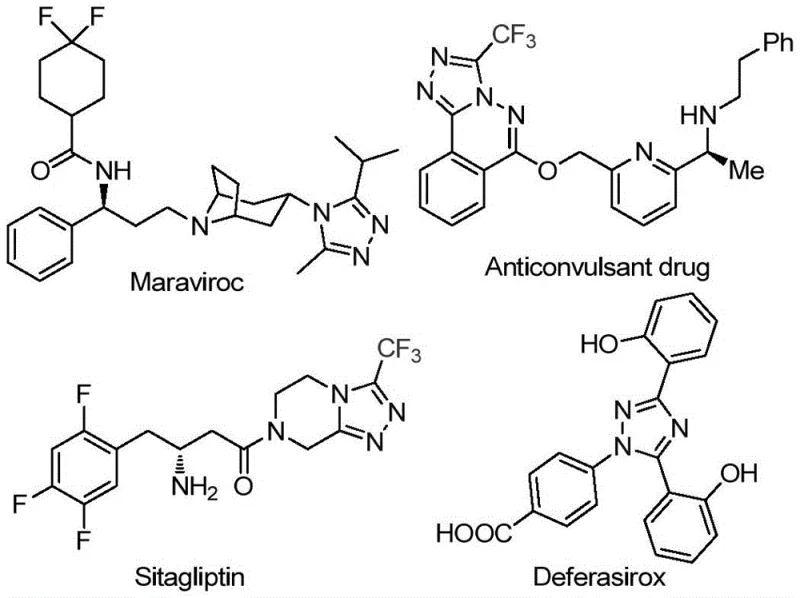
Advanced Iodine-Catalyzed Synthesis of 3,4,5-Trisubstituted Triazoles for Commercial Pharmaceutical Manufacturing
Patent CN113105402B introduces a groundbreaking synthetic methodology for producing 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses critical limitations in current pharmaceutical intermediate manufacturing. This innovative approach eliminates the need for transition metal catalysts while maintaining high structural complexity required for advanced drug molecules. The process leverages iodine-mediated chemistry to construct the triazole core under practical reaction conditions that significantly enhance operational feasibility for industrial implementation. By avoiding stringent anhydrous and oxygen-free requirements typically associated with heterocyclic synthesis, this method substantially reduces process complexity while maintaining excellent functional group tolerance across diverse substrate combinations. The patent demonstrates particular value for producing triazole-containing intermediates essential to modern pharmaceutical development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for polysubstituted triazoles frequently rely on transition metal-catalyzed cyclization reactions that introduce significant challenges for pharmaceutical manufacturing. These methods typically require strictly controlled anhydrous and oxygen-free environments that substantially increase operational complexity and equipment costs while limiting scalability potential. The necessity for expensive palladium or copper catalysts creates additional purification hurdles to remove trace metal residues that could compromise final drug product quality. Furthermore, conventional approaches often exhibit narrow substrate scope with poor functional group tolerance, particularly when incorporating both trifluoromethyl and acyl groups simultaneously into the triazole scaffold. These limitations collectively result in higher production costs and extended development timelines that hinder rapid commercialization of novel therapeutic agents requiring triazole-containing intermediates.
The Novel Approach
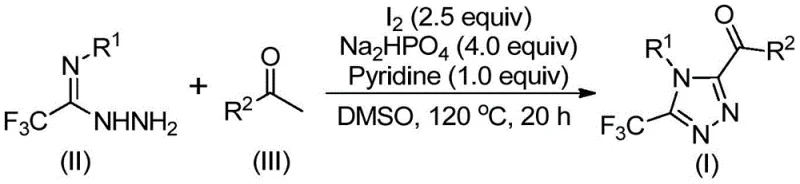
The patented methodology overcomes these constraints through an elegant iodine-promoted cascade reaction that operates under standard atmospheric conditions without requiring specialized equipment or handling procedures. By utilizing elemental iodine as a non-toxic promoter instead of transition metals, the process eliminates costly metal removal steps while maintaining excellent reaction efficiency across diverse substrate combinations. The two-stage reaction sequence begins with iodination/Kornblum oxidation of aryl ethyl ketones followed by condensation with trifluoroethylimide hydrazides to form the triazole core through intramolecular cyclization. This approach demonstrates remarkable functional group tolerance with various substituted aryl groups while producing high-purity intermediates suitable for direct incorporation into pharmaceutical manufacturing processes without extensive additional purification requirements.
Mechanistic Insights into Iodine-Promoted Triazole Formation
The reaction mechanism proceeds through a well-defined sequence where aryl ethyl ketones undergo iodination/Kornblum oxidation in dimethyl sulfoxide solvent to generate aryl diketone intermediates at elevated temperatures (90-110°C). This critical transformation occurs without requiring anhydrous conditions due to DMSO's dual role as both solvent and oxidizing agent. The resulting diketones then react with trifluoroethylimide hydrazides through dehydration and condensation to form hydrazone intermediates that subsequently undergo iodine-mediated intramolecular cyclization under mild basic conditions provided by sodium dihydrogen phosphate and pyridine. The precise molar ratio of reagents (trifluoroethylimide hydrazide:aryl ethyl ketone:sodium dihydrogen phosphate:pyridine:iodine = 1:2:4:1:2.5) ensures optimal reaction kinetics while minimizing side product formation throughout the cascade process.
