Revolutionizing 5-Nitro-4,5-Dihydrofuran Synthesis via Manganese Catalysis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational efficiency. Patent CN101343263A introduces a transformative methodology for the synthesis of 5-nitro-4,5-dihydrofuran derivatives, a critical structural motif found in numerous bioactive molecules and complex natural products. This innovation leverages manganese(III) acetylacetonate as a versatile oxidant and catalyst, enabling the direct cyclization of diverse electron-deficient nitroalkenes in an ethanol medium. Unlike traditional approaches that often suffer from harsh conditions and poor atom economy, this protocol operates at moderate temperatures between 30-70°C, drastically reducing energy consumption. For R&D directors and process chemists, this represents a significant leap forward in accessing functionalized heterocyclic scaffolds with improved selectivity and yield profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 5-nitro-4,5-dihydrofuran derivatives was largely constrained by the methodology reported by E.V. Trukhin, which relied on a multi-step sequence involving Michael addition followed by cyclization. This legacy process demanded stringent cryogenic conditions, initially requiring temperatures as low as -3 to -5°C for the addition step, followed by a slow warm-up phase. Furthermore, the cyclization step utilized triethylamine in anhydrous methanol over a protracted period of up to two days to achieve conversion. A critical drawback of this conventional route is the generation of nitrous acid as a hazardous byproduct, posing significant environmental and safety challenges for large-scale manufacturing. The overall yield of approximately 56% further exacerbates cost inefficiencies, making it less attractive for commercial production of high-purity pharmaceutical intermediates.
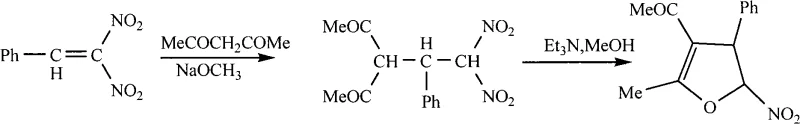
The Novel Approach
The patented technology fundamentally reimagines this transformation by employing a single-step oxidative cyclization driven by manganese(III) acetylacetonate. This novel approach eliminates the need for cryogenic cooling and reduces the reaction timeline from days to merely 1-4 hours. By utilizing absolute ethanol as the reaction medium, the process not only adheres to green chemistry principles but also simplifies the workup procedure, as ethanol is easily removed and recovered. The reaction tolerates a wide array of substituents on the aromatic or heteroaromatic rings, including electron-withdrawing and electron-donating groups, demonstrating exceptional versatility. This streamlined workflow directly addresses the bottlenecks of the prior art, offering a scalable solution for the cost reduction in pharmaceutical intermediate manufacturing.
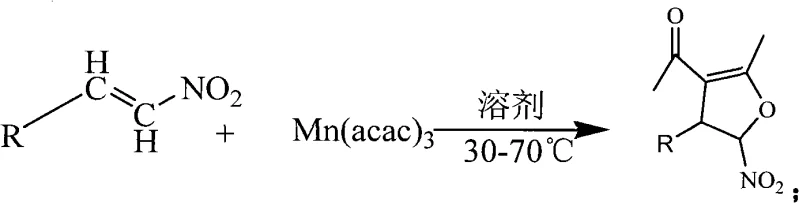
Mechanistic Insights into Mn(acac)3-Catalyzed Oxidative Cyclization
The core of this synthetic breakthrough lies in the unique reactivity of manganese(III) acetylacetonate, which acts as a single-electron oxidant to initiate radical pathways or coordinate with the electron-deficient double bond of the nitroalkene. The nitro group renders the vinyl moiety highly electrophilic, facilitating nucleophilic attack or radical addition depending on the specific mechanistic nuance employed by the metal center. In this system, the manganese species likely promotes the formation of a carbon-centered radical or cationic intermediate that undergoes intramolecular cyclization with the oxygen source or internal nucleophile to close the furan ring. This mechanism ensures high regioselectivity, preventing the formation of unwanted isomers that often plague non-catalyzed thermal cyclizations. The stability of the Mn(III) complex in ethanol allows for a controlled release of oxidative potential, minimizing side reactions such as polymerization or over-oxidation of the sensitive nitro functionality.
