Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using DMF as Carbon Source for Pharmaceutical Applications
Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using DMF as Carbon Source for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing privileged heterocyclic scaffolds, particularly those incorporating fluorine motifs which are critical for metabolic stability and bioavailability. A recent technological breakthrough documented in patent CN114920707B introduces a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This innovation leverages the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a reaction medium but as a vital carbon source, thereby streamlining the synthetic pathway. The significance of this development cannot be overstated for R&D teams targeting complex drug candidates, as the 1,2,4-triazole core is a fundamental structural element found in numerous bioactive molecules, including Factor IXa inhibitors and GlyT1 inhibitors. By utilizing a molecular iodine-promoted tandem cyclization under air atmosphere, this process eliminates the need for expensive transition metal catalysts or harsh anhydrous conditions, representing a paradigm shift towards greener and more economical pharmaceutical intermediate manufacturing.
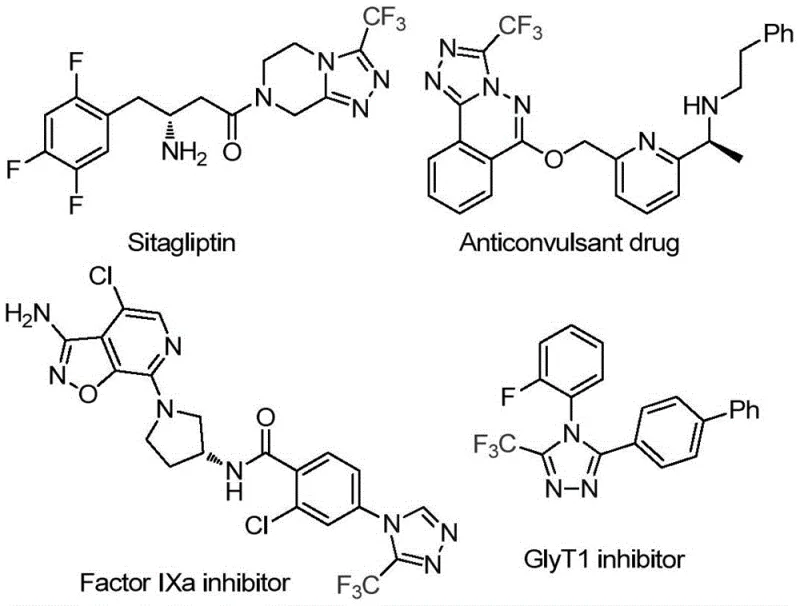
The strategic value of this synthesis lies in its ability to access diverse chemical space with minimal operational friction. As illustrated in the provided structural examples, the resulting triazole derivatives serve as key building blocks for next-generation therapeutics. The patent details a protocol where readily available trifluoroethyliminohydrazide reacts with DMF in the presence of iodine to yield the target heterocycles with high efficiency. For procurement managers and supply chain directors, this translates to a reliable pharmaceutical intermediate supplier capability that mitigates the risks associated with complex multi-step syntheses. The method's reliance on commodity chemicals like DMF and iodine ensures that raw material sourcing remains stable and cost-predictable, avoiding the volatility often seen with specialized organometallic reagents. Furthermore, the broad substrate scope allows for the rapid generation of analog libraries, accelerating the lead optimization phase in drug discovery programs without compromising on scalability or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder large-scale production. Conventional routes often rely on the condensation of hydrazines with nitriles or amidines, which frequently necessitate the use of toxic cyanide sources or unstable intermediates that pose significant safety hazards in a plant setting. Moreover, many established protocols require stringent reaction conditions, such as strictly anhydrous solvents and inert gas protection (nitrogen or argon), to prevent side reactions or catalyst deactivation. These requirements impose a heavy burden on infrastructure, demanding specialized glass-lined reactors and extensive purification steps to remove trace metals or hazardous byproducts. Additionally, the introduction of the trifluoromethyl group often involves separate fluorination steps using expensive reagents like Selectfluor or trifluoromethyl iodide, which drastically inflates the cost of goods sold (COGS). The cumulative effect of these limitations is a process that is difficult to scale, environmentally burdensome due to high E-factor waste generation, and economically inefficient for the production of high-purity pharmaceutical intermediates needed for clinical trials and commercial launch.
