Scalable Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using DMF as a Dual-Function Reagent
Introduction to Advanced Triazole Manufacturing
The pharmaceutical and agrochemical industries continuously demand efficient routes to fluorinated heterocycles, particularly 1,2,4-triazoles, which serve as privileged scaffolds in modern drug design. As illustrated in the structural diversity of bioactive molecules such as Sitagliptin and various Factor IXa inhibitors, the incorporation of a trifluoromethyl group onto the triazole core significantly enhances metabolic stability and lipophilicity. Patent CN114920707B discloses a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that fundamentally shifts the paradigm of heterocyclic synthesis. By leveraging the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium but as an active carbon source, this technology offers a streamlined, atom-economical approach that addresses long-standing challenges in process chemistry.
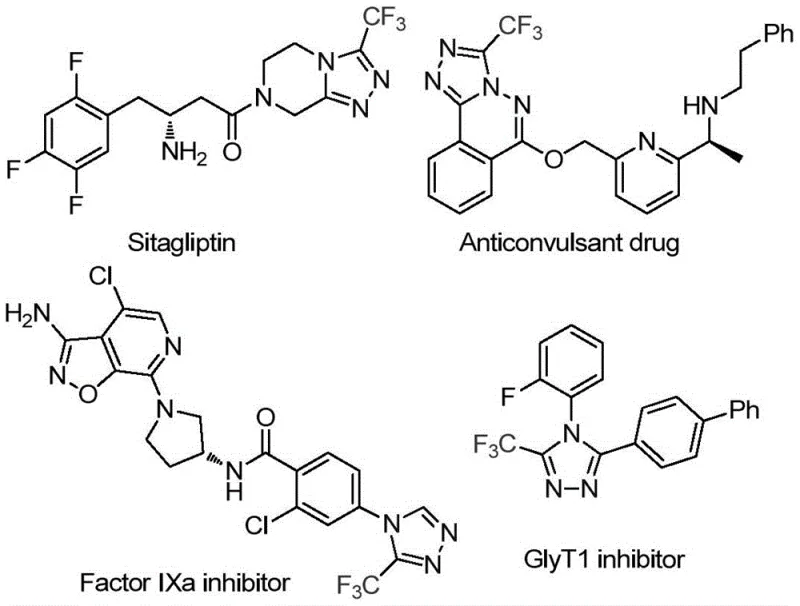
This innovation is particularly relevant for R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships. The method eliminates the dependency on specialized, moisture-sensitive reagents often required for traditional triazole construction. Instead, it utilizes commercially available trifluoroethyliminohydrazide and molecular iodine under aerobic conditions. This transition from complex, multi-step sequences to a direct tandem cyclization represents a significant leap forward in cost reduction in API manufacturing, enabling the rapid generation of diverse libraries for drug discovery while maintaining high standards of purity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-trifluoromethyl-1,2,4-triazoles has relied on cyclocondensation reactions between hydrazides and nitriles or amidines, often necessitating harsh thermal conditions or the use of toxic, expensive catalysts. Traditional protocols frequently require strict anhydrous and anaerobic environments to prevent the decomposition of sensitive intermediates, thereby imposing substantial infrastructure costs on production facilities. Furthermore, many existing routes utilize stoichiometric amounts of activating agents that generate significant chemical waste, complicating downstream purification and environmental compliance. The reliance on specialized C1 donors, distinct from the reaction solvent, adds another layer of logistical complexity and raw material expense, making scale-up economically challenging for high-volume applications.
The Novel Approach
In stark contrast, the methodology described in CN114920707B introduces a remarkably simple yet powerful strategy where the solvent itself participates in the bond-forming event. The reaction employs molecular iodine as a mild promoter to activate DMF, facilitating a tandem cyclization that constructs the triazole ring in a single operational step. As demonstrated by the substrate scope shown below, this approach exhibits exceptional tolerance for various functional groups, including electron-donating and electron-withdrawing substituents on the aromatic ring.
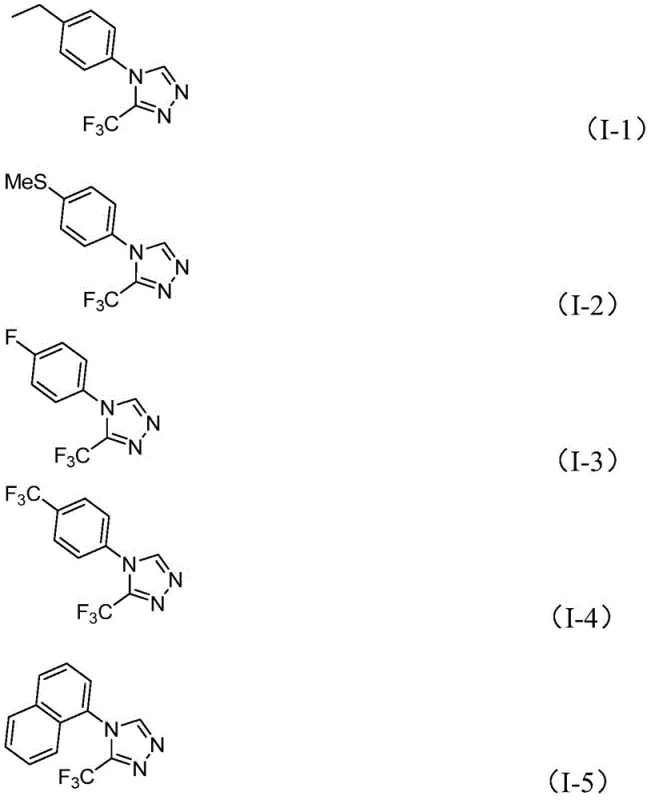
The ability to operate under air atmosphere at moderate temperatures (110-130°C) drastically simplifies the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. By merging the roles of solvent and reactant, the process minimizes the total mass intensity of the reaction, leading to reduced waste generation and lower solvent recovery costs. This dual-functionality design is a hallmark of green chemistry principles, offering a sustainable alternative that aligns with modern regulatory expectations for cleaner production technologies in the fine chemical sector.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the versatile activation of DMF by molecular iodine, which unlocks two potential pathways for carbon incorporation depending on which fragment of the DMF molecule participates. In the first pathway, the formyl group of DMF acts as the carbon source, undergoing condensation with the trifluoroethyliminohydrazide to form a hydrazone intermediate. This is followed by an intramolecular cyclization and the elimination of dimethylamine to yield the target 3-trifluoromethyl-1,2,4-triazole. Alternatively, the N-methyl group of DMF can serve as the synthon; here, iodine activates the solvent to form an amine salt, which subsequently reacts with the hydrazide via nucleophilic addition. This pathway involves the elimination of N-methylformamide to generate an azadiene intermediate, which then undergoes oxidative aromatization to finalize the heterocyclic core.