From an impurity control perspective, the choice of ethanol as a solvent plays a pivotal role in suppressing the formation of polar byproducts that are difficult to separate. The reaction conditions are sufficiently mild to preserve sensitive functional groups like halides and methoxy groups on the phenyl ring, which might otherwise degrade under the strongly basic or acidic conditions of older methods. The high yields observed, ranging from 74% to 94% across various substrates, indicate a clean reaction profile with minimal degradation. For quality assurance teams, this translates to a simpler purification process, often achievable through standard flash column chromatography, ensuring the final API intermediate meets stringent purity specifications required for downstream drug synthesis.
How to Synthesize 5-Nitro-4,5-Dihydrofuran Derivatives Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the efficiency of the manganese catalyst. The standard protocol involves dissolving the specific beta-nitroalkene substrate and manganese(III) acetylacetonate in absolute ethanol, typically maintaining a molar ratio of 1:3 to ensure complete conversion. The mixture is then heated to an optimal temperature of 55°C, where the reaction proceeds rapidly as monitored by thin-layer chromatography. Detailed standardized synthesis steps see the guide below.
- Dissolve the selected electron-deficient nitroalkene substrate and manganese(III) acetylacetonate in absolute ethanol solvent.
- Heat the reaction mixture to a temperature range of 30-70°C, preferably around 55°C, and maintain for 1-4 hours.
- Monitor reaction completion via TLC, then isolate the target dihydrofuran product using flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this manganese-catalyzed protocol offers tangible benefits beyond mere chemical elegance. The elimination of cryogenic equipment and the reduction of reaction time from 48 hours to under 4 hours significantly increases throughput capacity without requiring additional reactor volume. This efficiency gain directly contributes to substantial cost savings in manufacturing operations by lowering utility costs and labor hours per batch. Furthermore, the use of ethanol, a commodity solvent with a stable supply chain, mitigates the risks associated with sourcing specialized or regulated solvents, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive, toxic solvents and the removal of energy-intensive cooling steps drastically lower the operational expenditure per kilogram of product. By avoiding the generation of hazardous nitrous acid waste, the facility also saves on waste disposal fees and environmental compliance costs, creating a leaner and more profitable production model. The high isolated yields mean less raw material is wasted, optimizing the cost of goods sold for these valuable heterocyclic building blocks.
- Enhanced Supply Chain Reliability: The substrates required for this reaction, such as substituted beta-nitrostyrenes and heteroaryl nitroalkenes, are commercially available and inexpensive, ensuring a steady flow of starting materials. The robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment failure or sensitivity to ambient conditions. This reliability is crucial for maintaining continuous supply lines to downstream pharmaceutical clients who depend on just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of exothermic hazards associated with cryogenic additions. The use of ethanol aligns with modern sustainability goals, reducing the carbon footprint of the manufacturing process and simplifying regulatory approvals in regions with strict environmental laws. This eco-friendly profile enhances the marketability of the final product to global partners who prioritize green chemistry in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners.
Q: What are the primary advantages of using Mn(acac)3 over traditional base-catalyzed methods?
A: The Mn(acac)3 catalyzed method operates under significantly milder conditions (30-70°C) compared to the cryogenic requirements (-3 to -5°C) of prior art, while eliminating toxic nitrous acid byproducts and reducing reaction time from days to hours.
Q: Which substrates are compatible with this oxidative cyclization protocol?
A: The process demonstrates broad substrate scope, successfully converting various electron-deficient nitroalkenes including beta-nitrostyrenes, beta-nitrofuran vinyls, beta-nitrothiophene vinyls, and beta-nitropyrrole vinyls into the corresponding dihydrofuran derivatives.
Q: How does this method impact environmental compliance and waste management?
A: By utilizing ethanol as a green solvent instead of toxic organic solvents and avoiding the generation of hazardous nitrous acid waste, this method significantly simplifies post-processing and aligns with stricter environmental regulations for chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Nitro-4,5-Dihydrofuran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic routes like the one described in CN101343263A for producing high-value heterocyclic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this manganese-catalyzed method are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-nitro-4,5-dihydrofuran derivatives meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that your downstream synthesis remains uninterrupted and compliant with global regulatory frameworks.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can support your supply chain goals effectively.
