Advanced Rhodium-Catalyzed Synthesis of 2-Methylene-1-Indanone Derivatives for Commercial Scale-Up
Advanced Rhodium-Catalyzed Synthesis of 2-Methylene-1-Indanone Derivatives for Commercial Scale-Up
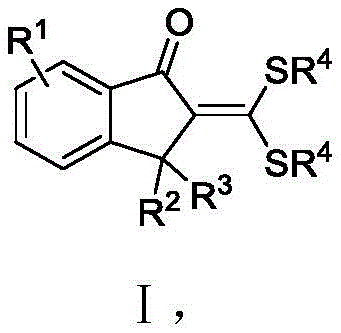
The pharmaceutical industry continuously seeks efficient pathways to access privileged scaffolds that serve as the backbone for novel therapeutic agents. Patent CN112961086B introduces a groundbreaking methodology for the synthesis of 2-methylene-1-indanone derivatives, a structural motif prevalent in bioactive natural products and drug candidates. This technology leverages a transition metal-catalyzed intermolecular oxidative coupling cyclization, transforming readily available S,S-dithioketal compounds and diazo compounds into complex indanone architectures in a single operational step. By bypassing the tedious multi-step sequences traditionally required for indanone construction, this innovation addresses critical bottlenecks in process chemistry, offering a streamlined route that enhances both synthetic efficiency and functional group tolerance for high-purity pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1-indanone skeleton has relied heavily on multi-step condensation reactions that impose significant burdens on process development and supply chain logistics. Traditional protocols often necessitate the pre-functionalization of starting materials, such as the bromination of benzyl groups or the selective reduction of benzoates, which introduces additional unit operations and increases the accumulation of impurities. Furthermore, existing methods frequently operate under harsh conditions that compromise atom economy and generate hazardous byproducts; for instance, certain prior art processes release methanethiol, a toxic and malodorous gas that complicates waste management and environmental compliance. These inefficiencies not only escalate the cost of goods sold but also extend the lead time for high-purity intermediates, creating vulnerabilities in the supply continuity for downstream API production.
The Novel Approach
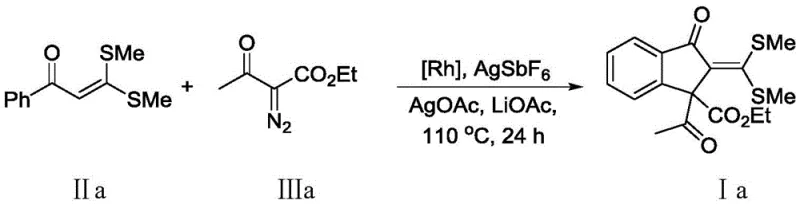
In stark contrast, the methodology disclosed in CN112961086B represents a paradigm shift by enabling the direct assembly of the indanone core through a rhodium-catalyzed oxidative coupling strategy. This novel approach utilizes S,S-dithioketal compounds and diazo compounds as robust building blocks, which undergo cyclization under mild thermal conditions to yield the target 2-methylene-1-indanone derivatives with impressive yields exceeding 80%. The reaction system is remarkably versatile, accommodating a broad spectrum of electronic and steric environments on the aromatic ring, thereby facilitating the rapid generation of diverse chemical libraries for drug discovery. By consolidating what was previously a multi-stage synthesis into a single pot, this technology drastically simplifies the manufacturing workflow and eliminates the need for isolating unstable intermediates.
Mechanistic Insights into Rhodium-Catalyzed Oxidative Coupling
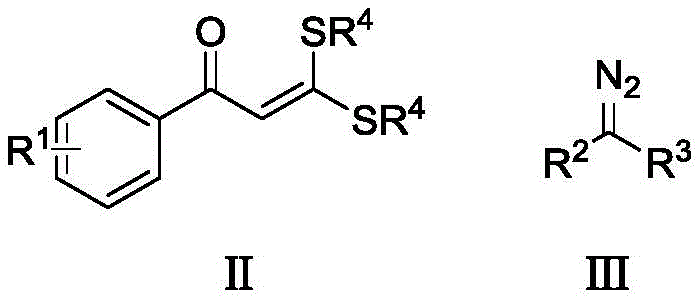
The core of this synthetic breakthrough lies in the sophisticated utilization of a cationic Cp*Rh(III) catalyst system, which orchestrates the precise activation of carbon-hydrogen bonds and subsequent carbon-carbon bond formation. The catalytic cycle initiates with the coordination of the rhodium species to the sulfur-containing substrate, followed by the insertion of the diazo compound to generate a reactive metal-carbene intermediate. This highly electrophilic species then engages in an intramolecular cyclization with the activated aromatic ring, driven by the presence of silver salts which act as both oxidants and halide scavengers to maintain the active cationic state of the catalyst. The inclusion of basic additives such as lithium acetate further stabilizes the transition state, ensuring that the reaction proceeds with high regioselectivity and minimizes the formation of polymeric side products often associated with diazo chemistry.
