Scalable Visible-Light Synthesis of 1-Benzyl-3,4-Dihydroisoquinoline Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways to access complex nitrogen-containing heterocycles, which serve as the core scaffolds for numerous bioactive alkaloids and therapeutic agents. Patent CN114874139A introduces a groundbreaking methodology for the synthesis of 1-benzyl or allyl 3,4-dihydroisoquinoline derivatives, leveraging visible-light photocatalysis to overcome the limitations of traditional thermal processes. This innovation specifically targets the functionalization of the C1 position of tetrahydroisoquinoline, a transformation that is historically challenging due to the inertness of the Csp3-H bond. By employing an arylazo protective group strategy combined with violet light irradiation (390-395 nm), the inventors have established a green, operationally simple route that proceeds under mild conditions without the need for stoichiometric oxidants or transition metal catalysts. This technical advancement represents a significant leap forward for manufacturers of pharmaceutical intermediates, offering a robust alternative to legacy methods that often suffer from poor atom economy and harsh reaction environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct α-allylation or benzylation of tertiary amines like tetrahydroisoquinoline has relied heavily on Cross-Dehydrogenative Coupling (CDC) strategies. These conventional approaches typically necessitate the use of stoichiometric amounts of strong oxidants, such as peroxides or hypervalent iodine reagents, to generate the reactive iminium ion intermediates required for nucleophilic attack. Such conditions not only pose significant safety hazards in a manufacturing setting due to the potential for exothermic runaway reactions but also generate substantial quantities of chemical waste, complicating downstream purification and environmental compliance. Furthermore, many traditional methods require elevated temperatures or specialized transition metal catalysts that can be prohibitively expensive and difficult to remove to the stringent parts-per-million levels required for high-purity pharmaceutical intermediates. The reliance on moisture-sensitive reagents and inert atmospheres further escalates the operational complexity and cost, creating bottlenecks in the commercial scale-up of complex alkaloid scaffolds.
The Novel Approach
In stark contrast, the methodology disclosed in CN114874139A utilizes a visible-light-induced electron donor-acceptor (EDA) complex mechanism to activate the Csp3-H bond adjacent to the nitrogen atom. By temporarily protecting the tetrahydroisoquinoline nitrogen with an arylazo group, the electronic properties of the molecule are tuned to facilitate single-electron transfer upon irradiation with low-energy violet light. This photo-induced activation generates a radical species at the C1 position, which subsequently undergoes coupling with benzyl or allyl bromides in the presence of a mild base like potassium carbonate. The reaction proceeds efficiently at room temperature (25°C) in acetonitrile, eliminating the need for external heating or cooling systems and significantly reducing energy consumption. This approach not only simplifies the operational workflow but also enhances the safety profile by avoiding hazardous oxidants, thereby providing a streamlined pathway for cost reduction in pharmaceutical intermediate manufacturing.
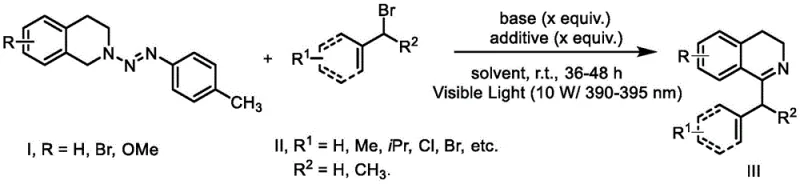
The versatility of this photochemical protocol is demonstrated by its broad substrate scope, accommodating various substituents on both the isoquinoline ring and the benzyl/allyl coupling partners. As illustrated in the reaction scheme, the process tolerates electron-donating groups like methoxy as well as electron-withdrawing halogens, allowing for the rapid diversification of the chemical library. This flexibility is crucial for medicinal chemists who require access to diverse analogues for structure-activity relationship (SAR) studies during drug discovery phases. The use of simple, commercially available starting materials such as substituted anilines and tetrahydroisoquinolines ensures that the supply chain remains resilient and cost-effective. Moreover, the reaction conditions are compatible with standard laboratory glassware and LED light sources, making the technology easily transferable from bench-scale research to pilot plant operations without the need for specialized high-pressure or high-temperature reactors.
