Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Drug Manufacturing
The pharmaceutical industry continuously seeks robust synthetic methodologies to access heterocyclic scaffolds that serve as the backbone for modern therapeutics. Patent CN113045503A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in the synthesis of these vital pharmacophores. Quinazolinones are ubiquitous in medicinal chemistry, exhibiting a diverse range of biological activities including antifungal, antiviral, and anticancer properties, yet their functionalization with trifluoromethyl groups has historically been challenging. The introduction of the trifluoromethyl moiety is strategically significant as it enhances metabolic stability, lipophilicity, and bioavailability of the parent drug molecule. This new protocol leverages a transition metal palladium-catalyzed carbonylation tandem reaction, offering a streamlined pathway that transforms cheap and easily obtainable trifluoroethylimidoyl chloride and various amines into high-value intermediates. For R&D directors and procurement specialists, this represents a pivotal shift towards more efficient, cost-effective, and scalable manufacturing processes for complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinone compounds has been plagued by significant operational and economic hurdles that hinder large-scale adoption. Conventional literature reports typically rely on cyclization reactions involving anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, which often necessitate harsh reaction conditions and expensive, moisture-sensitive reagents. Alternative routes involving the cyclization of anthranilic acid esters with unstable trifluoroacetamides or the use of isatoic anhydride frequently suffer from low yields and narrow substrate scopes, limiting the structural diversity accessible to medicinal chemists. Furthermore, methods promoted by coupling agents like T3P, while effective in small batches, introduce substantial cost burdens and complicate downstream purification due to the generation of phosphorus-containing byproducts. These legacy methods often require pre-activation steps that increase process time and waste generation, making them less attractive for the cost-conscious manufacturing of active pharmaceutical ingredients (APIs) where margin compression is a constant pressure.
The Novel Approach
In stark contrast to these inefficient legacy protocols, the novel approach detailed in the patent utilizes a sophisticated palladium-catalyzed carbonylation strategy that dramatically simplifies the synthetic landscape. By employing trifluoroethylimidoyl chloride and amines as starting materials, the method bypasses the need for expensive pre-activated substrates, relying instead on a catalytic cycle that efficiently constructs the quinazolinone core. The reaction operates under relatively mild thermal conditions at 110 °C in common organic solvents like 1,4-dioxane, utilizing TFBen as a safe and effective carbon monoxide surrogate. This eliminates the safety hazards associated with handling high-pressure carbon monoxide gas directly. The process demonstrates exceptional substrate compatibility, tolerating a wide array of functional groups including halogens, alkyls, and trifluoromethyl groups on both the aryl and amine components. This versatility allows for the rapid generation of diverse chemical libraries, accelerating the drug discovery timeline while simultaneously reducing the cost of goods sold (COGS) through the use of commodity chemicals.
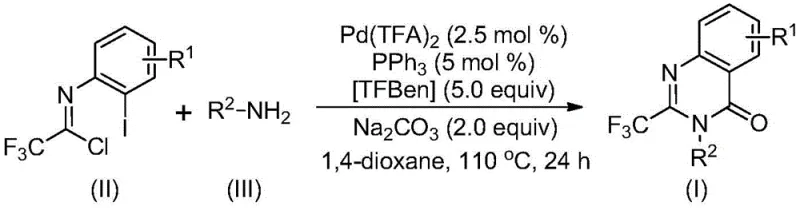
The mechanistic elegance of this transformation lies in its multi-step tandem nature, which seamlessly integrates carbon-nitrogen bond formation with carbonylation. The reaction likely initiates with an alkali-promoted intermolecular carbon-nitrogen bond coupling to generate a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst undergoes oxidative addition into the carbon-iodine bond of the substrate, forming a key divalent palladium intermediate. The TFBen additive then releases carbon monoxide under heating, which inserts into the carbon-palladium bond to create an acyl palladium species. This is followed by base-promoted palladium-nitrogen coordination to form a seven-membered ring palladium intermediate, finally concluding with reductive elimination to release the desired 2-trifluoromethyl-substituted quinazolinone product. This intricate catalytic cycle ensures high atom economy and minimizes side reactions, resulting in the observed high yields ranging from 74% to 98% across various substrates. Such mechanistic control is crucial for maintaining a clean impurity profile, a key requirement for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
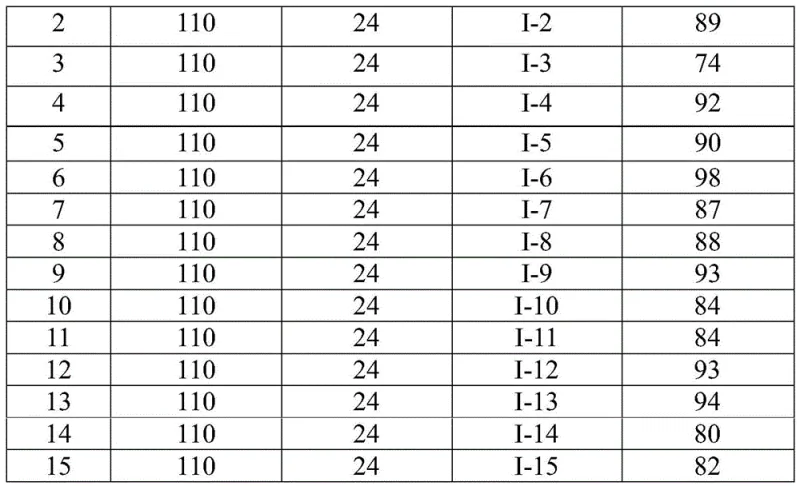
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized reaction parameters to maximize yield and purity. The protocol involves charging a reactor with specific molar ratios of palladium trifluoroacetate, triphenylphosphine, and sodium carbonate alongside the substrate and CO source. The detailed standardized synthesis steps below outline the exact procedure for achieving consistent results, ensuring that the transition from bench-scale discovery to process development is seamless. Operators should note that the choice of solvent, specifically 1,4-dioxane, plays a critical role in solubilizing the reactants and facilitating the catalytic turnover, while the reaction time of 16 to 30 hours allows for complete conversion without excessive thermal degradation.