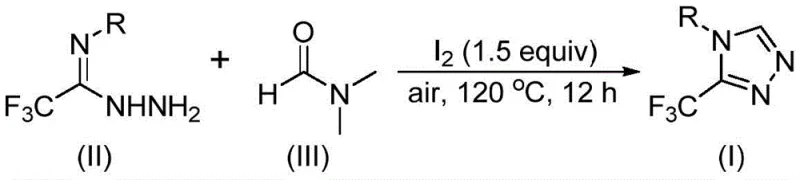
Understanding these mechanistic nuances is critical for impurity control and process optimization. The use of iodine as a promoter ensures high reaction efficiency without the need for transition metals that could contaminate the final API. The oxidative aromatization step, driven by air oxygen, further enhances the atom economy of the process. For quality assurance teams, this mechanism implies a clean impurity profile dominated primarily by unreacted starting materials or simple hydrolysis byproducts, which are easily removed via standard silica gel chromatography or crystallization. This predictability in the reaction outcome is essential for establishing robust control strategies during technology transfer from laboratory to pilot plant scales.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring minimal specialized equipment while delivering consistent results across a broad range of substrates. The protocol leverages the high boiling point and solvating power of DMF to maintain homogeneous reaction conditions throughout the heating cycle. Detailed standard operating procedures regarding exact stoichiometry, workup parameters, and purification techniques are critical for ensuring batch-to-batch reproducibility.
- Charge a reaction vessel with molecular iodine (1.5 equivalents) and trifluoroethyliminohydrazide substrate in an air atmosphere.
- Add organic solvent DMF (acting as both solvent and reactant) to dissolve the starting materials completely.
- Heat the mixture to 110-130°C for 10-15 hours, then perform standard post-treatment including filtration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this DMF-based synthesis route offers transformative benefits that extend beyond mere chemical yield. The reliance on commodity chemicals—specifically DMF and molecular iodine—decouples production from the volatility associated with specialized reagent markets. Since DMF is one of the most widely produced polar aprotic solvents globally, its availability is virtually guaranteed, ensuring supply continuity even during periods of market disruption. This stability allows procurement managers to negotiate long-term contracts with favorable pricing structures, effectively insulating the manufacturing budget from raw material price spikes.
- Cost Reduction in Manufacturing: The elimination of expensive, dedicated C1 building blocks and the removal of inert gas requirements result in substantial cost savings. By utilizing the solvent as a reactant, the process reduces the overall material bill, while the simplified workup decreases labor and utility consumption associated with complex purification trains. These efficiencies compound at scale, driving down the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction under air atmosphere removes the need for nitrogen or argon blanketing systems, reducing capital expenditure on reactor modifications. Furthermore, the broad substrate tolerance means that a single manufacturing line can be adapted to produce various analogues simply by changing the starting hydrazide, enhancing asset utilization and flexibility in responding to changing customer demands.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts simplifies waste stream management and reduces the burden on wastewater treatment facilities. The process generates fewer hazardous byproducts compared to traditional methods, aligning with increasingly stringent environmental regulations. This 'green' profile not only mitigates regulatory risk but also enhances the brand value of the end-product in markets that prioritize sustainable sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis platform. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF serves a dual role as both the polar aprotic solvent and the critical C1 carbon source, eliminating the need for separate, expensive formylating agents and simplifying the reaction stoichiometry.
Q: Does this process require inert gas protection?
A: No, the reaction proceeds efficiently under an air atmosphere, which significantly reduces operational complexity and equipment costs compared to traditional anaerobic methods.
Q: What is the substrate scope for the R group in this reaction?
A: The method tolerates a wide range of substituents including alkyl, alkoxy, alkylthio, halogens, and trifluoromethyl groups on the aromatic ring, allowing for diverse functionalization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 3-trifluoromethyl-1,2,4-triazole derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation.
We invite global partners to leverage our technical capabilities to optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how this innovative DMF-mediated synthesis can enhance your project's economic viability and speed to market.
