Scalable Palladium-Catalyzed Synthesis of 3-Arylquinolinone Derivatives for Commercial Production
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that serve as critical backbones in drug discovery, particularly for quinolinone derivatives known for their potent biological activities. Patent CN113045489B introduces a groundbreaking preparation method for 3-arylquinoline-2(1H) ketone derivatives, utilizing a sophisticated palladium-catalyzed aminocarbonylation strategy that redefines efficiency in organic synthesis. This innovation leverages benzisoxazole as a dual-function reagent, acting simultaneously as a nitrogen source and a formyl source, which drastically streamlines the synthetic pathway compared to traditional multi-step approaches. The significance of this chemical architecture is underscored by its presence in high-value therapeutic agents, ranging from MAP Kinase inhibitors to HBV inhibitors, highlighting the urgent demand for reliable manufacturing processes.
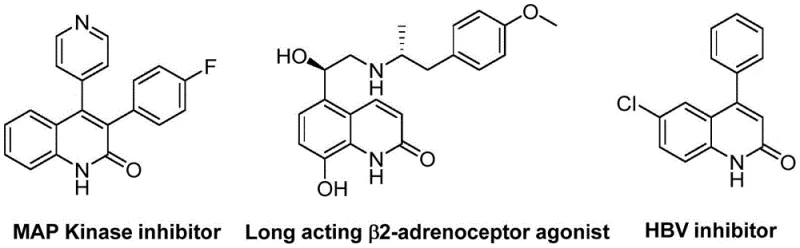
For R&D directors and procurement specialists, the ability to access these complex intermediates through a simplified, high-yielding process represents a strategic advantage in pipeline development. The method described in the patent not only ensures high reaction efficiency but also maintains excellent compatibility with a broad spectrum of functional groups, thereby reducing the risk of side reactions that often plague large-scale production. By adopting this technology, pharmaceutical manufacturers can secure a more stable supply chain for essential intermediates while minimizing the environmental footprint associated with wasteful synthetic steps. This report delves into the technical nuances and commercial implications of this novel methodology, providing a comprehensive analysis for stakeholders aiming to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H) ketone derivatives has relied heavily on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions, which often impose significant constraints on process scalability and operational safety. These traditional routes frequently require harsh reaction conditions, including extreme temperatures or the use of corrosive reagents, which can lead to equipment degradation and increased maintenance costs over time. Furthermore, conventional methods often suffer from limited substrate tolerance, necessitating extensive protection and deprotection steps that elongate the production timeline and accumulate chemical waste. The reliance on multiple discrete steps also introduces additional points of failure, where yield losses at each stage compound to result in suboptimal overall efficiency and higher cost of goods sold. For supply chain managers, these inefficiencies translate into longer lead times and greater vulnerability to raw material price fluctuations, making the search for alternative pathways a critical priority.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN113045489B utilizes a transition metal-catalyzed carbonylation coupling reaction that fundamentally simplifies the construction of the quinolinone core. By employing benzisoxazole and benzyl chloride compounds as starting materials, this method bypasses the need for pre-functionalized precursors, allowing for a more direct assembly of the target molecular architecture. The integration of a palladium catalyst system with carbonyl molybdenum facilitates a smooth aminocarbonylation process under relatively mild conditions, specifically at 100°C, which enhances energy efficiency and operational safety. This streamlined protocol not only reduces the number of unit operations required but also minimizes the generation of hazardous byproducts, aligning with modern green chemistry principles. For procurement teams, this translates to a more predictable manufacturing schedule and reduced dependency on complex, hard-to-source reagents, ultimately strengthening the resilience of the supply chain.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The core of this technological breakthrough lies in the intricate catalytic cycle driven by palladium acetate and the chiral ligand (S)-1,1'-binaphthyl-2,2'-bisdiphenylphosphine, commonly known as (S)-BINAP. In this system, the palladium catalyst activates the benzyl chloride substrate through oxidative addition, forming a reactive organopalladium intermediate that is poised for carbonyl insertion. The presence of carbonyl molybdenum serves as a solid carbon monoxide source, releasing CO in situ to facilitate the carbonylation step without the need for handling hazardous gaseous CO directly. This controlled release mechanism ensures a steady concentration of carbonyl species in the reaction mixture, promoting high conversion rates while maintaining safety standards suitable for industrial environments. The subsequent coordination and insertion of the benzisoxazole nitrogen atom close the ring structure, finalizing the formation of the 3-arylquinoline-2(1H) ketone scaffold with high regioselectivity.
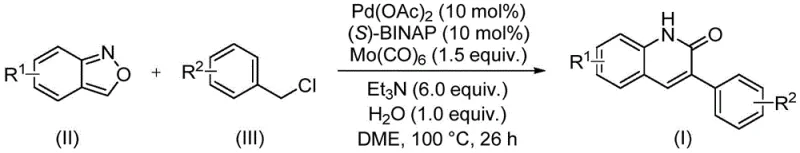
Beyond the primary catalytic cycle, the reaction design incorporates triethylamine and water as essential additives that play crucial roles in stabilizing intermediates and facilitating proton transfer steps. The wide functional group tolerance observed in this system is attributed to the mild nature of the catalytic species, which avoids aggressive interactions with sensitive moieties such as esters, nitriles, or halogens present on the aromatic rings. This chemoselectivity is paramount for R&D directors aiming to synthesize diverse libraries of analogs for structure-activity relationship studies without encountering compatibility issues. Moreover, the use of ethylene glycol dimethyl ether (DME) as the solvent provides an optimal medium for dissolving both organic substrates and inorganic catalysts, ensuring homogeneous reaction conditions that are vital for consistent batch-to-batch reproducibility. Understanding these mechanistic details allows technical teams to fine-tune parameters for maximum yield and purity during scale-up operations.
