Advanced Molybdenum-Copper Co-Catalysis for Scalable 3-Trifluoromethyl-1,2,4-Triazole Manufacturing
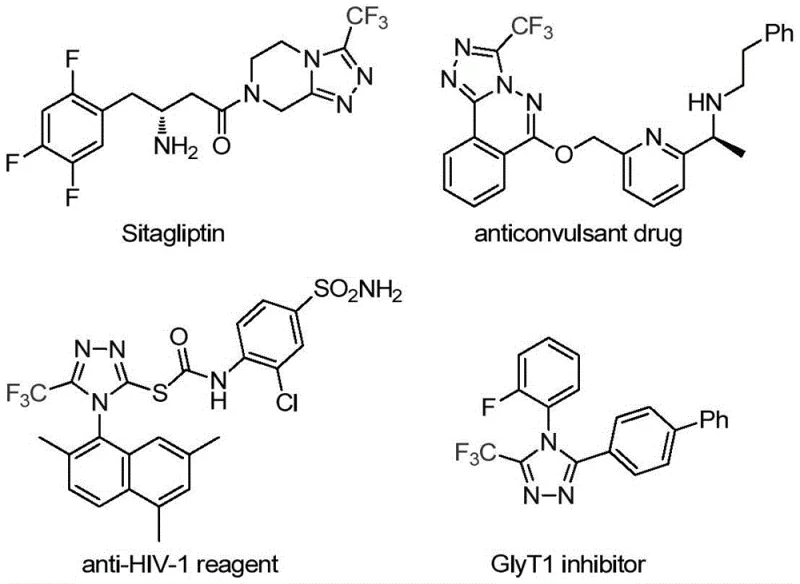
The pharmaceutical and agrochemical industries continuously demand efficient access to fluorinated heterocycles, particularly 1,2,4-triazoles, due to their profound impact on metabolic stability and bioavailability. A pivotal advancement in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology addresses the critical need for robust synthetic routes that can deliver high-purity intermediates essential for modern drug discovery. The significance of this structural motif is underscored by its presence in blockbuster medications such as Sitagliptin, where the triazole core plays a vital role in biological activity. By leveraging a unique molybdenum and copper co-catalytic system, this invention offers a streamlined pathway that bypasses the limitations of classical cyclization methods, positioning it as a valuable asset for any organization seeking a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has relied on methodologies that are often fraught with operational challenges and safety concerns. Traditional literature reports predominantly describe the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These legacy routes frequently necessitate harsh reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate waste management and increase overall production costs. Furthermore, alternative multi-component reactions involving diazonium salts or trifluorodiazoethane introduce significant safety risks due to the explosive nature of diazo compounds, making them less desirable for commercial scale-up. The reliance on such unstable precursors not only elevates the barrier to entry for manufacturing but also introduces variability in yield and purity that is unacceptable for GMP-grade production environments.
The Novel Approach
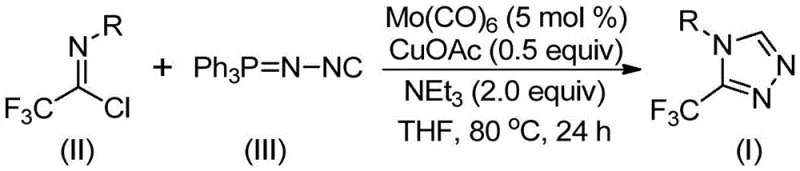
In stark contrast, the methodology presented in the patent utilizes a transition metal co-catalyzed cycloaddition strategy that fundamentally reshapes the synthetic landscape for these heterocycles. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as stable, commercially available starting materials, the process eliminates the need for dangerous diazo intermediates. The reaction proceeds under remarkably mild conditions, typically between 70°C and 90°C, which significantly reduces energy consumption and thermal stress on sensitive functional groups. This approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing process, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing. The ability to access diverse substitution patterns through simple substrate design further amplifies the utility of this method for generating focused libraries of drug candidates.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this transformation hinges on the synergistic interaction between molybdenum hexacarbonyl and cuprous acetate, which facilitates a complex cascade of bond-forming events. The proposed mechanism suggests that molybdenum hexacarbonyl initially activates the functionalized isonitrile to form a reactive metal complex, thereby lowering the energy barrier for subsequent nucleophilic attacks. Concurrently, the copper species promotes a [3+2] cycloaddition between the activated isonitrile and the trifluoroethylimidoyl chloride, leading to the formation of a five-membered ring intermediate. This intricate dance of coordination chemistry ensures high regioselectivity, favoring the formation of the desired 3-trifluoromethyl-1,2,4-triazole scaffold over potential isomeric byproducts. The final step involves the elimination of triphenylphosphine oxide, driven by the presence of water in the system, to yield the stable aromatic triazole product.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining a clean product profile. Unlike high-temperature thermal cyclizations that often promote decomposition or polymerization side reactions, this catalytic system operates within a narrow thermal window that preserves the integrity of sensitive substituents such as halogens and nitro groups. The use of molecular sieves in the reaction mixture further aids in controlling moisture levels, preventing hydrolysis of the imidoyl chloride precursor before it can participate in the cycle. This precise control over the reaction environment results in a simplified downstream purification process, where standard column chromatography is sufficient to achieve high-purity specifications required for clinical applications. Such robustness in impurity management is a key consideration for R&D directors evaluating the feasibility of scaling this chemistry.