Advanced Copper-Catalyzed Synthesis of 3-Acyl Pyridines for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery and material science. Patent CN107602452B discloses a groundbreaking synthetic method for 3-acyl pyridine compounds, addressing the long-standing challenges of complexity and cost in heterocyclic chemistry. This innovation utilizes a one-pot multi-step series reaction involving enaminone compounds and α,β-saturated ketones or aldehydes, driven by a robust copper catalytic system. By leveraging earth-abundant copper catalysts alongside TEMPO oxidants under aerobic conditions, this technology offers a sustainable alternative to traditional methods that often rely on expensive precious metals or harsh stoichiometric oxidants. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while adhering to green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acyl pyridine derivatives has been plagued by significant operational inefficiencies and environmental burdens. Conventional routes frequently involve multi-step sequences requiring the isolation and purification of unstable intermediates, which drastically reduces overall yield and increases solvent consumption. Many traditional protocols depend on stoichiometric amounts of toxic heavy metal oxidants or require extremely harsh reaction conditions, such as high temperatures and strong acids, which limit functional group tolerance. Furthermore, the atom economy of these older methods is often poor, generating substantial quantities of chemical waste that complicate disposal and increase the environmental factor (E-factor) of the manufacturing process. These limitations make conventional synthesis economically unviable for large-scale production of complex API intermediates, creating bottlenecks in the supply chain for downstream drug manufacturers.
The Novel Approach
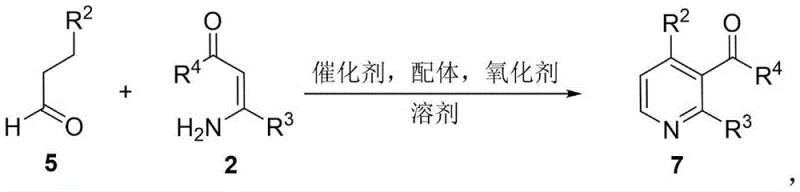
In stark contrast, the methodology described in CN107602452B introduces a streamlined, one-pot strategy that merges multiple transformation steps into a single operational unit. This novel approach employs readily available enaminones and saturated ketones or aldehydes as starting materials, which are converted directly into the target 3-acyl pyridines through an oxidative cyclization cascade. The reaction proceeds under relatively mild thermal conditions, typically between 110°C and 130°C, utilizing molecular oxygen from air as the terminal oxidant. This eliminates the need for hazardous chemical oxidants and simplifies the reaction setup. The use of a copper catalyst system, specifically copper acetate paired with bipyridine ligands and TEMPO, ensures high catalytic efficiency and broad substrate compatibility. This shift from multi-step batch processing to a telescoped one-pot reaction significantly enhances process mass intensity and reduces the carbon footprint of the synthesis.

The versatility of this new synthetic route is further demonstrated by its ability to accommodate a wide array of substituents on both the ketone and enaminone components. As illustrated in the reaction scope, the method tolerates electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens and trifluoromethyl groups on the aromatic rings. Moreover, the protocol is not limited to benzene derivatives; it successfully incorporates heterocycles such as thiophene and pyridine, as well as aliphatic structures like cyclohexyl and isopropyl groups. This broad scope is critical for medicinal chemists who require diverse libraries of analogues for structure-activity relationship (SAR) studies. The ability to access such structural diversity from cheap and easy-to-obtain raw materials underscores the commercial viability of this technology for producing high-purity OLED materials and pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the synergistic interaction between the copper catalyst and the nitroxyl radical oxidant. The mechanism likely initiates with the coordination of the copper species to the enaminone substrate, facilitating an initial condensation or addition step with the saturated ketone or aldehyde. The TEMPO radical plays a pivotal role in the subsequent oxidative dehydrogenation steps, effectively abstracting hydrogen atoms to drive the aromatization of the pyridine ring. This radical-mediated pathway allows the reaction to proceed under aerobic conditions, where oxygen serves to regenerate the active oxidant species, creating a catalytic cycle that minimizes reagent consumption. Understanding this mechanistic nuance is vital for process chemists aiming to optimize reaction parameters such as catalyst loading and oxygen flow rates for maximum efficiency.
