Scalable Synthesis of Bioactive 2,3-Dihydroquinolones via Safe Palladium-Catalyzed Carbonylation
Scalable Synthesis of Bioactive 2,3-Dihydroquinolones via Safe Palladium-Catalyzed Carbonylation
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN112239456B introduces a robust methodology for the preparation of substituted 2,3-dihydroquinolone compounds, a class of molecules renowned for their significant antitumor and analgesic properties. As illustrated in the structural diversity of known bioactive agents, the 2,3-dihydroquinolone core serves as a critical pharmacophore in various therapeutic candidates. This patent discloses a transition metal palladium-catalyzed carbonylation reaction that utilizes N-pyridine sulfonyl-o-iodoaniline and olefins as primary building blocks. By employing a solid carbon monoxide substitute, the process circumvents the severe safety hazards associated with traditional high-pressure CO gas protocols, thereby offering a safer and more operationally convenient pathway for the production of high-purity pharmaceutical intermediates.
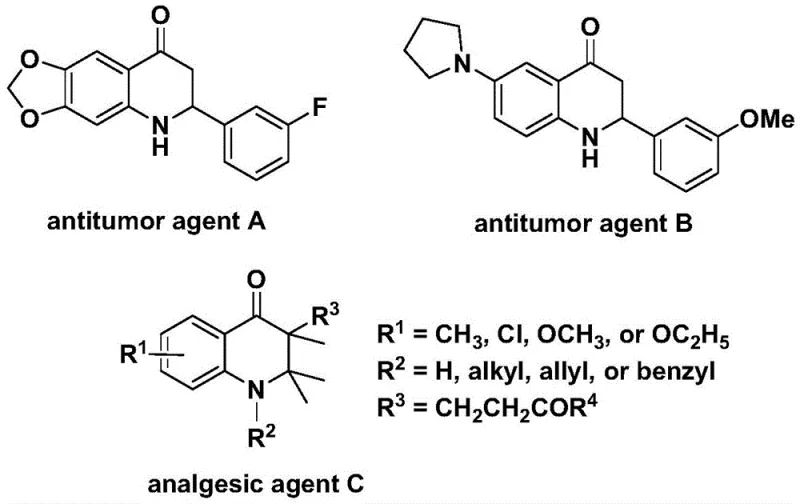
The strategic importance of this synthesis lies in its ability to construct the six-membered heterocyclic ring with high precision while tolerating a wide array of functional groups. For research directors overseeing process development, the ability to access these complex skeletons through a modular approach is invaluable. The method described allows for the systematic variation of substituents at the 2-position, enabling the rapid generation of analog libraries for structure-activity relationship (SAR) studies. Furthermore, the use of readily available starting materials ensures that the supply chain for these critical intermediates remains stable and cost-effective, addressing a key concern for procurement managers seeking reliable sources for complex organic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has often relied on direct carbonylation using carbon monoxide gas. While chemically effective, this approach presents substantial logistical and safety challenges in an industrial setting. The requirement for high-pressure reactors and specialized gas handling infrastructure significantly increases capital expenditure and operational complexity. Moreover, the toxicity of carbon monoxide necessitates rigorous safety monitoring and containment protocols, which can slow down production timelines and increase the risk profile of the manufacturing process. Alternative methods involving pre-functionalized acid chlorides or anhydrides often suffer from poor atom economy and generate stoichiometric amounts of salt waste, complicating downstream purification and environmental compliance. These factors collectively hinder the scalable production of these valuable intermediates, creating bottlenecks in the supply of active pharmaceutical ingredients.
The Novel Approach
The methodology outlined in CN112239456B represents a paradigm shift by utilizing 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide surrogate. This innovation effectively decouples the carbonylation reaction from the need for gaseous CO, allowing the reaction to proceed under standard atmospheric pressure conditions in common organic solvents like 1,4-dioxane. The reaction mechanism involves the in situ release of carbon monoxide from the TFBen molecule, which is then immediately consumed by the palladium catalytic cycle. This "bottled CO" strategy not only enhances safety but also improves the controllability of the reaction, leading to consistent yields across different substrate classes. The process is designed to be simple to operate, requiring only standard heating and stirring equipment, which drastically lowers the barrier to entry for manufacturing facilities lacking high-pressure capabilities.
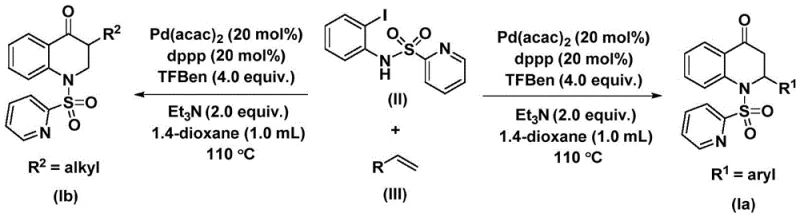
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The catalytic cycle driving this transformation is a sophisticated orchestration of organometallic steps that ensure high chemoselectivity and efficiency. The process initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide molecule, released thermally from the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate is the key precursor to the carbonyl functionality found in the final 2,3-dihydroquinolone product. The precise control over CO insertion is vital to prevent side reactions such as homocoupling of the aryl halide, which would otherwise degrade the overall yield and purity of the product.