Mechanistic Insights into Visible-Light Induced C-H Activation
The core of this innovation lies in the formation of an Electron Donor-Acceptor (EDA) complex between the azo-protected tetrahydroisoquinoline and the base or solvent system, which absorbs visible light to initiate the radical cascade. Upon irradiation with 390-395 nm light, the excited state of the EDA complex facilitates a single-electron transfer (SET) process, generating a radical cation intermediate. Subsequent deprotonation at the C1 position yields a nucleophilic carbon-centered radical, which is sufficiently reactive to couple with the electrophilic benzyl or allyl bromide. This radical-radical or radical-polar crossover mechanism bypasses the high energy barriers associated with thermal C-H activation, allowing the reaction to proceed under ambient conditions. The presence of 18-Crown-6 as an additive plays a critical role in solubilizing the potassium carbonate base in the organic phase, enhancing the efficiency of the deprotonation step and stabilizing the ionic species involved in the catalytic cycle. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal yield and selectivity.
Impurity control is another critical aspect addressed by this mechanism. Traditional oxidative methods often lead to over-oxidation byproducts or polymerization of the amine substrate, resulting in complex impurity profiles that are difficult to separate. In the visible-light mediated process, the mild nature of the radical generation minimizes side reactions, leading to a cleaner crude reaction mixture. The azo protecting group not only activates the C-H bond but also prevents N-alkylation side reactions, ensuring that the substitution occurs exclusively at the desired C1 position. Following the coupling reaction, the azo group can be readily removed or transformed, yielding the target 1-substituted 3,4-dihydroisoquinoline with high structural fidelity. This inherent selectivity reduces the burden on downstream purification steps, such as column chromatography or crystallization, thereby improving the overall mass balance and throughput of the manufacturing process. For supply chain heads, this translates to more predictable production timelines and reduced material loss.
How to Synthesize 1-Benzyl-3,4-Dihydroisoquinoline Efficiently
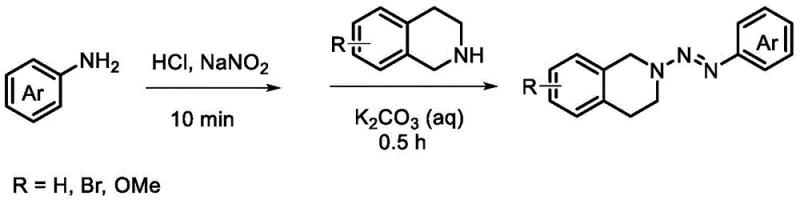
The synthesis of these valuable heterocyclic compounds begins with the preparation of the key azo-protected intermediate, which serves as the photo-active substrate for the subsequent coupling reaction. This two-step sequence ensures high reproducibility and allows for the isolation and characterization of the intermediate before proceeding to the light-driven step. Detailed standard operating procedures for the preparation of the intermediate and the final photocoupling reaction are essential for maintaining consistency across different production batches. The following guide outlines the critical stages involved in executing this synthesis effectively, emphasizing the importance of light intensity, reaction time, and reagent stoichiometry.
- Prepare the azo-protected tetrahydroisoquinoline intermediate by reacting tetrahydroisoquinoline with an aryl diazonium salt in the presence of potassium carbonate at 0°C.
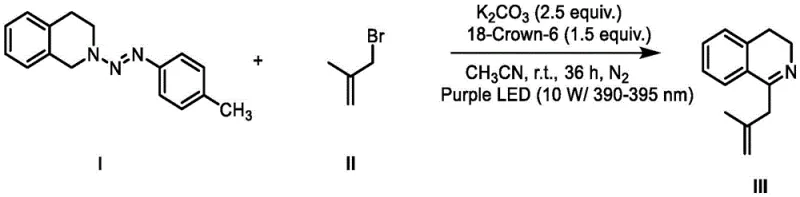
- Combine the protected intermediate with benzyl or allyl bromide, potassium carbonate (2.5 equiv.), and 18-Crown-6 (1.5 equiv.) in acetonitrile solvent under a nitrogen atmosphere.
