Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Manufacturing
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in Chinese Patent CN113307778A, which discloses a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are ubiquitous in medicinal chemistry, forming the core structure of numerous blockbuster drugs such as Sitagliptin, Deferasirox, and Maraviroc, as illustrated in the structural diversity of bioactive molecules below. The introduction of a trifluoromethyl group into these frameworks is particularly strategic, as it profoundly enhances physicochemical properties including metabolic stability, lipophilicity, and bioavailability, thereby addressing key challenges in modern drug design and development.
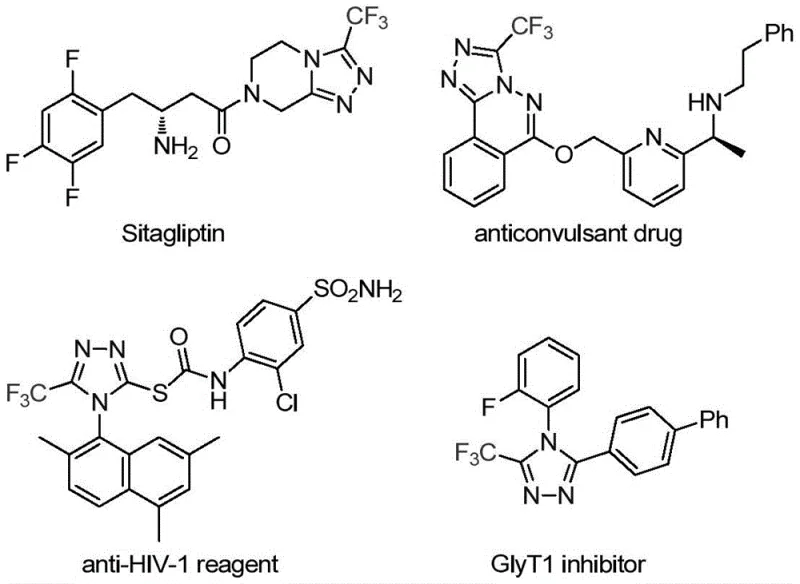
The significance of this patent extends beyond mere academic interest; it represents a tangible solution for reliable pharmaceutical intermediate suppliers aiming to streamline their manufacturing pipelines. By leveraging a molybdenum and copper co-catalytic system, the disclosed method overcomes many of the thermodynamic and kinetic barriers associated with traditional triazole synthesis. For R&D directors and process chemists, this translates to a versatile platform capable of generating diverse analogues with high precision. The ability to access these valuable scaffolds through a simplified, one-pot procedure marks a substantial advancement in the field of fine chemical intermediates, offering a pathway to reduce lead times for high-purity pharmaceutical intermediates while maintaining rigorous quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant synthetic challenges that hinder efficient commercial scale-up of complex heterocycles. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical routes often suffer from harsh reaction conditions, requiring extreme temperatures or pressures that pose safety risks in a plant environment. Furthermore, alternative strategies involving copper-catalyzed multi-component reactions frequently utilize diazonium salts and trifluorodiazoethane, reagents that are notoriously unstable, potentially explosive, and difficult to handle on a large scale. Such inherent hazards not only escalate operational costs due to stringent safety protocols but also limit the scope of compatible functional groups, thereby restricting the chemical space available for drug discovery teams.
The Novel Approach
In stark contrast to these legacy methods, the technology described in Patent CN113307778A introduces a paradigm shift by employing a mild, co-catalytic cycloaddition strategy. As depicted in the general reaction scheme below, the process utilizes trifluoroethylimidoyl chloride and a functionalized isonitrile (NIITP) as the primary building blocks. This approach eliminates the need for hazardous diazonium species and operates under remarkably gentle conditions, typically between 70°C and 90°C. The reaction is driven by a synergistic catalytic system comprising molybdenum hexacarbonyl and cuprous acetate, which activates the substrates efficiently without degrading sensitive functional groups. This innovation allows for cost reduction in pharmaceutical intermediate manufacturing by simplifying the operational workflow, reducing energy consumption, and utilizing starting materials that are cheap and easily obtainable from standard chemical suppliers.