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and minimize waste. The patent outlines a generalized procedure where the molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile is optimized, typically favoring a slight excess of the isonitrile to drive the equilibrium forward. The choice of solvent is also critical, with tetrahydrofuran (THF) identified as the superior medium for dissolving both organic substrates and inorganic catalysts effectively. Operators should note that while the reaction time can extend up to 30 hours, monitoring conversion via TLC or HPLC is recommended to determine the optimal quench point for specific substrates. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel catalytic method offers tangible benefits that extend beyond mere chemical elegance. The reliance on earth-abundant metals like copper and molybdenum, rather than expensive precious metals like palladium or rhodium, inherently lowers the raw material cost baseline. Additionally, the starting materials, specifically the functionalized isonitriles and imidoyl chlorides, are either commercially off-the-shelf or easily synthesized from bulk commodities, ensuring a stable and resilient supply chain. This accessibility mitigates the risk of production delays caused by sourcing bottlenecks, a common pain point in the global chemical market. By adopting this route, organizations can secure a more predictable supply of critical intermediates while simultaneously reducing their exposure to volatile pricing associated with rare metal catalysts.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the elimination of costly purification steps and the use of inexpensive catalytic systems. Since the reaction does not require cryogenic conditions or high-pressure equipment, the capital expenditure for reactor infrastructure is significantly minimized. Furthermore, the high atom economy of the cycloaddition reduces the volume of waste generated per kilogram of product, leading to substantial savings in waste disposal fees. The simplicity of the workup, which avoids complex aqueous extractions, further streamlines the manufacturing timeline, allowing for faster batch turnover and improved facility utilization rates without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly into improved supply chain continuity. Because the process tolerates a wide range of functional groups, manufacturers can source diverse substrates from multiple vendors without needing to revalidate the entire process for each new batch. This flexibility allows procurement teams to negotiate better terms with suppliers and switch sources rapidly if disruptions occur. The scalability of the method, demonstrated from milligram to gram scales in the patent data, suggests a smooth path to multi-kilogram production, ensuring that commercial demands can be met consistently without the teething problems often associated with scaling novel chemistries.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The use of THF, a recyclable solvent, and the absence of toxic heavy metal residues simplify the environmental compliance burden. The mild reaction temperatures reduce the carbon footprint associated with heating and cooling large-scale reactors. Moreover, the high selectivity of the catalyst minimizes the formation of hazardous byproducts, making the effluent easier to treat. These factors collectively enhance the sustainability profile of the manufacturing process, which is increasingly becoming a decisive factor for partners seeking long-term collaborations in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this triazole synthesis platform. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing a clear framework for technical evaluation. Understanding these nuances is essential for teams considering the integration of this technology into their existing pipelines. The answers reflect the practical realities of running this chemistry in a laboratory or pilot plant setting.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies using THF as the preferred solvent at temperatures between 70°C and 90°C for 18 to 30 hours, utilizing a catalytic system of Mo(CO)6 and CuOAc with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent substrate tolerance, successfully synthesizing derivatives with substituents such as methyl, methoxy, fluoro, chloro, nitro, and phenethyl groups with high efficiency.
Q: Is the purification process complex for large-scale production?
A: The post-treatment is straightforward, involving simple filtration followed by standard column chromatography, which avoids complex extraction or distillation steps often required in traditional heterocyclic synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced heterocyclic intermediates in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis for a specific analog or bulk supply for clinical trials, our infrastructure is designed to support your most demanding projects with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this innovative Mo/Cu catalyzed route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this methodology for your specific application. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us collaborate to build a more efficient and sustainable future for pharmaceutical manufacturing together.
