Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those bearing fluorine motifs which are critical for enhancing metabolic stability and bioavailability. Patent CN112480015B introduces a groundbreaking multi-component one-pot strategy for synthesizing 2-trifluoromethyl substituted quinazolinones, a scaffold prevalent in numerous bioactive molecules ranging from antifungals to anticancer agents. This technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to diversify their portfolio with high-value scaffolds. The quinazolinone core is found in established drugs such as Methaqualone and Afloqualone, as well as emerging candidates like Albaconazole, highlighting the immense commercial potential of mastering this synthetic route.
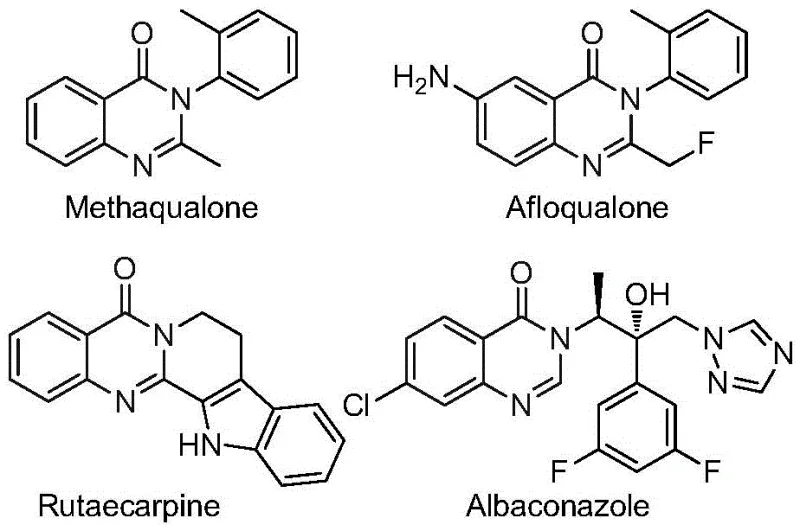
The introduction of the trifluoromethyl group into the quinazolinone framework is not merely a structural modification but a strategic enhancement of physicochemical properties. Fluorine atoms impart unique electronegativity and lipophilicity, often leading to improved membrane permeability and resistance to metabolic degradation. However, traditional methods to install this group often suffer from harsh conditions or limited substrate scope. The methodology disclosed in CN112480015B addresses these pain points by utilizing a palladium-catalyzed carbonylation cascade that operates under relatively mild thermal conditions compared to high-pressure alternatives. For R&D directors focused on purity and process robustness, this patent offers a pathway to access complex heterocycles with high efficiency and minimal impurity profiles, ensuring that the resulting high-purity quinazolinone derivatives meet stringent regulatory standards for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazolinone rings has relied on methodologies that pose significant challenges for large-scale manufacturing and cost-effective production. Conventional routes often involve the use of high-pressure carbon monoxide gas, which necessitates specialized autoclaves and rigorous safety protocols, thereby inflating capital expenditure and operational complexity. Furthermore, many existing protocols require pre-activated substrates such as 2-bromoformylaniline or acid anhydrides, which are not only more expensive than simple nitro compounds but also generate stoichiometric amounts of waste during their preparation. Iron-catalyzed condensations or ruthenium-catalyzed reductions often struggle with narrow substrate tolerance, failing to accommodate sensitive functional groups or sterically hindered positions. These limitations result in lower overall yields and a restricted ability to generate diverse libraries of analogs, which is a critical bottleneck for medicinal chemists exploring structure-activity relationships. Consequently, the cost reduction in API manufacturing is severely hampered by the inefficiency and safety risks associated with these legacy processes.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN112480015B leverages a sophisticated yet operationally simple palladium-catalyzed system that transforms cheap and readily available nitro compounds directly into the target heterocycles. By employing molybdenum hexacarbonyl [Mo(CO)6] as a solid carbon monoxide surrogate, the process completely eliminates the need for handling hazardous CO gas cylinders, drastically improving workplace safety and simplifying reactor design. The reaction utilizes trifluoroethylimidoyl chloride and nitro compounds as starting materials, both of which are commercially accessible and cost-effective. The one-pot nature of the synthesis means that multiple bond-forming events—reduction, coupling, and cyclization—occur in a single vessel without the need for intermediate isolation. This telescoping of steps not only saves time but also minimizes solvent usage and waste generation, aligning perfectly with green chemistry principles. For supply chain managers, this translates to a more streamlined production workflow that enhances throughput and reliability.
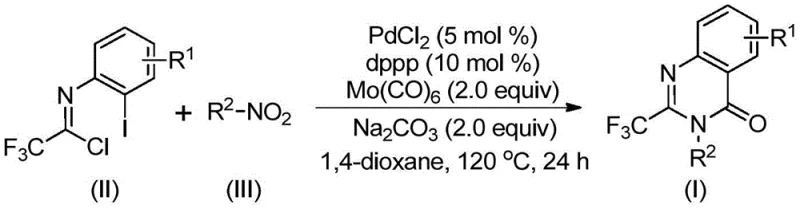
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction is believed to initiate with the reduction of the nitro group to an amine by Mo(CO)6, which serves a dual role as both a CO source and a reducing agent. Once the amine is generated in situ, it undergoes a base-promoted nucleophilic attack on the trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst, coordinated with the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride moiety (or a related intermediate), forming a reactive organopalladium species. The thermal decomposition of Mo(CO)6 releases carbon monoxide, which then inserts into the carbon-palladium bond to generate an acyl-palladium intermediate. This acyl species then undergoes intramolecular cyclization via nucleophilic attack by the nitrogen atom, facilitated by the base, to form a seven-membered palladacycle. Finally, reductive elimination releases the desired 2-trifluoromethyl quinazolinone product and regenerates the active palladium catalyst. This intricate cascade ensures high atom economy and precise control over the regioselectivity of the cyclization.
