Revolutionizing Pharmaceutical Intermediate Production Through Advanced Nickel-Catalyzed Synthesis of α-Cyano Quaternary Carbon Center Compounds
Patent CN111499542A introduces a groundbreaking method for synthesizing cycloalkenone compounds containing α-cyano group substituted quaternary carbon centers, representing a significant advancement in organic synthesis methodology for pharmaceutical intermediates. This innovative approach addresses longstanding challenges in constructing these biologically important structures through a nickel-catalyzed tandem reaction that demonstrates exceptional regioselectivity and enantioselectivity under remarkably mild conditions. The technology enables efficient production of compounds featuring all-carbon quaternary carbon stereocenters with cyanide groups—structures that prevent α-carbon oxidation while maintaining metabolic stability and enhancing hydrogen bonding properties essential for pharmaceutical applications. Unlike previous methods that struggled with transition metal-cyanide coordination issues leading to decomposition, this process provides a reliable pathway to these valuable pharmacophores with excellent functional group tolerance and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing compounds with α-cyano quaternary carbon centers have faced significant challenges due to the inherent properties of cyanide groups and their interactions with transition metals. Previous methods primarily relied on α-cyanocarbonyl electrophilic functionalization or ketene imine electrophilic functionalization, both of which suffered from poor enantioselectivity and limited substrate scope. The coordination affinity between cyanide groups and transition metals created substantial complications, while the small steric size of cyanide groups further exacerbated selectivity issues during asymmetric transformations. Most critically, disubstituted malononitriles were prone to decyanation decomposition under transition metal catalysis conditions, severely limiting practical applications despite their availability and lower toxicity profile compared to alternative precursors. Existing protocols demonstrated only two documented examples of asymmetric desymmetrization of bissubstituted malononitriles—Tanaka's rhodium-catalyzed [2+2+2] cycloaddition achieving only 75% yield with 33% ee, and Ikariya's ruthenium-catalyzed hydrolysis yielding merely 54% with 30% ee—both exhibiting poor enantioselectivity and limited substrate applicability.
The Novel Approach
The patented method overcomes these limitations through an innovative nickel-catalyzed tandem reaction where aryl or alkenyl boron reagents react with alkynyl substituted disubstituted malononitrile compounds under mild conditions to form α-cyano substituted quaternary carbon center cyclic ketene compounds with high efficiency and selectivity. This approach leverages nickel complexes paired with specifically designed chiral ligands that prevent cyanide decomposition while enabling precise stereochemical control at the quaternary carbon center. The process operates effectively at moderate temperatures (60-120°C) with excellent functional group tolerance, producing high yields without requiring expensive precious metal catalysts or complex purification procedures to remove heavy metal residues. Crucially, the method utilizes readily available starting materials and demonstrates remarkable versatility across diverse substrate combinations, generating structurally complex products with high enantiomeric excess values that were previously unattainable through conventional approaches.
Mechanistic Insights into Nickel-Catalyzed Tandem Reaction
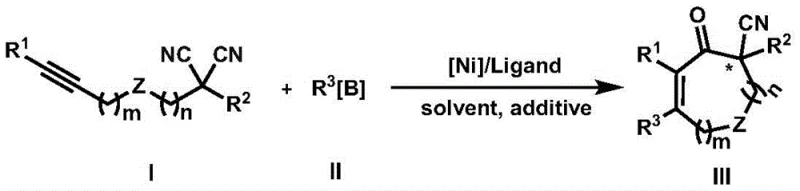
The nickel-catalyzed tandem reaction proceeds through a sophisticated mechanism involving alkyne insertion followed by cyclization steps that enable precise construction of the challenging quaternary carbon center bearing an α-cyano group. The nickel complex activates both reaction partners simultaneously—the alkyne moiety of compound I and the boron reagent II—facilitating oxidative addition followed by migratory insertion steps that form critical carbon-carbon bonds with high regiocontrol. The chiral ligand environment around the nickel center creates an asymmetric pocket that directs facial selectivity during cyclization, resulting in high enantioselectivity at the newly formed stereocenter adjacent to the cyano group. This dual activation strategy avoids problematic direct coordination between nickel and cyanide groups by positioning them away from the metal center during key bond-forming steps.
