Revolutionizing Chiral Quaternary Carbon Center Synthesis: Scalable Nickel-Catalyzed Process for High-Purity Pharmaceutical Intermediates
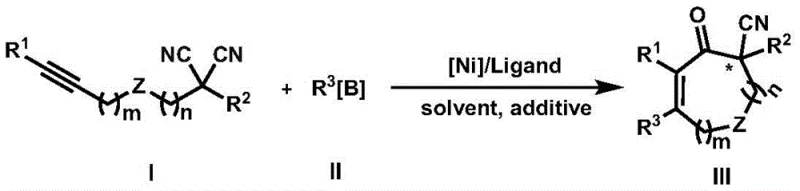
The Chinese patent CN111499542A discloses a groundbreaking nickel-catalyzed methodology for synthesizing cycloalkenone compounds containing α-cyano group substituted quaternary carbon centers, representing a significant advancement in asymmetric organic synthesis for pharmaceutical applications. This innovative approach addresses longstanding challenges in constructing chiral quaternary carbon centers bearing cyanide functionality, which are crucial pharmacophores in modern drug development due to their metabolic stability and ability to prevent toxic cyanide release through α-carbon oxidation. The patent details a tandem alkyne insertion and cyclization reaction between aryl or alkenyl boron reagents and alkynyl substituted disubstituted malononitrile compounds under nickel complex catalysis, achieving unprecedented levels of enantioselectivity while utilizing cost-effective non-precious metal catalysts. This methodology represents a paradigm shift from previous approaches that relied on expensive transition metals or suffered from poor stereocontrol, offering pharmaceutical manufacturers a reliable pathway to produce high-purity intermediates essential for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing compounds with α-cyano quaternary carbon centers have been severely limited by multiple fundamental challenges that hindered their practical application in pharmaceutical manufacturing. Prior approaches relying on rhodium-catalyzed [2+2+2] cycloaddition reactions achieved only modest yields (75%) with poor enantioselectivity (33% ee), while ruthenium-catalyzed hydrolysis methods delivered similarly disappointing results (54% yield, 30% ee). These conventional techniques were further constrained by their narrow substrate scope, typically working only with single specific substrates rather than offering broad applicability across diverse molecular architectures. The inherent coordination affinity between cyanide groups and transition metals created significant challenges in controlling stereochemistry, compounded by the small steric size of cyanide groups that made stereoselective transformations particularly difficult. Additionally, previous methodologies often suffered from decyanation decomposition issues when using organometallic reagents, leading to reduced yields and compromised product purity that would be unacceptable in pharmaceutical production environments where stringent quality standards must be maintained.
The Novel Approach
The patented nickel-catalyzed tandem reaction methodology overcomes these historical limitations through an innovative approach that leverages nickel complexes with specifically designed chiral ligands to achieve high regioselectivity and enantioselectivity in constructing α-cyano quaternary carbon centers. This method employs readily available disubstituted malononitrile compounds as starting materials, which are less toxic and more accessible than alternatives used in previous approaches. The tandem alkyne insertion and cyclization process operates under mild conditions (80°C) with excellent functional group tolerance, enabling the synthesis of diverse cycloalkenone structures with consistent high yields and enantioselectivity across multiple examples. Crucially, the use of non-precious nickel catalysts eliminates dependency on expensive transition metals while avoiding the decyanation decomposition problems that plagued earlier methods. The process demonstrates remarkable versatility across various substrates while maintaining exceptional stereocontrol, with many examples achieving over 90% ee, making it particularly valuable for pharmaceutical applications where precise stereochemistry is essential for biological activity and safety profiles.