How to Synthesize 3-Arylquinoline-2(1H) Ketone Efficiently
Implementing this synthesis route requires precise adherence to the optimized molar ratios and reaction conditions established in the patent data to ensure successful outcomes. The process begins with the careful weighing and mixing of palladium acetate, (S)-BINAP, carbonyl molybdenum, triethylamine, and water alongside the primary substrates, benzisoxazole and benzyl chloride compounds. It is critical to maintain the reaction temperature at 100°C within a sealed vessel to prevent solvent loss and maintain the necessary pressure for efficient carbonylation over the 26-hour duration. Following the reaction completion, the workup procedure involves filtration to remove insoluble catalyst residues, followed by silica gel mixing to prepare the crude mixture for purification. The final isolation of the pure 3-arylquinoline-2(1H) ketone derivative is achieved through column chromatography, a standard technique that ensures the removal of any trace impurities to meet stringent pharmaceutical quality standards.
- Prepare the reaction mixture by combining palladium acetate, (S)-BINAP, carbonyl molybdenum, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the sealed reaction vessel to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Perform post-processing via filtration and silica gel mixing, followed by column chromatography purification to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis method offers substantial strategic benefits for organizations focused on cost optimization and supply chain reliability in the pharmaceutical sector. The utilization of benzisoxazole and benzyl chloride as starting materials is particularly advantageous because these chemicals are commercially available at low costs and are widely produced by upstream suppliers, reducing the risk of raw material shortages. By eliminating the need for complex multi-step sequences associated with traditional methods, manufacturers can significantly reduce labor costs, energy consumption, and solvent usage, leading to a leaner and more economical production process. This efficiency gain is further amplified by the high reaction yields reported across various substrate examples, which minimizes material waste and maximizes the output per batch. For procurement managers, these factors combine to create a compelling value proposition that supports long-term budget planning and competitive pricing strategies in the global market.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot reaction design eliminates the need for intermediate isolation and purification steps, which are traditionally resource-intensive and costly. By reducing the number of processing stages, facilities can lower their operational overheads related to equipment usage, utility consumption, and waste disposal fees. Additionally, the use of inexpensive and readily available catalysts and reagents ensures that the variable costs per kilogram of product remain low, even when scaling to commercial volumes. This economic efficiency allows companies to reinvest savings into further R&D initiatives or pass on cost benefits to clients, enhancing overall market competitiveness without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing simplicity is a key driver for supply chain stability, and this method relies on commodity chemicals that are less susceptible to geopolitical disruptions or market volatility. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without requiring highly specialized infrastructure or rare equipment. This flexibility enables supply chain heads to diversify their manufacturing base and establish redundant production capabilities, ensuring continuous availability of critical intermediates for downstream drug formulation. Furthermore, the reduced lead time associated with a shorter synthetic route allows for faster response to market demand fluctuations, improving customer satisfaction and service levels.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that can be safely translated from laboratory benchtop to industrial reactor scales without significant re-optimization. The avoidance of hazardous gaseous carbon monoxide by using solid carbonyl molybdenum enhances workplace safety and simplifies regulatory compliance regarding emissions and worker exposure. Moreover, the high atom economy and reduced solvent requirements align with increasingly strict environmental regulations, helping companies meet sustainability goals and avoid potential fines or shutdowns. This forward-looking approach to manufacturing not only protects the environment but also future-proofs the production asset against tightening global chemical management policies.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and capabilities of this synthesis technology, we have compiled a set of answers based on the detailed experimental data provided in the patent documentation. These questions cover critical aspects such as reagent functionality, substrate scope, and operational parameters that are essential for technical teams evaluating this route for adoption. Understanding these specifics helps in making informed decisions about process integration and resource allocation within existing manufacturing frameworks. The following insights are derived directly from the validated examples and claims within the intellectual property, ensuring accuracy and relevance for professional assessment.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl source, which significantly simplifies the reaction setup and reduces the need for multiple expensive reagents.
Q: Is this catalytic system tolerant to diverse functional groups?
A: Yes, the patented method demonstrates wide substrate functional group tolerance, allowing for the synthesis of various derivatives with different substituents like halogens, alkoxy, and cyano groups.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds at 100°C in ethylene glycol dimethyl ether (DME) for about 26 hours using a palladium catalyst system with Mo(CO)6 as the carbonyl source.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline-2(1H) Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality pharmaceutical intermediates through innovative and scalable synthetic routes. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards for identity, potency, and impurity profiles. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are equipped to handle complex chemistries like the palladium-catalyzed aminocarbonylation described in this report with precision and reliability.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development needs, ensuring a competitive edge in time-to-market and cost efficiency. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements and project timelines. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Partnering with us means securing a dependable source for high-purity 3-arylquinoline-2(1H) ketone derivatives, supported by a team dedicated to your success in the global marketplace.