The Novel Approach
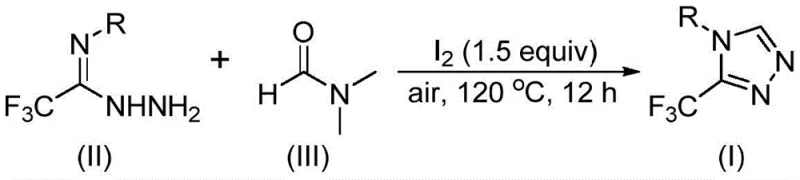
In stark contrast, the methodology disclosed in patent CN114920707B offers a streamlined solution that addresses these pain points through ingenious reaction design. The core innovation is the dual functionality of DMF, which acts simultaneously as the solvent and the C1 synthon, effectively collapsing multiple synthetic steps into a single pot operation. This approach utilizes molecular iodine as a mild and inexpensive promoter to activate the DMF, facilitating a tandem cyclization that constructs the triazole ring directly from trifluoroethyliminohydrazide. The reaction proceeds smoothly at temperatures between 110°C and 130°C under ambient air, completely removing the need for inert atmosphere handling. This operational simplicity is a game-changer for cost reduction in pharmaceutical intermediate manufacturing, as it allows for the use of standard stainless steel reactors without specialized drying systems. The versatility of the method is further evidenced by its compatibility with a wide range of functional groups, enabling the synthesis of diverse derivatives such as those shown in the reaction scheme below, which includes substrates with halogen, alkyl, and alkoxy substituents.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, this transformation represents a fascinating interplay between halogen activation and solvent participation. The reaction initiates with the interaction of molecular iodine with DMF, which activates the solvent molecule towards nucleophilic attack. Depending on the specific pathway, either the formyl group or the N-methyl group of DMF serves as the carbon source for the ring closure. In the formyl pathway, the carbonyl carbon undergoes condensation with the hydrazide moiety to form a hydrazone intermediate, followed by intramolecular cyclization and elimination of dimethylamine to yield the aromatic triazole system. Alternatively, when the N-methyl group participates, iodine facilitates the formation of an amine salt species which subsequently reacts with the hydrazide via nucleophilic addition. This pathway involves the elimination of N-methylformamide to generate an azadiene intermediate, which then undergoes oxidative aromatization to finalize the heterocyclic structure. Understanding these mechanistic nuances is critical for R&D directors aiming to optimize reaction parameters, as it highlights the importance of iodine stoichiometry (typically 1.5 equivalents) in driving the equilibrium towards the desired product while minimizing the formation of oligomeric impurities.
Impurity control is another critical aspect where this mechanism offers distinct advantages over transition-metal catalyzed alternatives. Since the process relies on main-group elements (iodine) rather than precious metals like palladium or copper, the risk of heavy metal contamination in the final API is virtually eliminated. This simplifies the downstream purification process, often reducing the number of crystallization or chromatography steps required to meet stringent regulatory specifications. The oxidative aromatization step, driven by air oxygen in conjunction with iodine, ensures that the final triazole ring is fully aromatic and stable, preventing the accumulation of partially reduced byproducts that could complicate the impurity profile. For quality assurance teams, this means a cleaner crude reaction mixture and a more robust process capable of consistently delivering high-purity 3-trifluoromethyl-1,2,4-triazoles suitable for sensitive biological applications. The ability to tune the electronic properties of the substrate without affecting the core cyclization mechanism further underscores the robustness of this chemical platform.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the precise weighing of trifluoroethyliminohydrazide and molecular iodine, which are then suspended in DMF. It is crucial to maintain the reaction temperature within the optimal window of 110°C to 130°C; temperatures below this range may result in incomplete conversion, while excessive heat could lead to solvent decomposition. The reaction mixture is stirred under air for a duration of 10 to 15 hours, allowing sufficient time for the tandem cyclization and aromatization events to reach completion. Monitoring the reaction progress via TLC or HPLC is recommended to determine the exact endpoint for specific substrates. Upon completion, the workup procedure is remarkably straightforward, involving simple filtration to remove inorganic salts followed by standard extraction and drying protocols. For detailed operational specifics and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Charge a reaction vessel with molecular iodine (1.5 equivalents), trifluoroethyliminohydrazide substrate, and DMF solvent.
