Advanced Iodide-Catalyzed Amide Synthesis for Scalable Pharmaceutical Intermediate Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing essential structural motifs, among which the amide bond remains paramount. Patent CN102584509A introduces a groundbreaking methodology for the preparation of amides that fundamentally shifts the paradigm from traditional transition metal catalysis to a more economical and environmentally benign iodide-catalyzed system. This innovation leverages a decarbonylation diradical cross-coupling reaction between aldehyde derivatives and formamide derivatives, utilizing tert-butyl hydroperoxide (TBHP) as the oxidant. By replacing expensive and toxic precious metals with readily available iodides, this technology offers a compelling solution for the scalable manufacturing of high-purity pharmaceutical intermediates. The significance of this patent lies not only in its chemical novelty but also in its potential to drastically reduce the cost of goods sold (COGS) for complex amide-containing active pharmaceutical ingredients (APIs) and their precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of amide bonds has relied heavily on methodologies that present significant logistical and economic challenges for large-scale industrial application. Traditional approaches often necessitate the use of carboxylic acid derivatives coupled with amines, which frequently require harsh activation reagents that generate substantial chemical waste. Furthermore, transition metal-catalyzed carbonylation reactions, while effective, typically depend on catalysts based on Ruthenium, Rhodium, Palladium, or Nickel. These metals are not only prohibitively expensive due to market volatility but also pose severe toxicity concerns that mandate rigorous and costly purification steps to meet residual metal specifications in final drug products. Additionally, many conventional methods suffer from narrow substrate scopes, requiring specific protecting groups or suffering from poor functional group tolerance, which complicates synthetic route design and increases the number of processing steps required to reach the target molecule.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102584509A utilizes a robust iodide-catalyzed oxidative coupling strategy that operates under remarkably mild conditions. The core of this innovation is the use of simple iodide salts, such as sodium iodide, potassium iodide, or tetrabutylammonium iodide, which act as efficient catalysts in conjunction with TBHP. This system enables the direct coupling of aldehydes and formamides through a radical mechanism, bypassing the need for pre-functionalized substrates or expensive metal complexes. 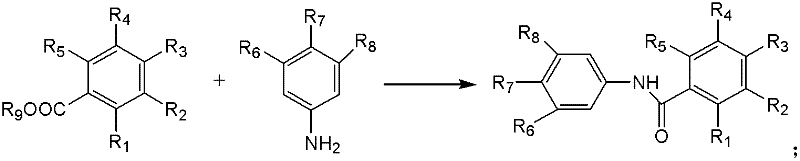
The reaction proceeds smoothly at temperatures ranging from 60°C to 100°C, often achieving excellent yields within 2 to 24 hours. This approach significantly simplifies the operational complexity, as the catalysts are commercially available commodities, and the work-up procedure involves standard quenching and extraction techniques followed by simple column chromatography. By eliminating the reliance on precious metals, this novel approach directly addresses the critical pain points of cost reduction in pharmaceutical intermediates manufacturing and enhances the overall sustainability profile of the synthesis.
Mechanistic Insights into Iodide-Catalyzed Decarbonylation Diradical Cross-Coupling
The mechanistic underpinning of this transformation is a sophisticated decarbonylation diradical cross-coupling process that distinguishes it from classical nucleophilic acyl substitution. In this catalytic cycle, the iodide species interacts with the oxidant TBHP to generate reactive radical intermediates. Crucially, the formamide derivative undergoes a decarbonylation event to generate an amino radical species, which then couples with the acyl radical derived from the aldehyde oxidation. This dual radical generation allows for the formation of the amide bond without the need for external activating agents. The use of TBHP as a terminal oxidant ensures that the reaction drives forward efficiently, regenerating the active iodine species and maintaining the catalytic cycle. This mechanism is particularly advantageous because it tolerates a wide array of functional groups on the aromatic ring of the aldehyde, including electron-withdrawing groups like nitro and cyano, as well as electron-donating groups like methoxy and methyl.
From an impurity control perspective, this radical pathway offers distinct advantages over ionic mechanisms that might be prone to side reactions such as over-oxidation or polymerization. The specificity of the radical coupling minimizes the formation of by-products, leading to cleaner reaction profiles and higher crude purity. 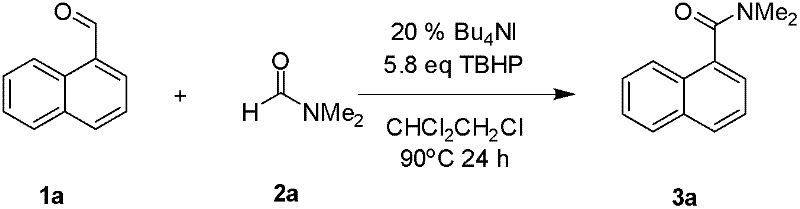
For instance, the conversion of 1-naphthaldehyde to N,N-dimethyl-1-naphthamide proceeds with high efficiency, demonstrating the system's capability to handle sterically demanding substrates. The ability to control the radical flux through the careful selection of iodide catalyst loading (typically 5-40 mol%) and oxidant equivalents (preferably 5.8 equivalents) allows process chemists to fine-tune the reaction for optimal selectivity. This level of control is essential for ensuring consistent quality in the commercial scale-up of complex pharmaceutical intermediates, where batch-to-batch reproducibility is a regulatory requirement.