From an impurity control perspective, this mechanism offers distinct advantages over acid- or base-promoted condensations that often lead to complex degradation profiles. The mild oxidative conditions prevent the over-oxidation of sensitive functional groups, while the specific ligand environment of the pentamethylcyclopentadienyl rhodium complex suppresses non-productive decomposition pathways of the diazo reagent. Consequently, the resulting crude reaction mixtures exhibit cleaner profiles, which significantly reduces the burden on downstream purification units. This level of chemical precision is critical for maintaining stringent purity specifications required for clinical-grade materials, ensuring that the final product meets the rigorous quality standards demanded by global regulatory agencies without extensive recrystallization efforts.
How to Synthesize 2-Methylene-1-Indanone Derivatives Efficiently
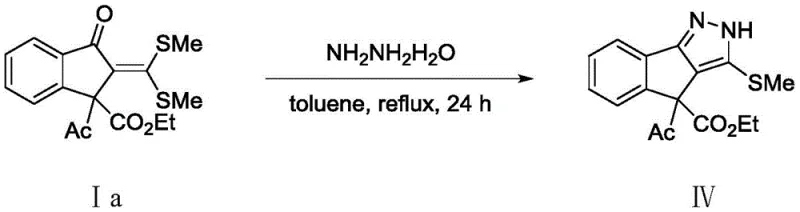
Implementing this synthesis requires careful attention to the stoichiometry of the catalyst system and the selection of appropriate solvents to maximize conversion rates. The protocol typically employs a molar ratio of 1:2 between the S,S-dithioketal substrate and the diazo compound to drive the equilibrium towards completion, utilizing toluene or 1,2-dichloroethane as the reaction medium. The catalyst loading is optimized to be cost-effective, using as little as 5 mol% of the rhodium dimer alongside silver hexafluoroantimonate to generate the active catalytic species in situ. For a detailed walkthrough of the specific experimental parameters, including temperature ramps and workup procedures, please refer to the standardized guide below which outlines the exact operational steps for reproducing these results in a pilot plant setting.
- Combine S,S-dithioketal compound and diazo compound in a solvent such as toluene or 1,2-dichloroethane with a Rhodium catalyst system.
- Add oxidant (AgOAc) and basic additive (LiOAc) to facilitate the oxidative coupling cyclization under inert atmosphere.
- Heat the reaction mixture to 110°C for 24 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Rh-catalyzed methodology translates into tangible strategic benefits regarding cost stability and vendor reliability. By eliminating the need for multiple isolation steps and hazardous reagents, the overall manufacturing footprint is significantly reduced, leading to substantial cost savings in terms of solvent consumption, energy usage, and waste disposal fees. The reliance on commercially available starting materials, such as simple ketones converted to dithioketals and standard diazo esters, mitigates the risk of raw material shortages that often plague complex synthetic routes dependent on custom-synthesized precursors. This robustness ensures a more predictable supply timeline, allowing production planners to forecast inventory needs with greater accuracy and reduce the safety stock levels required to buffer against supply disruptions.
- Cost Reduction in Manufacturing: The consolidation of the synthesis into a single step inherently lowers the operational expenditure by reducing labor hours and equipment occupancy time. Since the process avoids the use of expensive and difficult-to-remove heavy metal catalysts often found in cross-coupling reactions, the downstream purification costs are also minimized, contributing to a lower total cost of ownership for the intermediate. Furthermore, the high atom economy of the oxidative coupling means that a larger proportion of the input mass is converted into valuable product rather than waste, optimizing the yield per batch and maximizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions, which utilize common industrial solvents like toluene and operate at moderate temperatures around 110°C, ensures that the process can be easily transferred between different manufacturing sites without significant re-validation. This flexibility allows for a diversified supply base, reducing the dependency on a single production facility and enhancing the resilience of the supply chain against geopolitical or logistical shocks. Additionally, the stability of the S,S-dithioketal precursors allows for long-term storage and bulk purchasing, enabling companies to capitalize on favorable market pricing for raw materials and secure long-term supply contracts.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is straightforward due to the absence of exothermic hazards associated with rapid diazo decomposition, as the rhodium catalyst controls the release of nitrogen gas in a regulated manner. The process generates minimal hazardous waste compared to traditional methods that produce stoichiometric amounts of salt byproducts or toxic gases, aligning with modern green chemistry principles and simplifying environmental permitting. This environmental compatibility not only reduces regulatory compliance costs but also enhances the corporate sustainability profile, which is increasingly becoming a key criterion for supplier selection in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on the feasibility and limitations of the technology for potential licensees or manufacturing partners. Understanding these nuances is essential for R&D teams evaluating the fit of this chemistry within their existing pipeline and for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of this Rh-catalyzed method over traditional indanone synthesis?
A: Unlike conventional multi-step condensations requiring pre-functionalized substrates, this method constructs the indanone ring in a single step with high atom economy and avoids the release of toxic methanethiol.
Q: Is this process suitable for large-scale pharmaceutical intermediate production?
A: Yes, the reaction utilizes commercially available solvents like toluene and operates at moderate temperatures (110°C), making it highly amenable to commercial scale-up with robust safety profiles.
Q: What is the substrate scope for R1 and R2/R3 groups in this synthesis?
A: The method demonstrates wide adaptability, tolerating various substituents including halogens, alkyl groups, and alkoxy groups on the aromatic ring, as well as diverse ester and ketone functionalities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylene-1-Indanone Derivative Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this innovative patent into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch, thereby guaranteeing that our clients receive materials that meet the highest standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
