Advanced Visible-Light Synthesis of 1-Benzyl-3,4-dihydroisoquinoline Intermediates for Scalable Pharma Manufacturing
Introduction to Patent CN114874139A: A Green Breakthrough in Isoquinoline Chemistry
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing nitrogen-containing heterocycles, particularly the tetrahydroisoquinoline scaffold which serves as the core skeleton for numerous bioactive alkaloids and therapeutic agents. Patent CN114874139A introduces a transformative synthetic strategy that leverages visible light photocatalysis to achieve the α-allylation or benzylation of tetrahydroisoquinolines. Unlike traditional methods that often rely on harsh oxidative conditions or expensive transition metals, this invention utilizes an azo-aryl protecting group strategy combined with mild violet light irradiation (390-395 nm). This approach not only simplifies the operational complexity but also aligns with the growing demand for green chemistry principles in the manufacturing of high-purity pharmaceutical intermediates. By enabling the direct functionalization of the Csp3-H bond adjacent to the nitrogen atom under ambient temperature conditions, this technology offers a reliable pathway for producing diverse 1-benzyl or 1-allyl 3,4-dihydroisoquinoline derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct α-functionalization of tertiary amines like tetrahydroisoquinoline has been predominantly achieved through Cross-Dehydrogenative Coupling (CDC) strategies. While effective in academic settings, these conventional protocols frequently necessitate the use of stoichiometric amounts of strong oxidants, such as peroxides or hypervalent iodine reagents, which generate significant chemical waste and pose safety hazards during scale-up. Furthermore, many established methods require elevated temperatures or inert atmospheres that increase energy consumption and operational costs. The reliance on transition metal catalysts, often based on copper or iron, introduces the risk of heavy metal residues in the final active pharmaceutical ingredient (API), necessitating costly and time-consuming purification steps to meet stringent regulatory limits. These factors collectively hinder the economic viability and environmental sustainability of producing complex isoquinoline intermediates on a commercial scale.
The Novel Approach
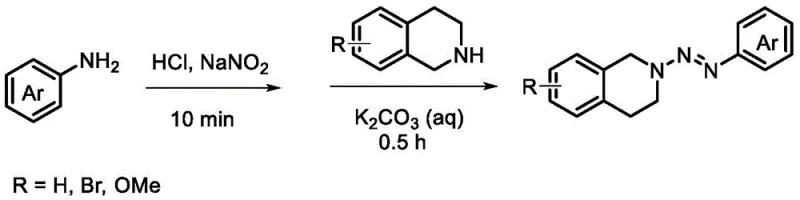
In stark contrast, the methodology disclosed in CN114874139A circumvents these challenges by employing a visible-light-induced radical coupling mechanism. The process utilizes readily available arylazo-protected tetrahydroisoquinolines as stable substrates, which react with benzyl or allyl bromides in the presence of a mild base (K2CO3) and a phase transfer catalyst (18-Crown-6). The reaction proceeds efficiently at room temperature (25°C) in acetonitrile solvent under nitrogen atmosphere, driven solely by purple LED light sources. This elimination of external oxidants and transition metal catalysts represents a paradigm shift towards cleaner synthesis. The use of an azo protecting group not only activates the specific C-H bond for functionalization but also stabilizes the intermediate, allowing for a broader substrate scope including various substituted benzyl bromides and allyl bromides without compromising yield or selectivity.
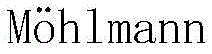
Mechanistic Insights into Visible-Light Induced Radical Coupling
The core innovation of this technology lies in the formation of an Electron Donor-Acceptor (EDA) complex between the azo-protected tetrahydroisoquinoline and the base under visible light irradiation. Upon absorption of photons in the 390-395 nm range, the EDA complex undergoes homolytic cleavage or single electron transfer to generate reactive radical species. Specifically, the light energy facilitates the activation of the Csp3-H bond at the α-position of the nitrogen atom, generating a carbon-centered radical that is sufficiently nucleophilic to attack the electrophilic benzyl or allyl bromide. This radical-radical or radical-nucleophile coupling occurs with high regioselectivity, ensuring that the substitution happens exclusively at the desired C1 position of the dihydroisoquinoline ring. The mildness of the photochemical conditions prevents the degradation of sensitive functional groups, such as methoxy or halogen substituents, which are often present in advanced pharmaceutical intermediates.
Furthermore, the stability of the starting materials plays a crucial role in the overall efficiency of the process. The azo-aryl protected tetrahydroisoquinolines are synthesized via a straightforward diazotization and coupling sequence involving aniline derivatives and tetrahydroisoquinoline in aqueous media. This precursor synthesis is robust, yielding stable yellow solids that can be isolated and stored, thereby decoupling the preparation of the starting material from the photocatalytic step. This modularity enhances supply chain reliability, as the key intermediates can be produced in bulk and utilized as needed. The subsequent photocatalytic step generates minimal byproducts, primarily inorganic salts, which are easily removed during the aqueous workup, resulting in a crude product profile that is significantly cleaner than those obtained from oxidative CDC methods.