- Heat the mixture to 110-130°C under air atmosphere and stir for 10-15 hours to facilitate the tandem cyclization.
- Upon completion, filter the mixture, wash, dry, and purify via silica gel column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary driver of value is the drastic simplification of the bill of materials (BOM). By replacing expensive, specialized C1 reagents and transition metal catalysts with commodity-grade DMF and iodine, the direct material costs are significantly reduced. This substitution also mitigates supply chain risks, as DMF and iodine are produced on a massive global scale with stable pricing and availability, unlike niche fluorinating agents which can be subject to market volatility. Furthermore, the elimination of anhydrous and anaerobic requirements translates to substantial capital expenditure (CapEx) savings, as existing general-purpose reactors can be utilized without modification. This flexibility enhances production scheduling and reduces lead times, allowing manufacturers to respond more agilely to fluctuating demand from downstream API producers. The environmental profile is also improved, as the process generates less hazardous waste and avoids the use of toxic heavy metals, aligning with increasingly strict global sustainability mandates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the replacement of high-cost reagents with low-cost solvents. Traditional methods often require stoichiometric amounts of expensive fluorinating agents or precious metal catalysts, which constitute a major portion of the variable cost. By utilizing DMF as both solvent and reactant, the process effectively monetizes a waste stream (solvent) into a value-added component of the product. Additionally, the simplified workup reduces the consumption of purification media such as silica gel and organic solvents, further driving down the cost per kilogram. The absence of noble metals also removes the need for costly metal scavenging steps, which are often required to meet ppm-level specifications for pharmaceutical ingredients. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on product quality or performance.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available raw materials. DMF is a staple solvent in the chemical industry, ensuring that production is never halted due to raw material shortages. Similarly, molecular iodine is a widely traded commodity with a robust supply network. This contrasts sharply with processes dependent on custom-synthesized intermediates or reagents sourced from single suppliers, which create single points of failure. The operational simplicity of the reaction—requiring only air and heat—also reduces the dependency on specialized utility infrastructure like nitrogen generators or solvent drying plants. This makes the technology transferable to a wider range of manufacturing sites, including those in emerging markets, thereby diversifying the supply base and reducing geopolitical risks. Consequently, partners can expect more consistent delivery schedules and greater flexibility in order volumes.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is inherently safer and more manageable due to the benign nature of the reagents and conditions. The exotherm of the reaction is moderate and easily controlled, posing minimal risk of thermal runaway compared to highly reactive fluorination chemistries. From an environmental standpoint, the process aligns well with green chemistry principles by maximizing atom economy and minimizing the use of hazardous substances. The avoidance of heavy metals simplifies wastewater treatment and solid waste disposal, reducing the environmental compliance burden and associated fees. The high conversion rates observed across various substrates mean that less unreacted starting material needs to be recovered or disposed of, further enhancing the overall sustainability profile. This makes the process not only commercially attractive but also future-proof against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of what partners can expect during technology transfer and scale-up. Understanding these nuances helps in setting accurate expectations for project timelines and resource allocation.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or methyl group), which drastically simplifies the reagent list and reduces raw material costs compared to traditional methods requiring separate C1 synthons.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, significantly lowering equipment and operational complexity.
Q: What is the substrate scope for the R-group in this reaction?
A: The method demonstrates broad substrate tolerance, successfully accommodating various substituted aryl groups including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, trifluoromethyl) substituents at ortho, meta, and para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted cyclization technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to seamlessly integrate this novel pathway into our existing manufacturing portfolio. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to commercial reactor is smooth and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and purity of every batch. Our facility is equipped to handle the specific thermal and material requirements of this DMF-based process, guaranteeing consistent quality and supply continuity for your critical drug development programs.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your next-generation therapeutics. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team to request specific COA data for our standard triazole intermediates and to discuss route feasibility assessments for your custom projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to accelerating your time-to-market while optimizing your manufacturing costs.
