Advanced One-Step Synthesis of 5-Nitro-4,5-Dihydrofuran Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly those serving as pivotal intermediates for bioactive molecules. Patent CN101343263A introduces a transformative approach for synthesizing 5-nitro-4,5-dihydrofuran derivatives, a class of compounds renowned for their versatility in drug discovery and natural product synthesis. This innovation leverages manganese(III) acetylacetonate as a single-electron oxidant to facilitate a direct oxidative cyclization of various electron-deficient nitroalkenes. By shifting away from multi-step sequences that require harsh conditions and toxic reagents, this technology offers a streamlined, one-pot solution that operates effectively in ethanol at moderate temperatures between 30-70°C. For R&D directors and process chemists, this represents a significant leap forward in atom economy and operational simplicity, providing a reliable pathway to access diverse chemical libraries with high purity and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 5-nitro-4,5-dihydrofuran derivatives was largely constrained by the methodology reported by E.V. Trukhin, which relied on a cumbersome two-step process involving Michael addition followed by cyclization. As illustrated in the historical reaction scheme below, this legacy approach necessitated the use of beta,beta-dinitrostyrene, a specialized and less accessible starting material, reacting with acetylacetone under strictly controlled low-temperature conditions followed by a prolonged room-temperature incubation.
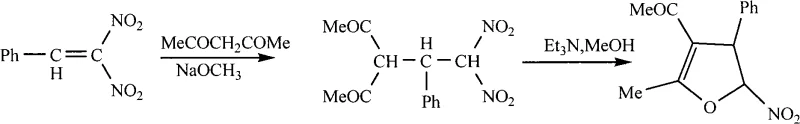
The conventional route suffered from severe drawbacks, including a total yield of only approximately 56 percent and the generation of nitrous acid as a hazardous byproduct, posing significant environmental and safety challenges during scale-up. Furthermore, the requirement for sodium methoxide and triethylamine introduced additional purification burdens to remove basic residues, while the extended reaction time of up to two days severely impacted throughput. These factors collectively rendered the traditional method economically unviable for large-scale manufacturing, creating a bottleneck for procurement teams seeking cost-effective sources of these critical intermediates.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a direct oxidative coupling strategy that merges the carbon-carbon bond formation and ring closure into a single operational step. The general reaction scheme demonstrates how readily available beta-nitrostyrenes and their heterocyclic analogs react directly with manganese(III) acetylacetonate in ethanol to furnish the target dihydrofuran core.
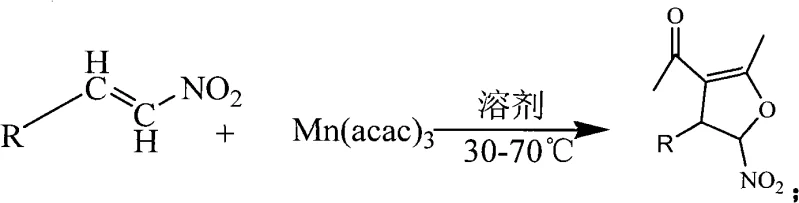
This innovative route eliminates the need for isolating unstable intermediates and avoids the use of strong bases or toxic solvents, instead employing ethanol as a green, renewable reaction medium. The process operates efficiently at 55°C, completing within just 1 to 4 hours, which drastically reduces energy consumption and reactor occupancy time. By expanding the substrate scope to include various substituted aromatic and heteroaromatic nitroalkenes, this method provides unparalleled flexibility for medicinal chemists to explore structure-activity relationships without being limited by synthetic feasibility, thereby accelerating the timeline from lead identification to candidate selection.
Mechanistic Insights into Mn(acac)3-Mediated Oxidative Cyclization
The core of this technological breakthrough lies in the radical-mediated mechanism facilitated by the manganese(III) species, which acts as both an oxidant and a Lewis acid promoter. The reaction initiates with the coordination of the manganese center to the enolizable beta-dicarbonyl equivalent or the nitroalkene, generating a radical species that undergoes intramolecular attack on the electron-deficient double bond. This radical cyclization is highly regioselective, driven by the stabilization of the transition state through the nitro group and the aromatic substituent, ensuring the formation of the 4,5-dihydrofuran ring with precise stereochemical control. The mild thermal conditions prevent the degradation of sensitive functional groups, allowing for the tolerance of halogens, alkoxy groups, and even additional nitro substituents on the aromatic ring.