- Irradiate the reaction mixture with purple LED light (390-395 nm) at room temperature for 36 hours, then quench with water and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this visible-light synthesis method offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of expensive transition metal catalysts and stoichiometric oxidants directly impacts the bill of materials, leading to significant cost savings in raw material procurement. Furthermore, the use of commodity chemicals like potassium carbonate and acetonitrile ensures a stable and diversified supply base, mitigating the risks associated with sourcing specialized reagents that may be subject to market volatility or geopolitical constraints. The operational simplicity of running reactions at room temperature under LED illumination also reduces the dependency on complex utility infrastructure, such as steam for heating or chilled brine for cooling, which contributes to lower overhead costs in manufacturing facilities.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates the need for costly metal scavenging steps and rigorous testing for residual metals, which are mandatory for pharmaceutical grade products. Additionally, the high atom economy of the radical coupling reaction minimizes waste generation, reducing the expenses associated with waste disposal and environmental compliance. The ability to run reactions in standard glassware or stainless steel reactors equipped with simple LED arrays further lowers the capital expenditure required for process implementation, making it an attractive option for both small-scale API production and large-volume intermediate manufacturing.
- Enhanced Supply Chain Reliability: By relying on widely available starting materials such as substituted anilines and tetrahydroisoquinolines, manufacturers can establish robust supply chains that are less susceptible to disruptions. The mild reaction conditions also enhance process safety, reducing the likelihood of accidents or shutdowns due to thermal runaways, thereby ensuring consistent delivery schedules. The scalability of the photochemical process, supported by the linear relationship between light exposure and reaction progress, allows for seamless translation from laboratory to commercial scale, ensuring that supply commitments can be met reliably even as demand fluctuates.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as the use of visible light as a traceless reagent and the avoidance of toxic oxidants, align perfectly with modern environmental regulations and corporate sustainability goals. The reduced generation of hazardous waste simplifies the permitting process for new manufacturing lines and lowers the long-term liability associated with chemical handling. Moreover, the energy efficiency of LED lighting compared to traditional thermal heating methods contributes to a lower carbon footprint for the manufacturing process, enhancing the company's reputation as a responsible and sustainable reliable pharmaceutical intermediate supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on reaction scope, safety considerations, and scalability potential. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: What are the key advantages of this visible-light method over traditional CDC strategies?
A: Unlike traditional Cross-Dehydrogenative Coupling (CDC) methods that often require stoichiometric oxidants and harsh conditions, this patent utilizes visible light and an azo-protecting group to enable mild, room-temperature activation without expensive metal catalysts.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process uses commercially available raw materials like tetrahydroisoquinoline and simple bromides, operates at room temperature with standard LED lights, and avoids hazardous oxidants, making it highly scalable and safe for industrial production.
Q: What is the typical yield and purity profile for these dihydroisoquinoline derivatives?
A: The patent reports yields reaching up to 63% for specific derivatives like 1-(2-methallyl)-3,4-dihydroisoquinoline, with products characterized by high purity via NMR and HRMS, suitable for downstream medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyl-3,4-Dihydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photochemical technologies in modern organic synthesis and are committed to delivering high-quality heterocyclic intermediates to the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114874139A can be successfully translated into robust industrial processes. We maintain stringent purity specifications across all our product lines, supported by state-of-the-art rigorous QC labs equipped with advanced analytical instrumentation to verify identity and potency. Whether you require custom synthesis of novel dihydroisoquinoline derivatives or reliable supply of established intermediates, our infrastructure is designed to meet the demanding requirements of the life sciences industry.
We invite you to collaborate with us to explore how this green synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, highlighting the economic benefits of switching to this visible-light methodology. Please contact our technical procurement team today to request specific COA data for our available inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in driving efficiency and innovation in pharmaceutical intermediate manufacturing.
