Advanced Synthesis of Donepezil Hydrochloride Key Intermediate via Optimized Aldol Condensation
Advanced Synthesis of Donepezil Hydrochloride Key Intermediate via Optimized Aldol Condensation
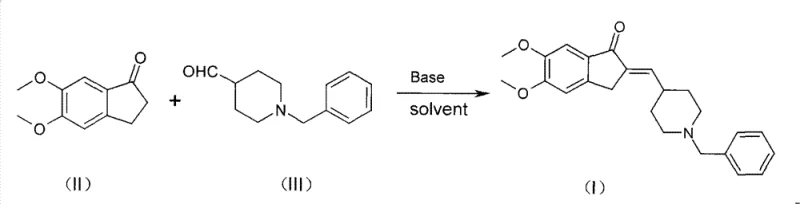
The pharmaceutical landscape for neurological disorders continues to evolve, with Donepezil Hydrochloride (E2020) remaining a cornerstone therapy for Alzheimer's dementia due to its high selectivity and favorable safety profile. Central to the efficient manufacturing of this critical active pharmaceutical ingredient is the robust production of its key precursor, 2-(1-benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indanone. Patent CN101628889B discloses a transformative method for preparing this intermediate through an optimized aldol condensation reaction. This technical insight report analyzes the strategic advantages of this novel synthetic route, which utilizes C6-C12 aromatic hydrocarbon solvents like toluene in the presence of alkali metal hydroxides. By shifting away from traditional alcoholic solvents and complex phase-transfer systems, this methodology offers a pathway to significantly higher purity and yield, addressing critical pain points for reliable pharmaceutical intermediate suppliers aiming to streamline their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pivotal indanone derivative has been plagued by significant chemical inefficiencies that hinder large-scale manufacturing. Prior art, such as methodologies disclosed in earlier literature and patents like WO2007108011, typically employed methanol or ethanol as the reaction solvent in the presence of sodium hydroxide. While conceptually straightforward, these alcoholic conditions promote the formation of stubborn by-products, specifically a structural isomer identified as Compound IV. Data indicates that under these conventional methanol-based conditions, the generation of this specific impurity can reach levels as high as 20%, creating a severe burden on downstream purification processes. Furthermore, alternative approaches described in patents like WO2007119118 attempted to mitigate these issues by introducing phase-transfer catalysts and mixed solvent systems involving water and organic phases. However, these modifications introduced new operational complexities, including the absolute necessity for nitrogen protection, extended reaction times ranging from 7 to 8 hours, and cumbersome work-up procedures involving large volumes of water and subsequent solvent removal. These factors collectively inflate production costs and compromise the overall throughput of the manufacturing line.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally re-engineers the reaction environment to favor the desired thermodynamic product while suppressing side reactions. By replacing polar protic solvents like methanol with non-polar aromatic hydrocarbons such as toluene, xylene, or parachlorotoluene, the reaction kinetics are altered to minimize impurity formation. This novel approach operates under mild basic conditions using readily available alkali metal hydroxides like sodium hydroxide or potassium hydroxide, eliminating the need for expensive phase-transfer catalysts or sensitive alkoxides. The process is remarkably efficient, with reaction times drastically reduced to a window of 0.5 to 3 hours at temperatures between 50°C and 80°C. The simplicity of the work-up procedure is another standout feature; the product often precipitates directly from the reaction mixture upon cooling, allowing for simple filtration and a brief methanol wash to achieve high purity. This shift represents a paradigm change in cost reduction in API manufacturing, moving from complex, multi-step aqueous work-ups to a streamlined crystallization-driven isolation process.
Mechanistic Insights into Base-Catalyzed Aldol Condensation
The core of this synthetic advancement lies in the nuanced understanding of solvent effects on the aldol condensation mechanism between 5,6-dimethoxy-1-indanone and 1-benzyl-4-piperidinecarboxaldehyde. In the presence of a strong base like sodium hydroxide, the alpha-protons of the indanone are deprotonated to form a resonance-stabilized enolate ion. In traditional methanol solvents, the high polarity and hydrogen-bonding capability of the solvent can stabilize transition states that lead to unwanted side reactions, potentially facilitating nucleophilic attacks at unintended positions or promoting retro-aldol pathways that result in the accumulation of Compound IV. The structure of this problematic impurity, which complicates purification, is distinct from the target molecule and requires rigorous chromatographic separation if formed in significant quantities. By utilizing toluene, a solvent with lower dielectric constant and different solvation properties, the reaction environment favors the formation of the thermodynamically stable conjugated enone system of the target intermediate. This solvent choice likely reduces the solubility of the product as it forms, driving the equilibrium forward through precipitation and physically removing the product from the reactive medium, thereby preventing further degradation or isomerization.
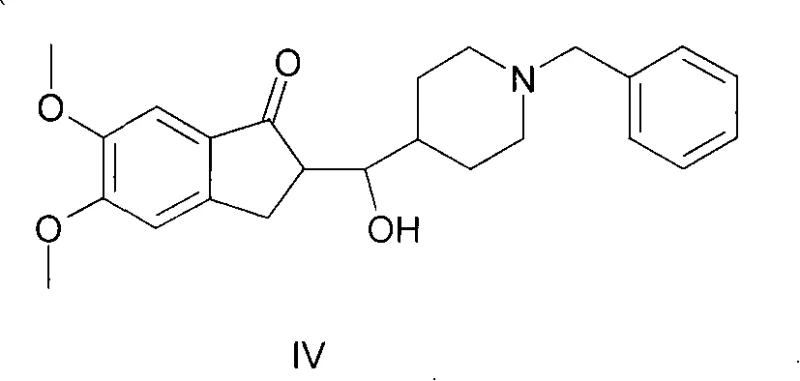
Furthermore, the control of impurities is critical for meeting the stringent regulatory standards required for high-purity pharmaceutical intermediates. The patent data highlights that while a minor isomer (less than 3%) may still form in the toluene system, this specific isomer is benign as it can be converted into the final Donepezil API during the subsequent hydrogenation step. This is a crucial mechanistic insight for process chemists, as it implies that absolute stereochemical or regiochemical perfection at this specific stage is not required, provided the major impurity (Compound IV) is suppressed. The use of aromatic solvents effectively suppresses the formation of Compound IV to negligible levels (often below 1% as seen in the experimental data), whereas methanol systems leave nearly 10% of this difficult-to-remove contaminant. This mechanistic advantage translates directly into a cleaner crude product, reducing the load on purification units and ensuring a more consistent quality profile for the downstream synthesis of the final neuroprotective agent.
