Scalable Synthesis of Donepezil Key Intermediate via Optimized Aldol Condensation
Scalable Synthesis of Donepezil Key Intermediate via Optimized Aldol Condensation
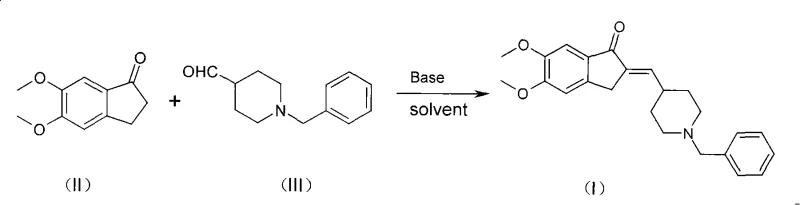
The global demand for effective treatments for Alzheimer's disease continues to drive innovation in the pharmaceutical intermediate sector, specifically for acetylcholinesterase inhibitors like Donepezil Hydrochloride. A pivotal advancement in this field is detailed in patent CN101628889A, which discloses a highly efficient method for preparing the critical key intermediate, 2-(1-benzyl-piperidin-4-yl methyl alkenyl)-5,6-dimethoxy-indanone. This compound serves as the direct precursor to the final active pharmaceutical ingredient (API), making its synthesis quality paramount for downstream drug efficacy. The disclosed technology represents a significant leap forward by utilizing a streamlined aldol condensation reaction in a benzene aromatic hydrocarbon solvent, specifically toluene, in the presence of alkali metal hydroxides. This approach not only simplifies the operational workflow but also addresses long-standing issues regarding impurity profiles and reaction times that have plagued previous synthetic routes. For R&D directors and procurement specialists alike, understanding the nuances of this patent provides a clear pathway toward more robust and cost-effective supply chains for neurological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this crucial indanone derivative has been fraught with chemical challenges that directly impact manufacturing economics and product quality. Prior art methods, such as those described in earlier literature and patents like WO2007108011, often relied on protic solvents like methanol or ethanol in the presence of sodium hydroxide. While conceptually simple, these conditions frequently led to the formation of significant amounts of a specific reduction byproduct, identified structurally as Compound IV, which is an alcohol derivative resulting from an unintended Michael addition followed by reduction. As illustrated in the structural analysis of impurities, this side reaction could account for up to 20% of the crude product mixture, necessitating complex and yield-depleting purification steps to meet pharmaceutical grade standards. Furthermore, other existing methodologies attempted to mitigate these issues by employing phase-transfer catalysts in mixed solvent systems of water and organic solvents under nitrogen protection. While these methods improved purity slightly, they introduced new bottlenecks, including extended reaction times of 7 to 8 hours, the high cost of specialized catalysts, and cumbersome post-reaction workups involving large volumes of water and solvent removal under reduced pressure.
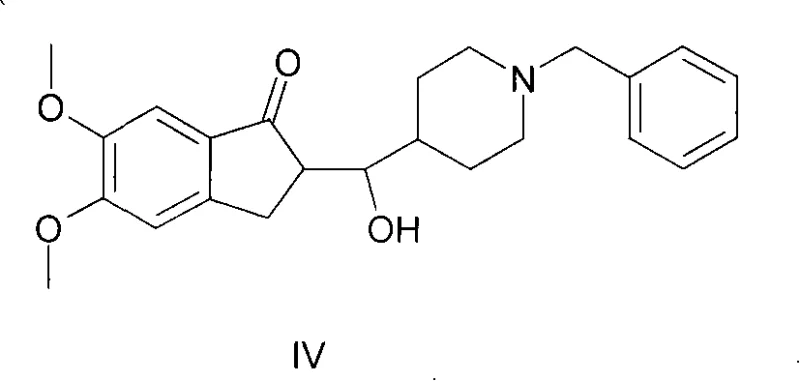
The Novel Approach
In stark contrast to these convoluted legacy processes, the method outlined in CN101628889A introduces a paradigm shift by utilizing a non-polar aromatic hydrocarbon solvent, preferably toluene, which fundamentally alters the reaction environment to favor the desired condensation product. By switching from a protic solvent like methanol to an aprotic solvent like toluene, the reaction effectively suppresses the mechanism leading to the formation of the alcohol impurity (Compound IV), thereby ensuring a much cleaner reaction profile from the outset. The process involves reacting 5,6-dimethoxy-1-indanone with 1-benzyl-4-piperidine carboxaldehyde in the presence of a simple alkali metal hydroxide, such as sodium hydroxide or potassium hydroxide, at moderate temperatures ranging from 50 to 80°C. This optimization results in a dramatic reduction in reaction time, often completing within 0.5 to 3 hours, compared to the half-day cycles required by older techniques. The simplicity of the workup is equally impressive; the product often precipitates directly upon cooling, allowing for simple filtration and a quick methanol wash to remove residual starting materials, yielding a high-purity solid without the need for column chromatography or extensive solvent exchanges.