How to Synthesize N,N-Dimethyl-1-naphthamide Efficiently
The practical implementation of this iodide-catalyzed protocol is straightforward and amenable to standard laboratory and pilot plant equipment. The synthesis typically involves charging a reaction vessel with the aldehyde substrate, the formamide derivative, the iodide catalyst, and the oxidant in a chlorinated or aromatic solvent. The mixture is then heated under aerobic conditions, allowing the radical coupling to proceed to completion. Following the reaction, a simple aqueous work-up removes inorganic salts and excess oxidant, and the product is isolated via crystallization or chromatography.
- Charge the reaction vessel with iodide catalyst (e.g., Bu4NI, 20mol%), aldehyde derivative, TBHP oxidant, and formamide derivative in a suitable solvent like 1,2-dichloroethane.
- Heat the reaction mixture in air at temperatures between 60°C and 100°C for a duration of 2 to 24 hours to facilitate the decarbonylation diradical cross-coupling.
- Quench the reaction with saturated sodium sulfite, extract with ethyl acetate, and purify the crude product via simple column chromatography to obtain the target amide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodide-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material supply chain. Unlike precious metal catalysts which are subject to geopolitical supply risks and extreme price fluctuations, iodide salts are abundant, stable, and sourced from a robust global market. This stability ensures consistent pricing and availability, mitigating the risk of production delays caused by raw material shortages. Furthermore, the elimination of heavy metals from the process removes the necessity for specialized scavenging resins or complex filtration steps, which are often bottlenecks in manufacturing timelines. This streamlining of the downstream processing directly contributes to enhanced supply chain reliability and shorter lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with commodity iodides results in significant raw material cost savings. Additionally, the simplified work-up procedure reduces solvent consumption and waste disposal costs, as there is no need for specialized treatment of heavy metal-contaminated waste streams. The mild reaction conditions also lower energy consumption compared to high-temperature or high-pressure alternatives, further driving down the operational expenditure per kilogram of product. These cumulative effects create a highly competitive cost structure for the manufacturing of amide-based intermediates.
- Enhanced Supply Chain Reliability: By utilizing commercially available reagents that are not subject to strict export controls or limited mining outputs, manufacturers can secure a more resilient supply chain. The robustness of the catalytic system means that minor variations in reagent quality do not critically impact the reaction outcome, providing a buffer against supply chain variability. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with various substrates under consistent conditions. The use of TBHP and iodides generates less hazardous waste compared to stoichiometric coupling reagents, aligning with increasingly stringent environmental regulations. The ability to operate at atmospheric pressure and moderate temperatures reduces the safety risks associated with high-pressure reactors, facilitating easier technology transfer from lab to plant and ensuring compliance with modern safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodide-catalyzed amide synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for process evaluation.
Q: What are the primary advantages of using iodide catalysts over traditional transition metals for amide synthesis?
A: Iodide catalysts such as sodium iodide or tetrabutylammonium iodide are significantly less expensive and less toxic than traditional precious metal catalysts like Ruthenium, Rhodium, or Palladium. This substitution eliminates the need for complex heavy metal removal steps, simplifying post-treatment and reducing overall production costs while adhering to stricter environmental regulations.
Q: What is the substrate scope for this iodide-catalyzed oxidative coupling reaction?
A: The method demonstrates high functional group compatibility, successfully converting various aldehyde derivatives including naphthyl, heterocyclic, alkenyl, and monosubstituted aryl aldehydes. It also accommodates diverse formamide derivatives, making it a versatile platform for synthesizing complex amide structures found in pharmaceutical intermediates.
Q: How does the reaction mechanism ensure high atom efficiency?
A: The process utilizes a decarbonylation diradical cross-coupling mechanism where formamide serves as both the nitrogen source and the carbonyl source via decarbonylation. This direct coupling of aldehydes and formamides avoids the generation of stoichiometric waste associated with activating agents used in traditional amidation, thereby aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amides Supplier
The technological advancements described in CN102584509A represent a significant leap forward in the efficient production of amide derivatives, yet realizing their full potential requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of oxidative coupling reactions, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity pharmaceutical intermediates meets the exacting standards required by global regulatory bodies.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for fine chemical intermediates.