Impurity control is achieved through careful optimization of reaction parameters including solvent choice (toluene or THF), additive composition (4 equivalents of water), temperature control (80°C), and precise catalyst loading ratios (nickel complex to ligand at 1:1 to 1:1.2). The water additive plays a critical role in suppressing side reactions while promoting proton transfer steps necessary for final product formation without compromising enantioselectivity. The use of non-precious nickel catalysts eliminates concerns about heavy metal contamination in final products, addressing a major regulatory hurdle for pharmaceutical applications where strict limits on residual metals apply.
How to Synthesize Cycloalkenone Compounds Efficiently
This section details the standardized synthesis procedure developed from patent CN111499542A that enables reliable production of high-purity cycloalkenone compounds containing α-cyano group substituted quaternary carbon centers at commercial scale. The methodology represents a significant improvement over previous approaches by eliminating multiple purification steps while maintaining exceptional stereochemical control throughout the process.
- Combine nickel complex catalyst (10 mol%) and chiral ligand (12 mol%) in toluene solvent under argon atmosphere, stirring at room temperature for 30 minutes.
- Add disubstituted malononitrile compound (0.2 mmol) and aryl/alkenyl boron reagent (0.4 mmol), followed by additional toluene (1 mL) and water (4 equivalents).
- Heat reaction mixture to 80°C under oil bath conditions for approximately 24 hours until TLC monitoring confirms complete conversion of starting materials.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits by addressing critical pain points in pharmaceutical intermediate supply chains while significantly improving cost structures through process intensification and elimination of expensive materials.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a fundamental cost advantage by removing expensive palladium or rhodium components from the process while simultaneously eliminating costly metal removal steps required by regulatory standards for pharmaceutical production. The use of readily available starting materials combined with simplified purification procedures reduces overall manufacturing costs through decreased raw material expenses and reduced processing time without requiring specialized equipment or hazardous reagents.
- Enhanced Supply Chain Reliability: The process utilizes common solvents like toluene and standard laboratory equipment without requiring specialized infrastructure or rare materials, significantly improving supply chain resilience compared to methods dependent on scarce or geopolitically sensitive components. The robust nature of the nickel catalysis system tolerates minor variations in starting material quality while maintaining consistent product specifications, reducing batch failures and associated supply disruptions common in more sensitive synthetic routes.
- Scalability and Environmental Compliance: The mild reaction conditions (80°C) enable straightforward scale-up from laboratory to commercial production without requiring specialized high-pressure or cryogenic equipment typically needed for alternative methods involving transition metals or extreme temperatures. The elimination of heavy metal catalysts substantially reduces environmental impact by avoiding toxic metal waste streams while simplifying waste treatment procedures required for regulatory compliance in manufacturing facilities worldwide.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this patented technology in commercial pharmaceutical intermediate manufacturing operations based on technical details provided in patent CN111499542A.
Q: How does this nickel-catalyzed method overcome previous challenges with cyanide-containing compounds?
A: The method addresses previous limitations by utilizing nickel catalysis instead of precious metals, which avoids strong coordination between cyanide groups and transition metals that previously caused decomposition issues during synthesis.
Q: What makes this synthesis particularly valuable for pharmaceutical applications?
A: The resulting compounds contain α-cyano substituted quaternary carbon centers that prevent α-carbon oxidation and toxic cyanide release, while maintaining metabolic stability and enhancing hydrogen bonding properties crucial for drug efficacy.
Q: How does this process improve manufacturing scalability compared to previous methods?
A: The process uses mild reaction conditions (80°C), readily available starting materials, and non-precious metal catalysts, eliminating complex purification steps required by previous methods that used transition metal catalysts requiring extensive removal procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cycloalkenone Compound Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our CDMO expertise ensures seamless technology transfer from laboratory-scale development to full commercial manufacturing while preserving all critical quality attributes required for pharmaceutical applications involving sensitive cyanide-containing compounds.
We invite your technical procurement team to request our Customized Cost-Saving Analysis which details specific COA data and route feasibility assessments tailored to your production requirements for these valuable α-cyano quaternary carbon center compounds.