Mechanistic Insights into Base-Catalyzed Aldol Condensation
The core of this synthetic breakthrough lies in the precise control of the aldol condensation mechanism within a non-polar medium. In this reaction, the alkali metal hydroxide acts as a base to abstract an acidic alpha-proton from the 5,6-dimethoxy-1-indanone, generating a resonance-stabilized enolate ion. This nucleophilic enolate then attacks the electrophilic carbonyl carbon of the 1-benzyl-4-piperidine carboxaldehyde. In protic solvents like methanol, the solvent molecules can participate in hydrogen bonding that stabilizes transition states leading to side reactions, or even act as hydride donors in specific conjugate reduction scenarios. However, in the toluene system described in the patent, the lack of proton donation capability prevents the reduction of the double bond in the intermediate aldol adduct. Instead, the reaction proceeds smoothly through the dehydration step to form the stable alpha,beta-unsaturated ketone (the enone system), which is the desired Compound I. The use of heterogeneous base conditions (solid NaOH/KOH in toluene) further enhances the selectivity, as the reaction likely occurs at the solid-liquid interface, limiting the exposure of the sensitive intermediates to bulk solution conditions that might promote degradation.
From an impurity control perspective, the mechanistic advantage is clear: the suppression of the Michael addition-reduction pathway is the primary driver for the high purity observed. In the conventional methanol route, the conjugated enone system formed initially is susceptible to nucleophilic attack by methoxide or hydride transfer, leading to the saturated alcohol impurity. By eliminating the protic solvent, the patent effectively removes the source of the hydride or the specific solvation environment required for this reduction. Additionally, the patent notes that while a small amount of isomer (less than 3%) may form, this isomer is chemically compatible with the subsequent hydrogenation step used to produce the final Donepezil API. This tolerance for minor isomeric impurities reduces the pressure on the intermediate synthesis to achieve absolute stereochemical perfection at this stage, allowing for a more forgiving and robust process that prioritizes overall yield and throughput over unnecessary intermediate purification.
How to Synthesize 2-(1-benzyl-piperidin-4-yl methyl alkenyl)-5,6-dimethoxy-indanone Efficiently
Implementing this synthesis on a production scale requires strict adherence to the optimized parameters regarding solvent volume, reactant ratios, and thermal control to maximize the benefits of this novel route. The process is designed to be operationally simple, avoiding the need for inert gas blankets or anhydrous conditions that typically drive up manufacturing costs. The key to success lies in the careful selection of the base and the maintenance of the reaction temperature within the optimal window of 60 to 80°C, which balances reaction rate with selectivity. Operators should note that the molar ratio of the aldehyde to the indanone is slightly skewed, typically between 1:1.0 to 1:1.2, to ensure complete consumption of the more valuable or critical indanone starting material. The following guide outlines the standardized procedure derived from the patent embodiments, serving as a foundational protocol for process engineers looking to adopt this technology.
- Charge a reaction vessel with toluene, 5,6-dimethoxy-1-indanone, and sodium hydroxide under magnetic agitation.