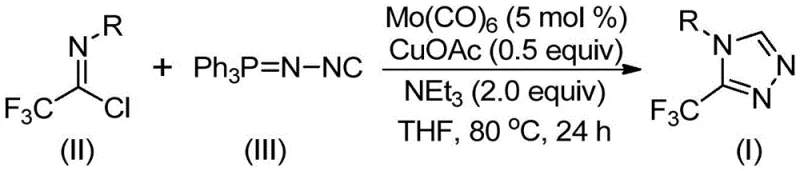
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The efficacy of this synthetic route lies in the intricate interplay between the molybdenum and copper catalysts, which orchestrate the formation of the five-membered triazole ring with high regioselectivity. Mechanistically, the molybdenum hexacarbonyl serves as a crucial metal activator for the functionalized isonitrile, forming a transient metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Concurrently, the cuprous acetate promotes the [3+2] cycloaddition of this activated species onto the trifluoroethylimidoyl chloride. This dual-catalyst synergy lowers the activation energy barrier for the ring-closing step, facilitating the rapid assembly of the heterocyclic core. Following the cyclization, the intermediate undergoes a spontaneous elimination of triphenylphosphine oxide, driven by the presence of water in the system or during workup, to yield the final 3-trifluoromethyl-substituted 1,2,4-triazole product. This elegant mechanism ensures high atom economy and minimizes the formation of difficult-to-remove byproducts.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring the production of high-purity OLED material or pharmaceutical grades. The mild nature of the reaction prevents the decomposition of the trifluoromethyl group, a common issue in harsher acidic or basic conditions. Moreover, the specific stoichiometry—typically employing a slight excess of the functionalized isonitrile relative to the imidoyl chloride—drives the reaction to completion, minimizing the presence of unreacted starting materials in the crude mixture. The tolerance for various substituents on the aromatic ring, including electron-withdrawing groups like nitro and halogens as well as electron-donating groups like methyl and methoxy, demonstrates the robustness of the catalytic cycle. This broad substrate scope, evidenced by the successful synthesis of derivatives I-1 through I-5 shown below, confirms that the method is not limited to simple substrates but is applicable to complex, functionalized architectures required for advanced drug candidates.
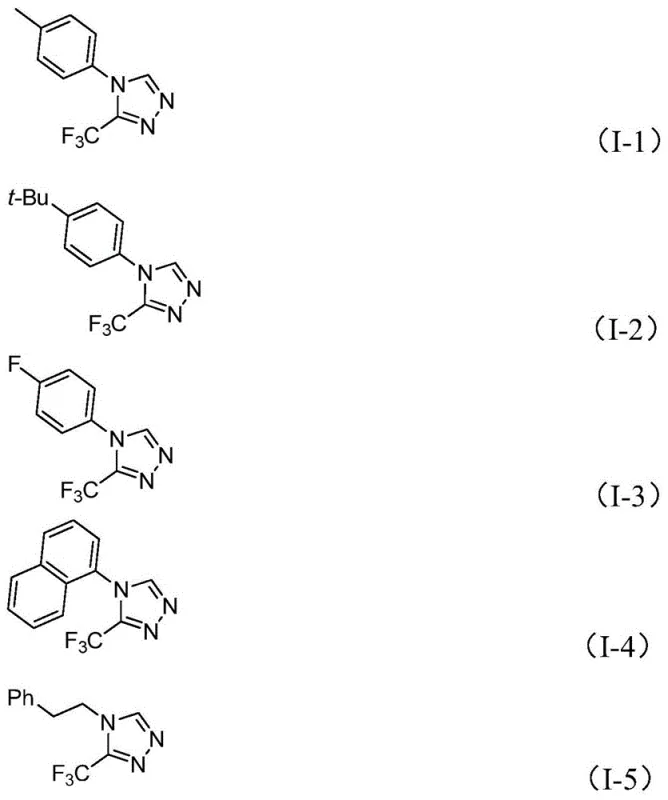
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is straightforward, making it highly accessible for process development teams aiming to replicate these results. The protocol involves charging a reaction vessel with the catalysts, base, and substrates in a suitable organic solvent such as THF, followed by heating and stirring. The simplicity of the workup procedure, which typically involves filtration and standard column chromatography, further enhances its appeal for rapid library synthesis. For those interested in the precise operational parameters, stoichiometry, and purification techniques validated by the patent data, the detailed standardized synthesis steps are provided in the guide below.