From an impurity control perspective, the mildness of the copper/TEMPO system offers distinct advantages over aggressive acidic or basic cyclization methods. Harsh conditions often lead to polymerization of the enaminone starting material or over-oxidation of the sensitive acyl side chain, resulting in complex impurity profiles that are difficult to separate. By maintaining the reaction temperature within the 110-130°C range and using a selective radical oxidant, the formation of these by-products is significantly suppressed. This results in a cleaner crude reaction mixture, which simplifies downstream purification processes like crystallization or chromatography. For quality assurance teams, this translates to more consistent batch-to-batch purity and reduced risk of genotoxic impurities carrying over into the final active pharmaceutical ingredient.
How to Synthesize 3-Acyl Pyridine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the sequential addition of reagents and the maintenance of an aerobic environment. The standard protocol involves dissolving the enaminone in a polar aprotic solvent such as N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), followed by the addition of the ketone substrate. The catalyst system, comprising copper acetate, a bipyridine ligand, and TEMPO, is then introduced before heating the mixture. Detailed standardized synthesis steps see the guide below.
- Dissolve the enaminone compound in a suitable solvent such as DMF or toluene within a reaction vessel.
- Sequentially add the alpha,beta-saturated ketone or aldehyde, copper catalyst (e.g., Cu(OAc)2), ligand (e.g., bipyridine), and oxidant (e.g., TEMPO).
- Heat the reaction mixture to 110-130°C under air atmosphere for approximately 20 hours, followed by aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost reduction and supply reliability. The primary economic driver is the substitution of expensive noble metal catalysts, such as palladium or rhodium, with inexpensive copper salts. Copper is abundant, widely available, and significantly cheaper, which directly lowers the raw material cost per kilogram of the final product. Additionally, the use of air as the oxidant removes the need to purchase and store hazardous chemical oxidants, further reducing material costs and safety compliance expenses associated with handling dangerous goods.
- Cost Reduction in Manufacturing: The one-pot nature of this reaction eliminates the need for intermediate isolation and purification steps, which are traditionally labor-intensive and solvent-heavy. By telescoping the synthesis into a single vessel, manufacturers can drastically reduce solvent consumption, energy usage for heating and cooling cycles, and labor hours. This process intensification leads to substantial cost savings in utility and waste disposal, making the production of complex pharmaceutical intermediates much more economically attractive without compromising on yield or quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically enaminones and simple aromatic ketones, are commodity chemicals that are readily available from multiple global suppliers. This diversifies the supply base and mitigates the risk of shortages that often plague specialized reagents. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent production schedules and reliable delivery timelines for downstream clients.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the absence of sensitive reagents and the use of standard heating equipment. The high atom economy and the generation of water as the primary by-product align perfectly with modern environmental regulations and corporate sustainability goals. Reduced chemical waste simplifies effluent treatment and lowers the environmental compliance burden, allowing manufacturers to operate more sustainably while avoiding potential regulatory fines or shutdowns associated with high-waste processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on operational feasibility and performance metrics for potential adopters of this technology.
Q: What are the key advantages of this copper-catalyzed method over traditional pyridine synthesis?
A: This method utilizes a one-pot multi-step series reaction that significantly simplifies the operational procedure compared to traditional multi-step syntheses. It employs inexpensive and earth-abundant copper catalysts instead of precious metals, uses air as a green oxidant, and demonstrates excellent atom economy, reducing waste generation and overall production costs.
Q: What is the substrate scope for this 3-acyl pyridine synthesis?
A: The protocol exhibits broad substrate tolerance, accommodating various alpha,beta-saturated ketones and aldehydes with substituents such as methyl, methoxy, halogens (fluorine, chlorine, bromine), and trifluoromethyl groups on the phenyl ring. It also supports heterocyclic substrates like thiophene and pyridine, as well as aliphatic chains like cyclohexyl and isopropyl groups.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly suitable for scale-up due to its use of commercially available raw materials, mild reaction conditions (110-130°C), and simple workup procedures involving standard extraction and chromatography. The avoidance of sensitive reagents and the robustness of the copper/TEMPO catalytic system facilitate reliable commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acyl Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis for the global pharmaceutical market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-acyl pyridine intermediate delivered meets the highest industry standards for safety and efficacy.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this advanced synthetic route into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how this method can optimize your specific production costs. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the manufacturing of high-value nitrogen heterocycles.