Subsequent coordination and migratory insertion of the olefin substrate into the acyl-palladium bond extends the carbon skeleton and establishes the stereochemistry at the 2-position of the quinolone ring. The nature of the olefin, whether it be an electron-rich aryl alkene or a simple aliphatic alkene, influences the rate of this insertion step, yet the catalyst system demonstrates remarkable tolerance. Finally, an intramolecular nucleophilic attack by the nitrogen atom onto the carbonyl carbon, followed by reductive elimination, closes the heterocyclic ring and regenerates the active palladium catalyst. This mechanistic pathway ensures that the nitrogen atom from the starting aniline is seamlessly incorporated into the final heterocycle, maintaining high atom economy. The use of the pyridine sulfonyl group acts as both a protecting group and a directing element, stabilizing intermediates and preventing unwanted polymerization of the olefin.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The experimental protocol detailed in the patent provides a clear roadmap for executing this carbonylation reaction with high reproducibility. The procedure emphasizes the importance of precise stoichiometry, particularly the ratio of the palladium catalyst to the ligand and the CO source, to maximize turnover numbers. Operators are instructed to combine bis(acetylacetone)palladium, the dppp ligand, triethylamine, and the TFBen CO surrogate in 1,4-dioxane before introducing the substrates. This order of addition helps to pre-form the active catalytic species, ensuring a smooth initiation of the reaction upon heating. The reaction is typically conducted at 110 °C for a duration of 48 hours to ensure complete conversion of the starting iodoaniline, as shorter reaction times may lead to incomplete consumption of materials and lower isolated yields.
- Charge a reaction vessel with Pd(acac)2 catalyst, dppp ligand, triethylamine additive, and the solid CO source TFBen in 1,4-dioxane solvent.
- Add the starting material N-pyridine sulfonyl-o-iodoaniline and the specific olefin substrate (aryl or alkyl) to the mixture under inert atmosphere.
- Heat the reaction mixture to 110 °C and stir for 48 hours, followed by filtration and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The elimination of high-pressure carbon monoxide gas removes a significant regulatory and safety burden from the manufacturing facility, potentially lowering insurance costs and simplifying site audits. Furthermore, the reliance on commercially available starting materials such as olefins and substituted iodoanilines ensures a robust supply chain that is less susceptible to the volatility often seen with specialized reagents. The simplicity of the post-treatment process, which involves standard filtration and column chromatography, reduces the demand for complex separation technologies, thereby streamlining the production workflow and reducing labor costs associated with purification.
- Cost Reduction in Manufacturing: The substitution of hazardous CO gas with a solid surrogate like TFBen eliminates the need for expensive high-pressure autoclaves and specialized gas delivery infrastructure. This capital expenditure saving is compounded by the reduced need for extensive safety monitoring systems and emergency response protocols associated with toxic gas handling. Additionally, the high atom economy of the carbonylation reaction minimizes raw material waste, leading to more efficient use of reagents and lower disposal costs for chemical byproducts. The ability to run the reaction in common solvents like dioxane further simplifies solvent recovery and recycling processes, contributing to overall operational cost efficiency.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, including various substituted olefins and iodoanilines, are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source vendor issues. The stability of the solid CO surrogate also simplifies logistics, as it can be stored and transported without the stringent regulations applicable to compressed gases. Consequently, manufacturers can maintain higher inventory levels of critical reagents with greater ease, ensuring continuous production schedules and reliable delivery of the final pharmaceutical intermediates to downstream clients.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions make this process highly amenable to scale-up from gram to kilogram and ton scales. The simplified waste profile, characterized primarily by organic solvents and solid residues rather than heavy metal sludge or toxic gas scrubber waste, facilitates easier compliance with environmental regulations. The robustness of the catalyst system allows for consistent performance even at larger volumes, reducing the risk of batch failures during technology transfer. This scalability ensures that the method can meet the growing demand for 2,3-dihydroquinolone derivatives in the pharmaceutical market without requiring significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this carbonylation protocol. Understanding these nuances is essential for process chemists aiming to adapt this method for specific target molecules. The answers are derived directly from the experimental data and mechanistic insights provided in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: What is the primary advantage of using TFBen over carbon monoxide gas?
A: Using 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas cylinders and specialized containment equipment, significantly enhancing laboratory and plant safety while simplifying the operational workflow.
Q: What types of olefin substrates are compatible with this carbonylation method?
A: The method demonstrates broad substrate compatibility, successfully accommodating both substituted aryl olefins to form 2-aryl derivatives and alkyl or silyl-substituted olefins to form 3-alkyl derivatives, allowing for diverse structural modifications.
Q: What are the typical reaction conditions required for high conversion?
A: Optimal conversion is achieved using Pd(acac)2 and dppp ligands in 1,4-dioxane at 110 °C for approximately 48 hours, ensuring complete consumption of the iodoaniline starting material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug discovery and development pipeline. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,3-dihydroquinolone intermediate meets the highest industry standards. Our commitment to safety and efficiency aligns perfectly with the advanced synthetic methodologies described in recent patents, allowing us to offer competitive pricing without compromising on quality or delivery timelines.
We invite you to collaborate with us to leverage this innovative carbonylation technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your development timeline and optimize your supply chain costs.