From an impurity control perspective, the single-step nature of the reaction significantly minimizes the formation of side products typically associated with multi-step syntheses, such as polymerization of the nitroalkene or over-oxidation of the furan ring. The use of ethanol as a protic solvent further aids in quenching reactive radical intermediates and stabilizing the transition states, leading to cleaner reaction profiles as evidenced by the high isolated yields ranging from 74 percent to 94 percent across various substrates. This mechanistic elegance translates directly to simplified downstream processing, as the crude reaction mixtures contain fewer impurities, reducing the load on purification columns and enhancing the overall mass balance of the manufacturing process.
How to Synthesize 5-Nitro-4,5-Dihydrofuran Derivatives Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the efficiency of the manganese oxidant. The protocol involves dissolving the specific beta-nitroalkene substrate and three equivalents of manganese(III) acetylacetonate in anhydrous ethanol, followed by heating the mixture to 55°C under vigorous stirring. Reaction progress is monitored via thin-layer chromatography, typically reaching completion within two hours for standard substrates, after which the mixture is subjected to flash column chromatography using a petroleum ether and acetone eluent system to isolate the pure crystalline product. For detailed standard operating procedures and safety guidelines regarding the handling of manganese salts and nitro compounds, please refer to the standardized synthesis steps outlined below.
- Dissolve the selected beta-nitroalkene substrate and manganese(III) acetylacetonate in anhydrous ethanol with a molar ratio favoring the oxidant.
- Heat the reaction mixture to a controlled temperature range of 30-70°C, optimally 55°C, and maintain stirring for 1 to 4 hours.
- Monitor reaction completion via TLC, then perform flash column chromatography using a petroleum ether and acetone system to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this manganese-catalyzed route offers substantial strategic benefits regarding cost structure and logistical reliability. The shift from a two-step batch process to a telescoped one-pot reaction inherently reduces the number of unit operations, which directly correlates to lower labor costs, reduced equipment wear, and minimized solvent consumption. By eliminating the need for specialized low-temperature cooling infrastructure and shortening the reaction cycle from days to hours, manufacturing facilities can achieve significantly higher throughput rates without requiring additional capital investment in reactor capacity.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous solvents with commodity-grade ethanol results in drastic savings on raw material procurement and waste disposal fees. Furthermore, the high atom economy of the oxidative cyclization ensures that a greater proportion of the input mass is converted into valuable product, minimizing the cost per kilogram of the final API intermediate. The avoidance of complex workup procedures associated with base-catalyzed reactions also reduces the consumption of auxiliary chemicals and water, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The broad substrate scope of this method means that manufacturers are not dependent on a single, hard-to-source starting material like beta,beta-dinitrostyrene. Instead, they can utilize a wide variety of commercially available beta-nitrostyrenes and heterocyclic vinyls, diversifying the supply base and mitigating the risk of shortages. The robustness of the reaction conditions allows for consistent quality output across different batches, ensuring that downstream customers receive materials with uniform specifications, which is critical for maintaining regulatory compliance in pharmaceutical supply chains.
- Scalability and Environmental Compliance: Operating at moderate temperatures in a green solvent aligns perfectly with modern environmental, health, and safety (EHS) standards, facilitating easier permitting and reducing the carbon footprint of the manufacturing site. The simplicity of the process makes it highly amenable to scale-up from laboratory grams to multi-ton commercial production, as the heat transfer and mixing requirements are far less demanding than those of the cryogenic conditions required by legacy methods. This scalability ensures that supply can be rapidly ramped up to meet market demand without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis platform. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the operational parameters and scope limitations for potential licensees and manufacturing partners.
Q: What are the primary advantages of using ethanol over traditional solvents in this synthesis?
A: Ethanol serves as a green, non-toxic reaction medium that significantly simplifies post-processing and waste treatment compared to chlorinated solvents or methanol, while maintaining high reaction efficiency and selectivity at moderate temperatures.
Q: How does the substrate scope of this Mn(acac)3 method compare to previous techniques?
A: Unlike prior art restricted to specific dinitrostyrenes, this method accommodates a broad range of electron-deficient nitroalkenes, including substituted beta-nitrostyrenes and heterocyclic variants like furan, thiophene, and pyridine derivatives.
Q: Does this process eliminate the generation of hazardous byproducts found in older methods?
A: Yes, the oxidative cyclization pathway avoids the formation of nitrous acid, a harmful byproduct associated with the traditional two-step Michael addition and cyclization sequences, thereby enhancing environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro-4,5-Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly evaluated the manganese-mediated oxidative cyclization described in CN101343263A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity 5-nitro-4,5-dihydrofuran derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities to ensure every batch exceeds client expectations.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced technology for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to obtain specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can drive value and efficiency in your supply chain.