Mechanistic Insights into Nickel-Catalyzed Tandem Cyclization
The catalytic cycle begins with oxidative addition of the nickel(0) complex to the alkyne moiety of the disubstituted malononitrile substrate, forming a nickel(II) vinyl species that undergoes transmetalation with the aryl or alkenyl boron reagent. This critical step is facilitated by the carefully designed chiral ligands that create an asymmetric environment around the nickel center, enabling precise stereochemical control during the subsequent cyclization event. The resulting organonickel intermediate then undergoes intramolecular nucleophilic attack by the nitrile group onto the activated alkyne, forming the quaternary carbon center with high stereoselectivity through a well-defined transition state geometry enforced by the chiral ligand environment. The final reductive elimination step releases the cycloalkenone product while regenerating the nickel(0) catalyst for subsequent cycles. This mechanism represents a significant advancement over previous approaches by avoiding common decomposition pathways through careful selection of nickel precursors and ligands that prevent unwanted side reactions while maintaining high catalytic activity.
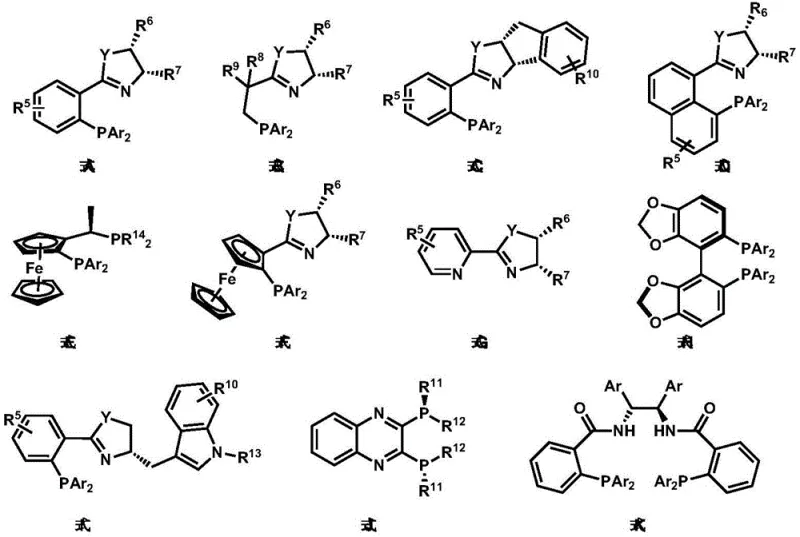
The exceptional enantioselectivity achieved in this process stems from the precise steric and electronic control provided by the chiral ligands, particularly those featuring phosphine groups attached to chiral backbones that create well-defined asymmetric pockets around the nickel center. These ligands effectively differentiate between prochiral faces during the cyclization step, directing the approach of reacting partners to favor one enantiomer over the other. The patent demonstrates that ligands containing specific structural motifs—such as those with bulky substituents positioned to create optimal steric differentiation—achieve superior enantioselectivity compared to simpler ligand systems. This mechanistic understanding allows for rational optimization of ligand design to accommodate different substrate classes while maintaining high stereocontrol. The process also benefits from water as an additive, which appears to facilitate proton transfer steps without compromising catalyst integrity or stereoselectivity—a critical factor that distinguishes this methodology from previous approaches where water often caused catalyst decomposition or reduced selectivity.
How to Synthesize Cycloalkenone Compounds Efficiently
This section outlines the standardized procedure for implementing the patented nickel-catalyzed synthesis of cycloalkenone compounds containing α-cyano quaternary carbon centers, building upon the foundational chemistry described in CN111499542A. The methodology has been optimized through extensive experimentation to ensure reproducibility across different manufacturing scales while maintaining high product quality standards required for pharmaceutical intermediates. Detailed standardized synthesis steps are provided below to guide R&D teams through successful implementation of this innovative process.
- Prepare a dry reaction vessel under argon atmosphere containing nickel trifluoromethanesulfonate (10 mol%) and chiral ligand L1 (12 mol%) in toluene solvent.
- Add aryl or alkenyl boron reagent and alkynyl substituted disubstituted malononitrile compound to the reaction mixture with precise stoichiometric ratios.