Impurity control is achieved through the stepwise nature of the reaction mechanism where each transformation occurs under distinct temperature regimes (first stage at 90-110°C for 4-6 hours; second stage at 110-130°C for 12-20 hours) that prevent competing side reactions. The absence of transition metals eliminates potential metal-catalyzed decomposition pathways that commonly generate impurities in conventional syntheses. Post-reaction purification via standard column chromatography effectively removes any residual starting materials or minor byproducts while maintaining excellent yields across diverse substrate combinations. This controlled reaction profile ensures consistent production of high-purity triazole intermediates meeting the stringent quality requirements essential for pharmaceutical applications where impurity profiles directly impact drug safety and efficacy.
How to Synthesize Triazole Intermediates Efficiently
This innovative synthetic route provides pharmaceutical manufacturers with a practical pathway to produce complex triazole-containing intermediates without the limitations of traditional metal-catalyzed methods. The process demonstrates exceptional versatility across various substituted aryl groups while maintaining consistent product quality through its well-defined reaction mechanism. Detailed standardized synthesis procedures have been developed based on the patented methodology to ensure reproducible results across different production scales. The following step-by-step guide outlines the essential operational parameters required for successful implementation in industrial settings while maintaining the high purity standards demanded by pharmaceutical applications.
- Combine aryl ethyl ketone with iodine in DMSO solvent at 90-110°C for 4-6 hours to form aryl diketone intermediate through iodination/Kornblum oxidation
- Add trifluoroethylimide hydrazide with sodium dihydrogen phosphate and pyridine to the reaction mixture maintaining precise molar ratios of 1: 2:4:1:2.5
- Heat the mixture to 110-130°C for 12-20 hours to complete cyclization followed by standard column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic approach delivers substantial value to procurement and supply chain operations by addressing multiple pain points inherent in traditional triazole intermediate manufacturing. The elimination of expensive transition metal catalysts reduces raw material costs while simplifying quality control procedures through the absence of metal residue testing requirements. The use of commercially available starting materials from established chemical suppliers enhances supply chain resilience by reducing dependency on specialized reagent sources that often create procurement bottlenecks in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The complete avoidance of transition metal catalysts removes multiple cost drivers including expensive catalyst procurement and complex post-reaction purification steps required to eliminate metal residues. The simplified process flow reduces equipment requirements and operational complexity while maintaining excellent yields across diverse substrate combinations. This streamlined approach significantly lowers overall production costs without compromising product quality or purity specifications required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilization of readily available starting materials from multiple global suppliers creates robust sourcing options that mitigate supply chain disruption risks. The process operates under standard atmospheric conditions without specialized equipment requirements enabling rapid technology transfer between manufacturing sites worldwide. This flexibility allows for strategic production allocation based on regional supply chain dynamics while maintaining consistent product quality through the well-defined reaction parameters documented in the patent.
- Scalability and Environmental Compliance: The demonstrated scalability from laboratory to gram-level production provides a clear pathway for industrial implementation with minimal process re-engineering requirements. The elimination of toxic heavy metals from the manufacturing process substantially reduces environmental impact while simplifying waste stream management. This environmentally friendly approach aligns with growing regulatory pressures for sustainable chemical manufacturing practices while maintaining excellent functional group tolerance across diverse substrate combinations required for complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions and answers address key technical and commercial considerations regarding implementation of this patented triazole synthesis methodology in pharmaceutical manufacturing operations. These insights are derived directly from the patent documentation and reflect practical experience with the technology's implementation across various production scales.
Q: How does this method eliminate heavy metal contamination concerns in pharmaceutical intermediate production?
A: The iodine-promoted synthesis completely avoids transition metal catalysts that typically require complex removal processes. This eliminates potential heavy metal residues in the final product which is critical for pharmaceutical applications requiring stringent purity specifications.
Q: What makes this process suitable for commercial scale-up of complex pharmaceutical intermediates?
A: The reaction operates under standard atmospheric conditions without anhydrous or oxygen-free requirements and uses readily available starting materials. The demonstrated scalability from laboratory to gram-level production provides a clear pathway for industrial implementation with minimal process re-engineering.
Q: How does the process address impurity profile challenges in triazole compound manufacturing?
A: The mechanism involves controlled sequential reactions where aryl diketone formation precedes hydrazone condensation and cyclization. This stepwise approach minimizes side reactions while the column chromatography purification ensures removal of potential impurities to meet pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Intermediate Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications through rigorous QC labs. We have successfully implemented this patented iodine-promoted triazole synthesis technology across multiple client projects with consistent delivery of high-purity intermediates meeting exacting pharmaceutical quality standards. Our technical team has developed optimized protocols that ensure seamless technology transfer from laboratory to commercial manufacturing while maintaining all critical quality attributes required for regulatory compliance.
Leverage our expertise to accelerate your development timeline through a Customized Cost-Saving Analysis that identifies specific opportunities within your supply chain. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your unique manufacturing requirements and quality specifications.