From an impurity control perspective, the choice of ligand and base plays a pivotal role in suppressing side reactions. The use of 1,3-bis(diphenylphosphino)propane (dppp) stabilizes the palladium center, preventing premature aggregation or decomposition that could lead to metallic impurities. Furthermore, the use of sodium carbonate as a mild base helps to neutralize the HCl byproduct generated during the amidine formation without promoting hydrolysis of the sensitive imidoyl chloride or the final product. The reaction conditions, specifically the temperature of 120°C in 1,4-dioxane, are optimized to balance the rate of CO release from Mo(CO)6 with the kinetics of the cyclization step. This careful tuning minimizes the formation of urea byproducts or homocoupling impurities that are common in less optimized carbonylation reactions. The result is a clean reaction profile that simplifies downstream purification, a key factor in achieving the commercial scale-up of complex heterocycles with consistent quality.
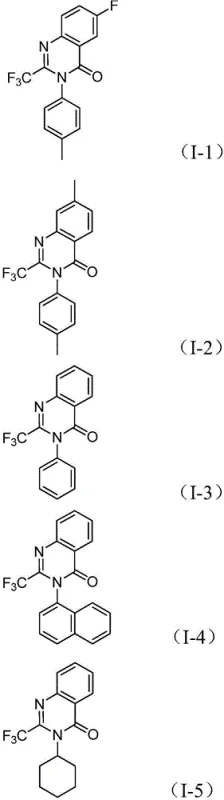
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO source, and substrates in an inert atmosphere to prevent catalyst oxidation. The choice of solvent is critical; while various aprotic solvents can support the reaction, 1,4-dioxane has been identified as the optimal medium for achieving high conversion rates. The reaction mixture is then heated to 120°C and maintained for a period ranging from 16 to 30 hours, depending on the electronic nature of the substituents on the nitro compound. Electron-deficient nitro compounds may react faster, while sterically hindered substrates might require the full duration to reach completion. Detailed standardized synthesis steps for this procedure are provided in the guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2-trifluoromethyl quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits that extend beyond mere chemical novelty. The shift from high-pressure gas systems to solid reagents fundamentally alters the risk profile and cost structure of the manufacturing process. By removing the dependency on specialized high-pressure infrastructure, facilities can utilize standard glass-lined or stainless steel reactors, significantly lowering the barrier to entry for production. Moreover, the reliance on commodity chemicals like nitro compounds and sodium carbonate ensures a stable and resilient supply chain, insulating production schedules from the volatility often associated with specialized reagents. This robustness is essential for maintaining continuous supply lines to downstream pharmaceutical clients who demand reliability above all else.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the operational workflow and the use of inexpensive starting materials. Eliminating the need for high-pressure carbon monoxide equipment removes a major capital expense and reduces maintenance costs associated with safety inspections and leak detection systems. Additionally, the one-pot nature of the reaction reduces labor costs and solvent consumption by avoiding intermediate workups and isolations. The high yields reported across a broad range of substrates mean that less raw material is wasted, directly improving the cost of goods sold (COGS). While specific percentage savings depend on local utility and labor rates, the qualitative reduction in process complexity invariably leads to substantial cost savings in large-scale API manufacturing.
- Enhanced Supply Chain Reliability: The availability of raw materials is a critical factor in supply chain planning. Nitro compounds and trifluoroethylimidoyl chlorides are widely produced industrial chemicals with established global supply networks, reducing the risk of shortages that can plague proprietary or exotic reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain. This reliability allows for more accurate forecasting and inventory management, reducing lead time for high-purity intermediates and ensuring that customer orders are fulfilled consistently without unexpected delays caused by process failures or raw material scarcity.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this methodology is inherently scalable due to its homogeneous nature and moderate temperature requirements. The use of Mo(CO)6 as a solid CO source avoids the mass transfer limitations associated with bubbling gas into liquid, making the reaction easier to control at larger volumes. From an environmental standpoint, the process generates less waste compared to multi-step sequences, and the absence of toxic gas emissions simplifies compliance with environmental regulations. The ability to run the reaction in standard solvents like dioxane, which can be recovered and recycled, further enhances the sustainability profile of the manufacturing process, aligning with modern corporate responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in patent CN112480015B, providing a reliable foundation for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing production capabilities.
Q: What are the key advantages of this one-pot synthesis method over traditional routes?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap nitro compounds and solid Mo(CO)6 as a safe CO source, significantly simplifying operational safety and reducing raw material costs while maintaining high reaction efficiency and broad functional group tolerance.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the method is operable at the gram level and is designed for scalability. The use of standard organic solvents like 1,4-dioxane and commercially available catalysts (PdCl2/dppp) makes the transition from laboratory to pilot plant feasible without requiring specialized high-pressure infrastructure.
Q: What is the substrate scope for the nitro compounds in this reaction?
A: The reaction exhibits excellent substrate compatibility, tolerating various substituents on the aromatic ring including alkyl groups (methyl), halogens (fluorine, chlorine, bromine), and electron-withdrawing groups like trifluoromethyl. Both aryl and alkyl nitro compounds can be effectively utilized to generate diverse quinazolinone derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN112480015B for accelerating drug discovery and development. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this efficient synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to our deep reservoir of process knowledge and our commitment to quality. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for success.
