Advanced Asymmetric Synthesis of Chiral Gamma-Alkynyl Alpha-Keto Esters for Pharma
Advanced Asymmetric Synthesis of Chiral Gamma-Alkynyl Alpha-Keto Esters for Pharma
The landscape of modern pharmaceutical manufacturing is constantly evolving, driven by the demand for high-purity chiral building blocks that serve as critical precursors for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN114478245A, which discloses a robust and versatile asymmetric synthesis method for chiral γ-alkynyl-α-keto ester compounds. These compounds are invaluable organic synthons, capable of being transformed into a myriad of complex structures found in natural products and drug candidates. The innovation lies in a sophisticated catalytic system that not only achieves high yields and exceptional enantioselectivity but also offers the unique capability to selectively access either enantiomer through precise modulation of the catalyst components. This level of control is paramount for R&D directors seeking to optimize synthetic routes for active pharmaceutical ingredients (APIs) where stereochemistry dictates biological activity.
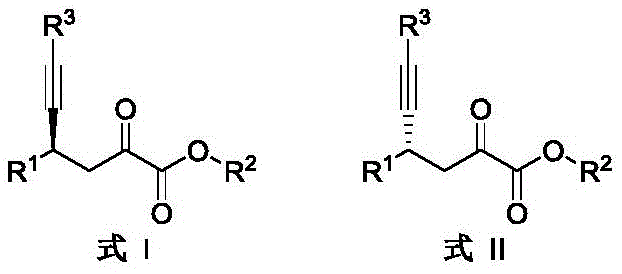
For procurement managers and supply chain heads, the implications of this technology extend beyond mere chemical elegance. The method utilizes readily available starting materials such as β,γ-unsaturated-α-ketoesters and terminal alkynes, coupled with a catalytic system comprising Lewis acids, silver salts, and chiral phosphoric acids. The operational simplicity, characterized by mild reaction temperatures ranging from 0°C to 60°C and the use of common solvents like dichloromethane, translates directly into reduced operational expenditures and enhanced process safety. Furthermore, the broad substrate scope ensures that this methodology can be applied to a wide array of derivatives without necessitating extensive re-optimization, thereby securing a stable and flexible supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral γ-alkynyl-α-ketoesters has been plagued by significant challenges that hinder their widespread adoption in industrial settings. Conventional methods primarily rely on the conjugate addition of terminal alkynyl groups to β,γ-unsaturated-α-carbonyl compounds. However, existing literature and prior art indicate that these traditional approaches suffer from severe limitations regarding substrate applicability. Many protocols are restricted to narrow classes of substrates, failing to tolerate diverse functional groups or steric hindrances often present in complex drug molecules. Additionally, achieving high enantioselectivity has frequently required harsh reaction conditions, expensive transition metal catalysts that are difficult to remove, or cryogenic temperatures that escalate energy costs. These factors collectively create bottlenecks in the manufacturing process, leading to inconsistent quality, higher impurity profiles, and ultimately, increased costs for the final API.
The Novel Approach
In stark contrast, the novel approach outlined in the patent introduces a dual-activation strategy that overcomes these historical barriers. By employing a synergistic combination of a Lewis acid, a silver salt, and a chiral phosphoric acid, the method activates both the nucleophile and the electrophile simultaneously, facilitating a highly efficient conjugate addition. A standout feature of this innovation is the switchable stereoselectivity; by merely adjusting the catalytic cocktail—specifically, the inclusion of a second silver salt and specific additives—the synthesis can be directed to produce either the S-configuration or the R-configuration enantiomer with high fidelity. This flexibility is a game-changer for process chemistry, allowing manufacturers to adapt quickly to changing regulatory or biological requirements without redesigning the entire synthetic route.

The reaction scheme demonstrates the streamlined nature of this process, where substrates are combined in a single pot under mild stirring conditions. The use of standard laboratory equipment and ambient pressure further underscores the scalability of this method. For a reliable pharmaceutical intermediate supplier, this means the ability to ramp up production from gram-scale discovery to multi-ton commercial manufacturing with minimal friction. The post-processing is equally straightforward, involving simple concentration and column chromatography, which avoids the need for complex work-up procedures that often lead to product loss or degradation. This efficiency ensures that the final product meets stringent purity specifications required by global regulatory bodies.
Mechanistic Insights into Lewis Acid and Chiral Phosphoric Acid Co-Catalysis
To fully appreciate the technical depth of this synthesis, one must delve into the mechanistic interplay between the catalysts. The core of this transformation relies on the cooperative catalysis between a Lewis acid (such as Indium Tribromide or Iron Tribromide) and a chiral phosphoric acid (CPA). The Lewis acid coordinates with the carbonyl oxygen of the β,γ-unsaturated-α-ketoester, significantly increasing its electrophilicity and rendering the β-position more susceptible to nucleophilic attack. Simultaneously, the chiral phosphoric acid, acting as a Brønsted acid, activates the terminal alkyne through hydrogen bonding or ion-pairing interactions, organizing the transition state within a chiral environment. This dual activation lowers the activation energy barrier while imposing strict stereochemical control, resulting in the observed high enantiomeric excess (ee) values, often exceeding 90%.