How to Synthesize 2-(1-benzyl-piperidin-4-ylmethyl)-5,6-dimethoxy-indanone Efficiently
Implementing this optimized synthesis protocol requires precise control over reaction parameters to maximize the benefits of the toluene solvent system. The process begins with the careful charging of the aromatic solvent and the base, followed by the controlled addition of the aldehyde to manage exotherms and ensure uniform mixing. The reaction temperature is maintained within a specific range to balance reaction rate with selectivity, avoiding the degradation that can occur at excessive temperatures while ensuring complete conversion within a short timeframe. Upon completion, the cooling phase is critical for inducing crystallization, which serves as the primary purification step. For a detailed breakdown of the specific operational parameters, stoichiometry, and work-up instructions derived from the patent examples, please refer to the standardized synthesis guide below.
- Charge a reaction vessel with toluene solvent, 5,6-dimethoxy-1-indanone, and an alkali metal hydroxide base such as sodium hydroxide or potassium hydroxide.
- Add 1-benzyl-4-piperidinecarboxaldehyde under magnetic agitation and heat the mixture to a temperature range of 50-80°C for 0.5 to 3 hours.
- Cool the reaction mixture to ambient temperature, filter the precipitated solid, wash with methanol, and vacuum dry to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of specialized reagents such as phase-transfer catalysts and the replacement of mixed solvent systems with a single, commodity-grade aromatic hydrocarbon significantly simplifies the raw material sourcing strategy. Toluene is a widely available industrial solvent with a stable supply chain, reducing the risk of procurement bottlenecks that can occur with specialty chemicals. Moreover, the drastic reduction in reaction time from nearly 8 hours in prior art methods to under 3 hours in this new process dramatically increases reactor turnover rates. This efficiency gain allows manufacturing facilities to produce larger volumes of the intermediate using existing infrastructure, effectively expanding capacity without the need for capital-intensive equipment upgrades. The simplified work-up, which avoids large-scale aqueous extractions and complex pH adjustments, also translates to reduced utility consumption and lower wastewater treatment costs, aligning with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the material bill and the reduction of processing time. By removing the requirement for phase-transfer catalysts and avoiding the use of expensive or hazardous reagents like concentrated hydrochloric acid for pH adjustment found in other methods, the direct material costs are lowered. Additionally, the high yield achieved (consistently above 95% in optimized examples) means that less starting material is wasted, maximizing the return on investment for every kilogram of 5,6-dimethoxy-1-indanone purchased. The ability to isolate the product via simple filtration rather than energy-intensive distillation or chromatography further drives down the operational expenditure per unit of production.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for maintaining continuous API production schedules. This method enhances reliability by utilizing robust, non-sensitive reaction conditions that do not require strict nitrogen protection or anhydrous environments, unlike some alternative routes. The use of common solvents like toluene and bases like sodium hydroxide ensures that raw materials can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. The shortened cycle time also provides greater flexibility in production planning, allowing manufacturers to respond more agilely to fluctuations in market demand for Donepezil without maintaining excessive inventory buffers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this solvent-based system is inherently scalable due to its homogeneous nature and manageable exotherm profile. The reduction in solvent diversity (using primarily toluene and a small amount of methanol for washing) simplifies solvent recovery and recycling loops, which is a key factor in green chemistry metrics. By minimizing the generation of aqueous waste streams associated with phase-transfer catalysis and extensive washing, the environmental footprint of the manufacturing process is significantly reduced. This alignment with sustainability goals not only lowers disposal costs but also future-proofs the supply chain against increasingly stringent environmental regulations in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology compares to established industry practices. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is toluene preferred over methanol for this condensation reaction?
A: Toluene significantly reduces the formation of difficult-to-remove impurities, specifically Compound IV, which is generated in substantial amounts (around 20%) when methanol is used as the solvent. Additionally, toluene allows for simpler product isolation via filtration.
Q: What are the typical yield and purity improvements with this method?
A: The patented method achieves yields exceeding 95% with high HPLC purity (often >96%), whereas conventional methanol-based methods often result in lower purity due to significant impurity generation requiring complex purification.
Q: Does this process require expensive phase-transfer catalysts?
A: No, this optimized route eliminates the need for phase-transfer catalysts and mixed solvent systems, relying instead on simple alkali metal hydroxides in a single aromatic hydrocarbon solvent, which simplifies the supply chain and reduces material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Donepezil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your final API production is only as strong as the quality of your starting materials. Our technical team has extensively analyzed advanced synthetic routes like the one described in CN101628889B to ensure we offer the most competitive and robust intermediates in the market. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you are in the clinical trial phase or full-scale commercialization, our supply capabilities match your needs. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Donepezil intermediate meets the high standards required for neurological therapeutics.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term production goals for Alzheimer's treatments.