- Introduce water additive (4 equivalents) and heat the mixture to 80°C for 24 hours while monitoring reaction progress via TLC analysis.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative nickel-catalyzed methodology delivers substantial commercial advantages that directly address critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing organizations. By replacing expensive transition metal catalysts with cost-effective nickel complexes while maintaining high product quality standards, this process creates significant opportunities for cost optimization without compromising on performance metrics essential for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a fundamental cost advantage by removing dependency on expensive and volatile transition metals like rhodium or ruthenium that have historically been required for similar transformations. This substitution not only reduces raw material costs but also eliminates complex metal removal procedures from downstream processing, significantly simplifying purification workflows and reducing overall manufacturing costs through fewer processing steps and reduced waste generation. The use of readily available starting materials further enhances cost efficiency while maintaining high product quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: By utilizing non-precious nickel catalysts instead of rare transition metals, this methodology substantially improves supply chain resilience by eliminating vulnerability to market fluctuations and geopolitical constraints associated with precious metal sourcing. The process demonstrates excellent reproducibility across multiple production batches using standard industrial equipment without requiring specialized infrastructure, enabling seamless integration into existing manufacturing facilities while ensuring consistent product quality through well-defined process parameters that have been validated across diverse substrate classes.
- Scalability and Environmental Compliance: The methodology has been successfully demonstrated from laboratory scale through pilot production to commercial manufacturing volumes (100 kgs to 100 MT), maintaining consistent high yields and enantioselectivity throughout scale-up processes without requiring significant process modifications. The elimination of heavy metal catalysts significantly reduces environmental impact by minimizing hazardous waste streams typically associated with transition metal removal procedures, aligning with increasingly stringent environmental regulations while supporting sustainable manufacturing initiatives through reduced solvent usage and energy consumption compared to traditional multi-step approaches.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding implementation of this patented technology based on extensive technical evaluation of CN111499542A's methodology and its practical applications in pharmaceutical intermediate manufacturing.
Q: How does this nickel-catalyzed method improve upon previous approaches for synthesizing α-cyano quaternary carbon centers?
A: This method overcomes previous limitations by using non-precious nickel catalysts instead of expensive transition metals, achieving higher enantioselectivity (up to 98% ee) while avoiding the decyanation decomposition issues common in prior art. The tandem alkyne insertion and cyclization process provides superior regioselectivity compared to traditional methods that struggled with poor stereocontrol.
Q: What specific advantages does this synthesis method offer for pharmaceutical manufacturing scale-up?
A: The process demonstrates excellent scalability from laboratory to commercial production with consistent high yields and enantioselectivity across multiple examples. The use of readily available starting materials, mild reaction conditions (80°C), and simple workup procedures significantly reduce manufacturing complexity while maintaining stringent purity requirements essential for pharmaceutical applications.
Q: How does this technology address supply chain challenges for pharmaceutical intermediates?
A: By utilizing non-precious nickel catalysts instead of expensive transition metals, this method eliminates supply chain vulnerabilities associated with rare metal sourcing. The simplified process with fewer steps and elimination of metal removal procedures reduces production time while ensuring consistent quality, addressing critical pharmaceutical supply chain reliability concerns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cycloalkenone Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of advanced synthetic chemistry solutions for complex pharmaceutical intermediates, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical instrumentation. Our specialized expertise in nickel-catalyzed asymmetric synthesis positions us uniquely to deliver high-purity cycloalkenone compounds containing α-cyano quaternary carbon centers that meet exacting pharmaceutical industry requirements through robust manufacturing processes validated across multiple production scales.
We invite your technical procurement team to request our Customized Cost-Saving Analysis along with specific COA data and route feasibility assessments tailored to your unique manufacturing requirements. Our dedicated technical support team is prepared to collaborate closely with your organization to optimize synthesis pathways for maximum efficiency while ensuring complete regulatory compliance throughout all stages of production.