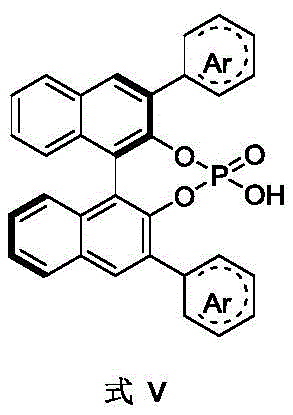
The structure of the chiral phosphoric acid, as depicted in Formula V, plays a pivotal role in defining the stereochemical outcome. The bulky aryl groups (Ar) attached to the binaphthyl backbone create a well-defined chiral pocket that discriminates between the two faces of the prochiral substrate. Substituents on these aryl rings, such as halogens or trifluoromethyl groups, can be tuned to optimize steric and electronic properties, further enhancing selectivity. Moreover, the addition of silver salts serves to generate a reactive silver-acetylide species in situ, which acts as the actual nucleophile. The precise stoichiometry of these components is critical; for instance, the presence of a second silver salt and additives like methanol or trifluoroethanol is essential for flipping the selectivity to the R-enantiomer, likely by altering the coordination geometry around the metal center or modifying the hydrogen-bonding network of the chiral catalyst.
How to Synthesize Chiral Gamma-Alkynyl Alpha-Keto Esters Efficiently
Implementing this synthesis in a practical setting requires adherence to specific procedural guidelines to maximize yield and optical purity. The general protocol involves charging a reactor with the unsaturated ketoester, the terminal alkyne, the chosen Lewis acid, silver salt(s), and the chiral phosphoric acid in an anhydrous solvent. The mixture is then stirred at a controlled temperature, typically room temperature, for a period sufficient to drive the reaction to completion, usually between 6 to 24 hours. Monitoring the reaction progress via TLC or HPLC is recommended to prevent over-reaction or decomposition. Once the starting material is consumed, the reaction mixture is concentrated, and the crude residue is purified via flash column chromatography. For a detailed breakdown of the specific molar ratios, solvent choices, and work-up procedures tailored to different substrates, please refer to the standardized synthesis steps provided below.
- Prepare the reaction vessel by adding beta,gamma-unsaturated-alpha-ketoester, terminal alkyne, Lewis acid (e.g., InBr3), silver salt, and chiral phosphoric acid in a suitable solvent like dichloromethane.
- Stir the mixture at a controlled temperature between 0°C and 60°C for a duration of 6 to 24 hours to ensure complete conversion and high enantioselectivity.
- Upon completion, concentrate the reaction mixture using a rotary evaporator and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this asymmetric synthesis method offers substantial strategic advantages for procurement and supply chain management. The primary benefit stems from the drastic simplification of the manufacturing process. Traditional routes often involve multiple protection-deprotection steps or the use of stoichiometric chiral auxiliaries, which generate significant waste and increase raw material costs. In contrast, this catalytic approach is atom-economical and generates minimal byproducts, leading to a significantly reduced environmental footprint and lower waste disposal costs. The ability to operate at mild temperatures eliminates the need for energy-intensive cooling or heating systems, further contributing to cost reduction in pharmaceutical intermediate manufacturing. These efficiencies translate into a more competitive pricing structure for the final intermediates without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric chiral reagents and the use of catalytic amounts of relatively inexpensive Lewis acids and silver salts result in substantial cost savings. The high yields reported, often reaching up to 90%, minimize the loss of valuable starting materials, ensuring that every kilogram of input translates effectively into output. Furthermore, the simplified downstream processing reduces the consumption of solvents and silica gel during purification, lowering the overall cost of goods sold (COGS). This economic efficiency is crucial for maintaining margins in the highly competitive generic and specialty pharma markets.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against varying substrate structures ensures a consistent supply of intermediates even when feedstock specifications fluctuate slightly. The use of common, commercially available solvents like dichloromethane and ethyl acetate mitigates the risk of supply disruptions associated with exotic or regulated reagents. Additionally, the short reaction times and ambient pressure conditions allow for faster batch turnover, enabling manufacturers to respond swiftly to urgent orders or changes in demand forecasts. This agility is a key factor in reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API production schedules are met without delay.
- Scalability and Environmental Compliance: The method is inherently scalable, having been demonstrated effectively on multi-millimole scales in the patent examples, with clear pathways to kilogram and ton-scale production. The mild reaction conditions reduce the safety risks associated with exothermic runaways or high-pressure operations, facilitating easier regulatory approval for commercial plants. Moreover, the high selectivity reduces the formation of diastereomeric impurities, simplifying the purification burden and reducing the volume of hazardous waste generated. This aligns with modern green chemistry principles and helps companies meet increasingly stringent environmental compliance standards, avoiding potential fines and reputational damage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What is the primary advantage of this synthesis method over conventional approaches?
A: The primary advantage is the ability to selectively synthesize either the S or R enantiomer by simply adjusting the catalytic system components, specifically the addition of a second silver salt and additives, overcoming the limited substrate scope of previous methods.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction operates under mild conditions, typically at room temperature (0-60°C) in common organic solvents like dichloromethane, with reaction times ranging from 6 to 24 hours, making it highly practical for scale-up.
Q: Can this method accommodate diverse substrate variations?
A: Yes, the method demonstrates excellent substrate applicability, tolerating various substituents on the aryl rings of both the ketoester and the alkyne, including halogens, alkyl groups, and ester functionalities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Gamma-Alkynyl Alpha-Keto Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team of expert chemists has extensively evaluated the technology described in patent CN114478245A and possesses the technical expertise to implement this advanced asymmetric synthesis at scale. We understand that transitioning a novel laboratory method to commercial production requires rigorous process optimization and quality control. Therefore, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure that your supply needs are met with unwavering consistency. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess, to guarantee that every batch meets the highest industry standards.
We invite you to collaborate with us to unlock the full potential of this synthesis method for your specific drug development programs. Whether you require custom synthesis of novel derivatives or large-scale supply of established intermediates, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your project requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your timeline and optimize your budget. Let us be your partner in delivering excellence in chiral chemistry.