- Combine palladium trifluoroacetate, triphenylphosphine, sodium carbonate, TFBen, trifluoroethylimidoyl chloride, and amine in an organic solvent like dioxane.
- Heat the reaction mixture to 110 °C and stir for 16 to 30 hours to allow the carbonylation tandem reaction to proceed.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl-substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical efficiency. The reliance on commercially available and inexpensive starting materials such as trifluoroethylimidoyl chloride and various amines drastically reduces the raw material cost burden compared to specialized precursors required by older methods. Furthermore, the elimination of hazardous gases and the use of standard heterogeneous catalysts simplify the engineering controls required for the reactor setup, thereby lowering capital expenditure (CAPEX) for new production lines. The robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality intermediates that can reliably feed downstream API synthesis campaigns without unexpected delays or quality failures.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive activating agents and complex precursors with commodity chemicals that are readily sourced from the global market. By removing the need for pre-activation steps and utilizing a catalytic amount of palladium rather than stoichiometric reagents, the overall material costs are significantly lowered. Additionally, the simplified workup procedure, which involves basic filtration and standard chromatography, reduces the consumption of solvents and silica gel, leading to substantial savings in waste disposal and operational expenditures. This lean manufacturing approach directly contributes to a more competitive pricing structure for the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of raw materials that are not subject to the same geopolitical or logistical constraints as exotic reagents. Since the amines and imidoyl chlorides used are widely produced for various industrial applications, the risk of supply disruption is markedly reduced. The method's tolerance for diverse functional groups also means that alternative suppliers for specific substituted amines can be qualified more easily, providing procurement teams with greater flexibility in vendor management. This diversification of the supply base ensures continuity of supply even in volatile market conditions, safeguarding production schedules for critical drug candidates.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns well with green chemistry principles by avoiding the use of toxic carbon monoxide gas and minimizing heavy metal waste through efficient catalysis. The reaction can be safely scaled from gram levels in the lab to multi-kilogram or tonne scales in production facilities without requiring specialized high-pressure equipment. The straightforward post-treatment process facilitates easier purification and isolation of the product, reducing the environmental footprint associated with solvent-intensive purification techniques. This scalability ensures that the technology can meet the growing demand for quinazolinone-based therapeutics while adhering to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this technology can be integrated into existing workflows. Understanding these nuances helps stakeholders make informed decisions about adopting this route for their specific project needs, ensuring alignment between technical feasibility and business objectives.
Q: What are the primary advantages of this Pd-catalyzed method over traditional cyclization routes?
A: Unlike traditional methods requiring harsh conditions or expensive activating agents like T3P, this novel approach utilizes cheap, readily available starting materials and achieves significantly higher yields with broader substrate tolerance.
Q: Can this synthesis method be scaled for industrial production of drug intermediates?
A: Yes, the process is designed for scalability, utilizing simple post-treatment procedures like filtration and column chromatography, making it suitable for expanding from gram-level research to commercial manufacturing.
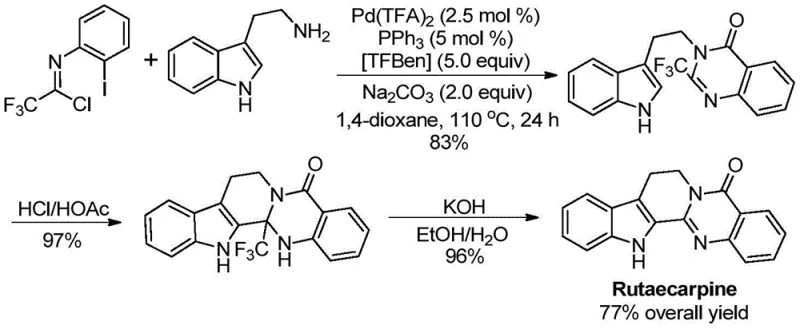
Q: What specific drug molecules can be synthesized using this quinazolinone platform?
A: This methodology has been successfully applied to the high-yield total synthesis of complex natural products and drug candidates, most notably demonstrating efficiency in the production of the alkaloid Rutaecarpine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is supported by robust manufacturing capabilities. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 2-trifluoromethyl quinazolinone derivatives we produce. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our processes are designed to deliver exactly that.
We invite you to collaborate with us to leverage this efficient synthetic route for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and accelerate your time to market.