How to Synthesize 1-Benzyl-3,4-dihydroisoquinoline Efficiently
The practical implementation of this synthesis involves a two-stage process designed for maximum operational simplicity and yield optimization. First, the azo-protected tetrahydroisoquinoline is prepared by reacting the corresponding aniline with sodium nitrite and hydrochloric acid at 0°C to form the diazonium salt, which is then coupled with tetrahydroisoquinoline in the presence of potassium carbonate. After isolation and recrystallization, this stable intermediate is subjected to the photocatalytic conditions. In a typical procedure, the protected amine is dissolved in acetonitrile along with the alkyl halide partner, potassium carbonate, and 18-Crown-6. The mixture is degassed and sealed under nitrogen to prevent oxygen quenching of the radical species. Detailed standardized synthesis steps for specific derivatives follow below.
- Preparation of Azo-Protected Tetrahydroisoquinoline: Diazotize an aryl amine with NaNO2/HCl at 0°C, then couple with tetrahydroisoquinoline in aqueous K2CO3 to form the stable azo-intermediate.
- Photocatalytic Coupling Reaction: Combine the azo-intermediate with benzyl or allyl bromide in acetonitrile with K2CO3 and 18-Crown-6 under nitrogen atmosphere.
- Visible Light Irradiation and Workup: Irradiate the mixture with 390-395 nm violet LED light for 36 hours at room temperature, followed by aqueous quenching and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this visible-light mediated synthesis offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio; the process relies on commodity chemicals such as potassium carbonate, acetonitrile, and commercially available benzyl bromides, rather than specialized oxidants or precious metal catalysts. This shift significantly reduces the cost of goods sold (COGS) by eliminating the procurement of expensive reagents and the associated logistics of handling hazardous oxidizing agents. Moreover, the absence of transition metals removes the need for specialized scavenging resins or complex filtration systems typically required to meet heavy metal specifications in API manufacturing, leading to substantial cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and transition metal catalysts directly lowers reagent costs and waste disposal fees. Since the reaction proceeds at room temperature using low-energy LED lights, energy consumption is minimized compared to thermal processes requiring reflux or cryogenic cooling. The simplified workup procedure, involving basic aqueous quenching and extraction, reduces solvent usage and labor hours, contributing to a more lean and cost-effective production model that enhances overall margin potential for high-volume intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial commodities ensures a resilient supply chain less susceptible to geopolitical disruptions or market volatility associated with rare earth metals or specialized reagents. The stability of the azo-protected intermediates allows for strategic stockpiling, buffering against potential raw material shortages. Additionally, the mild reaction conditions reduce equipment wear and tear, extending the lifecycle of reactor vessels and lighting arrays, which supports consistent production schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable using modern flow chemistry reactors or large-scale LED arrays, facilitating the commercial scale-up of complex alkaloid derivatives. The green chemistry profile, characterized by the absence of toxic heavy metals and hazardous oxidants, simplifies regulatory compliance and environmental permitting. This eco-friendly attribute aligns with corporate sustainability goals and reduces the burden of wastewater treatment, making the process highly attractive for long-term manufacturing partnerships in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing clarity on reaction parameters, substrate compatibility, and downstream processing. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this route into existing production pipelines.
Q: What are the advantages of using visible light catalysis over traditional CDC methods for isoquinoline synthesis?
A: Visible light catalysis eliminates the need for stoichiometric oxidants and expensive transition metal catalysts required in Cross-Dehydrogenative Coupling (CDC). This results in a greener process with simpler workup, lower raw material costs, and reduced heavy metal contamination risks in the final pharmaceutical intermediate.
Q: Is the azo-protection group stable enough for large-scale storage and handling?
A: Yes, the patent data indicates that the azo-aryl protected tetrahydroisoquinoline intermediates are structurally stable solids that can be prepared simply and stored. This stability allows for a modular synthesis approach where the protected precursor can be stockpiled before the final photocatalytic step.
Q: What is the typical yield and purity profile for this visible-light mediated alkylation?
A: The disclosed method achieves yields up to 63% for complex substrates like 1-(2-methallyl)-3,4-dihydroisoquinoline under mild conditions (25°C). The mild reaction environment minimizes side reactions, facilitating the production of high-purity intermediates suitable for downstream drug synthesis without extensive purification burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyl-3,4-dihydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light photocatalysis in modernizing the synthesis of complex nitrogen heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114874139A are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical clients. We are committed to delivering high-quality isoquinoline intermediates that adhere to the highest standards of safety and efficacy.
We invite you to collaborate with our technical team to explore how this green synthesis route can optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise in photocatalytic chemistry can accelerate your drug development timeline while ensuring a sustainable and reliable supply of critical intermediates.