- Charge a reaction vessel with molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile (NIITP) in an organic solvent such as THF.
- Heat the reaction mixture to a temperature range of 70-90°C (preferably 80°C) and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling strategic benefits that extend far beyond the laboratory bench. The shift towards this Mo/Cu co-catalyzed method addresses critical pain points in the sourcing and production of heterocyclic intermediates. By replacing hazardous and expensive reagents with stable, commodity-grade chemicals, the process inherently de-risks the supply chain. The reliance on trifluoroethylimidoyl chloride and functionalized isonitrile, both of which are commercially available or easily synthesized from bulk amines, ensures a steady flow of raw materials. This stability is crucial for maintaining continuous production schedules and avoiding the delays often associated with the procurement of specialized, high-risk reagents used in conventional triazole synthesis.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the elimination of costly and dangerous reagents. Traditional methods often require bespoke synthesis of diazonium salts or oxazolinones, which adds multiple steps and significant expense to the bill of materials. In contrast, this novel route utilizes a direct cycloaddition that converges two readily available precursors in a single pot. The use of relatively inexpensive catalysts like cuprous acetate and molybdenum hexacarbonyl, coupled with the ability to run the reaction at moderate temperatures, drastically reduces energy consumption and catalyst loading costs. Furthermore, the high reaction efficiency and clean profile minimize the burden on downstream purification, leading to substantial cost savings in solvent usage and waste disposal.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction conditions. The tolerance for a wide range of functional groups means that a single manufacturing platform can produce a diverse array of intermediates without requiring extensive retooling or process re-optimization. This flexibility allows manufacturers to respond swiftly to changing market demands for different API precursors. Additionally, the avoidance of explosive intermediates simplifies logistics and storage requirements, reducing the regulatory hurdles and insurance costs associated with transporting hazardous materials. This results in a more agile and reliable supply network capable of meeting tight delivery windows for global pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is significantly lower than that of legacy methods, aligning with modern green chemistry principles and increasingly stringent environmental regulations. The reaction generates minimal hazardous waste, and the primary byproduct, triphenylphosphine oxide, is easily separated and managed. The scalability of the process has been demonstrated at the gram level with high yields, providing a clear path for expansion to kilogram and metric ton scales. The mild conditions reduce the risk of thermal runaways, enhancing plant safety and ensuring compliance with occupational health and safety standards. This combination of safety, efficiency, and environmental stewardship makes the process ideal for long-term commercial partnerships focused on sustainable manufacturing.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial potential of this synthesis method, we have compiled answers to common questions based on the patent specifications and experimental data. These insights address concerns regarding substrate compatibility, reaction safety, and the practical aspects of implementing this technology in a GMP environment. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the key advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often require harsh conditions, explosive diazonium salts, or complex multi-step sequences, this novel approach utilizes mild temperatures (70-90°C) and readily available starting materials. The co-catalytic system ensures high reaction efficiency and broad substrate tolerance, significantly simplifying the purification process and improving overall safety profiles for industrial scale-up.
Q: Can this synthesis method accommodate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates exceptional functional group tolerance. The patent data confirms successful synthesis with various substituents including methyl, tert-butyl, fluoro, chloro, methoxy, and nitro groups at ortho, meta, and para positions. This versatility allows for the rapid generation of diverse libraries of 3-trifluoromethyl-1,2,4-triazole derivatives for SAR studies.
Q: Is this process suitable for large-scale commercial production?
A: The process is explicitly designed for scalability. The use of stable, non-hazardous reagents like trifluoroethylimidoyl chloride and functionalized isonitrile, combined with a robust catalytic system in common solvents like THF, facilitates easy transition from gram-level laboratory synthesis to kilogram or ton-scale manufacturing without compromising yield or purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
The technological advancements presented in Patent CN113307778A underscore the immense potential of 3-trifluoromethyl-1,2,4-triazoles as pivotal intermediates in next-generation therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methodologies into robust, industrial-scale processes. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from pilot plant to full-scale manufacturing. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to leverage our expertise to optimize your supply chain and accelerate your drug development timelines. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this efficient synthesis route can enhance your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us collaborate to bring your high-purity triazole projects to market faster, safer, and more economically.
