Advanced Metal-Free Synthesis of 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective pathways to synthesize complex heterocyclic scaffolds, particularly those containing nitrogen-rich motifs like 1,2,4-triazoles. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a novel preparation method for 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot oxidative cyclization strategy. By leveraging a metal-free catalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), the process achieves high conversion rates under relatively mild thermal conditions (80-100°C). For R&D directors and procurement managers alike, this patent offers a compelling value proposition: the ability to access high-value bioactive intermediates without the burden of heavy metal contamination or the logistical nightmare of stringent inert atmosphere operations. The versatility of this method allows for the introduction of various functional groups, making it a powerful tool for medicinal chemistry campaigns aiming to optimize drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been fraught with inefficiencies that hinder large-scale commercial adoption. Traditional routes often rely on quinoline-2-carboxylic acid as a primary starting material, necessitating a cumbersome five-step reaction sequence to arrive at the target molecule. This multi-step approach not only results in a dismal total yield of approximately 17%, representing a massive loss of raw material value, but also requires severe reaction conditions that demand specialized equipment and rigorous safety protocols. Furthermore, conventional methodologies frequently employ transition metal catalysts, which introduce significant downstream processing challenges. The removal of trace heavy metals to meet pharmaceutical purity standards often requires additional purification steps, such as scavenging or recrystallization, which further erodes the overall yield and increases the cost of goods sold (COGS). These factors combined make traditional synthesis routes economically unviable for the mass production of these critical intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN113307790B offers a disruptive alternative that addresses these pain points directly. By utilizing cheap and readily available starting materials—specifically 2-methylquinoline and trifluoroethylimide hydrazide—the new process collapses the synthesis into a single pot. The reaction is promoted by an organic catalytic system rather than precious metals, fundamentally altering the economic landscape of production. The absence of strict anhydrous or oxygen-free requirements means that standard reactor vessels can be used, drastically reducing capital expenditure and operational complexity. This approach not only simplifies the workflow but also enhances the safety profile of the manufacturing process. The ability to tolerate a wide range of substituents on both the quinoline and the triazole rings ensures that this method is not just a niche solution but a versatile platform technology capable of generating diverse libraries of compounds for drug discovery and development.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core innovation of this technology lies in its elegant mechanistic pathway, which avoids the pitfalls of organometallic chemistry while maintaining high efficiency. The reaction initiates with the oxidative activation of the methyl group on the 2-methylquinoline substrate. In the presence of tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), the methyl group is oxidized in situ to generate a reactive 2-quinoline carbaldehyde intermediate. This transformation is critical as it bypasses the need to isolate unstable aldehyde intermediates, which are often prone to degradation. Subsequently, this generated aldehyde undergoes a condensation reaction with the trifluoroethylimide hydrazide to form a dehydrated hydrazone species. This intermediate then enters a cascade of oxidative iodination and intramolecular electrophilic substitution reactions.
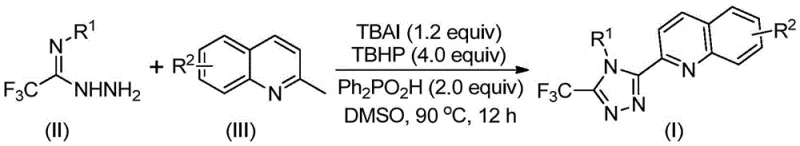
The final aromatization step yields the stable 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole core. The inclusion of diphenylphosphoric acid as an additive plays a pivotal role in facilitating these proton transfer steps and stabilizing the transition states, thereby driving the equilibrium towards the desired product. From a purity perspective, this mechanism is advantageous because it minimizes the formation of metal-complexed byproducts. The primary impurities are likely to be organic in nature, which are generally easier to separate via standard chromatographic techniques or crystallization compared to metal chelates. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the reaction trajectory is well-defined and controllable through the modulation of oxidant equivalents and temperature.
How to Synthesize 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires precise attention to reagent stoichiometry and thermal management to maximize yield. The patent outlines a robust protocol where the molar ratio of the catalyst system to substrates is optimized to balance reaction rate and cost. Typically, the process involves dissolving the hydrazide and quinoline derivatives in a polar aprotic solvent like DMSO, which effectively solubilizes the ionic intermediates. The addition of the oxidant must be controlled to prevent exothermic runaway, although the reaction is generally manageable at temperatures between 80°C and 100°C. For detailed operational parameters and specific workup procedures that ensure GMP compliance, please refer to the standardized guide below.
- Combine tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroethylimide hydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete conversion via oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the high-purity 3-quinolinyl-5-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of heavy metal catalysts removes a significant bottleneck in the supply chain, specifically the dependency on volatile precious metal markets and the complex logistics associated with their handling and disposal. Moreover, the use of commodity chemicals as starting materials ensures a stable and resilient supply base, reducing the risk of production stoppages due to raw material shortages. The simplified operational conditions also translate to lower energy consumption and reduced waste generation, aligning with modern sustainability goals and regulatory requirements.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of expensive transition metal catalysts and the reduction of synthetic steps. By collapsing a five-step sequence into a single pot, manufacturers can significantly reduce labor costs, solvent usage, and reactor occupancy time. The avoidance of heavy metals also eliminates the costly downstream purification steps typically required to meet residual metal specifications, leading to substantial overall cost savings. Furthermore, the high atom economy and improved yields mean that less raw material is wasted, directly improving the margin profile of the final active pharmaceutical ingredient (API) or intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, non-specialized reagents. Unlike proprietary catalysts that may have single-source suppliers, components like TBAI and TBHP are bulk commodities with robust global supply networks. This diversification reduces the risk of supply disruptions. Additionally, the tolerance for ambient atmospheric conditions means that production does not require specialized inert gas infrastructure, allowing for greater flexibility in manufacturing site selection and faster turnaround times for batch production. This agility is crucial for responding to fluctuating market demands in the pharmaceutical sector.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this method is inherently designed for expansion. The absence of pyrophoric reagents or extreme pressure conditions makes the transition from gram-scale to ton-scale production smoother and safer. From an environmental standpoint, the reduction in hazardous waste—particularly heavy metal sludge—simplifies effluent treatment and lowers disposal costs. This aligns with increasingly stringent environmental regulations, ensuring long-term operational continuity without the threat of regulatory shutdowns. The process exemplifies green chemistry principles, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does this synthesis method require toxic heavy metal catalysts?
A: No, the patented process utilizes an organic catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), completely eliminating the need for expensive and toxic transition metal catalysts often found in traditional cross-coupling reactions.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: Unlike many sensitive organometallic reactions, this method does not require strict anhydrous or anaerobic conditions. It can be operated under ambient atmospheric conditions, significantly simplifying the equipment requirements and operational complexity for large-scale production.
Q: Is this method suitable for producing diverse derivatives?
A: Yes, the method demonstrates excellent substrate tolerance. By varying the substituents on the aryl group of the hydrazide or the quinoline ring (such as methyl, methoxy, halogen, or nitro groups), a wide library of diversified 1,2,4-triazole compounds can be synthesized with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolinyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
The technological advancements presented in patent CN113307790B highlight the immense potential of metal-free oxidative cyclization in modern pharmaceutical manufacturing. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative academic and patent literature into reliable commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 1,2,4-triazole intermediate delivered meets the highest global standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective synthesis route for their drug development programs. By partnering with our technical team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring your supply chain is optimized for both performance and profitability.
