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term supply goals. Let us partner with you to bring your vital medicines to market faster and more efficiently.