- Add 1-benzyl-4-piperidine carboxaldehyde and heat the mixture slowly to 60-70°C for 0.5 to 1.5 hours.
- Cool the reaction to 20°C, filter the solid precipitate, wash with methanol, and vacuum dry to obtain the pure intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this toluene-based synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of phase-transfer catalysts and the shift to a single-solvent system drastically simplify the raw material sourcing strategy, reducing the number of SKUs that need to be managed and qualified. Furthermore, the significant reduction in reaction time from nearly a full day to merely a few hours translates directly into increased asset utilization, allowing manufacturing facilities to produce more batches per month without capital expenditure on new reactors. The simplified workup, which relies on filtration rather than complex extraction and distillation sequences, also implies a substantial reduction in energy consumption and solvent waste, aligning with modern green chemistry initiatives and lowering the overall environmental compliance burden.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of expensive auxiliary chemicals and the optimization of solvent usage. By avoiding phase-transfer catalysts and utilizing common, low-cost solvents like toluene which can be easily recovered and recycled, the variable cost per kilogram of the intermediate is significantly lowered. Additionally, the high yield achieved (often exceeding 95%) means that less raw material is wasted, directly improving the material efficiency of the process. The avoidance of complex purification steps such as column chromatography or multiple recrystallizations further reduces labor costs and consumable expenses, making this a highly cost-competitive route for large-scale API manufacturing.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures greater continuity of supply. The reagents involved, including 5,6-dimethoxy-1-indanone and 1-benzyl-4-piperidine carboxaldehyde, are commercially available commodity chemicals, reducing the risk of supply bottlenecks associated with exotic or custom-synthesized reagents. The process tolerance to minor variations in conditions, combined with the ability to handle isomeric impurities in downstream steps, creates a buffer against batch-to-batch variability. This reliability is crucial for maintaining the steady flow of intermediates required for Just-In-Time manufacturing models prevalent in the pharmaceutical industry, minimizing the risk of production stoppages due to quality failures.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its reliance on standard unit operations such as heating, stirring, and filtration, which are easily transferred from laboratory to pilot to commercial scale. The use of toluene, while requiring appropriate safety measures, is well-understood in industrial settings, and the reduced solvent volume (5 to 30 times the mass of reactants) minimizes the physical footprint of waste storage and treatment facilities. The absence of heavy metal catalysts or persistent organic pollutants simplifies the effluent treatment process, ensuring that the manufacturing site remains compliant with increasingly stringent environmental regulations regarding wastewater discharge and volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on why this specific route is superior to traditional methods. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on process efficiency.
Q: Why is toluene preferred over methanol for this condensation reaction?
A: Toluene is preferred because it is an aprotic solvent that prevents the unwanted Michael addition reduction side reaction often observed in protic solvents like methanol, thereby significantly reducing impurity formation (specifically Compound IV) and simplifying purification.
Q: What is the typical yield and purity achievable with this method?
A: According to the patent data, this method consistently achieves yields exceeding 95% with HPLC purity levels above 95%, significantly outperforming prior art methods which often struggle with impurity levels around 20%.
Q: Does this process require expensive phase-transfer catalysts?
A: No, this novel approach eliminates the need for phase-transfer catalysts or complex mixed solvent systems, relying instead on simple alkali metal hydroxides in a single organic solvent, which drastically reduces raw material costs and waste treatment complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Donepezil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex neurological agents like Donepezil depends on the availability of high-quality, cost-effective intermediates produced via robust synthetic routes. Our technical team has extensively analyzed the methodology described in CN101628889A and possesses the expertise to implement this optimized toluene-based condensation process at any scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2-(1-benzyl-piperidin-4-yl methyl alkenyl)-5,6-dimethoxy-indanone meets the exacting standards required for GMP API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this high-yield process for your specific volume requirements. We encourage you to contact us today to request specific COA data from our recent pilot batches and to receive detailed route feasibility assessments tailored to your project timelines. Let us help you secure a reliable, high-purity pharmaceutical intermediate supply that drives down costs and accelerates your time to market.
