Advanced Pd-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates Production
Advanced Pd-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B discloses a highly efficient preparation method for substituted 2,3-dihydroquinolone compounds, a privileged scaffold found in numerous therapeutic agents. As illustrated in the background art, these structures are critical; for instance, specific analogs have demonstrated significant antitumor activity against human cells, while others possess potent analgesic properties. 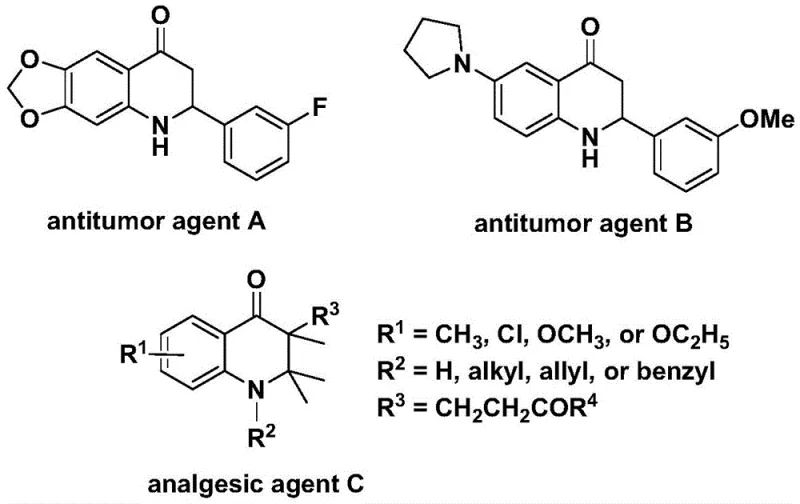 . The disclosed technology leverages a transition metal palladium-catalyzed carbonylation strategy, utilizing N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials. This approach represents a significant advancement over traditional methods by employing a solid carbon monoxide surrogate, thereby enhancing operational safety and simplifying the reaction setup for industrial applications.
. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation strategy, utilizing N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials. This approach represents a significant advancement over traditional methods by employing a solid carbon monoxide surrogate, thereby enhancing operational safety and simplifying the reaction setup for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones often relied on direct carbonylation using carbon monoxide gas. While effective, this traditional approach presents severe logistical and safety challenges for large-scale manufacturing. The use of CO gas requires specialized high-pressure reactors, rigorous leak detection systems, and extensive safety protocols to protect personnel from toxicity. Furthermore, controlling the stoichiometry of a gaseous reagent in a liquid-phase reaction can be difficult, often leading to inconsistent reaction rates and potential side reactions if the gas flow is not perfectly regulated. These factors contribute to higher capital expenditures (CAPEX) and increased operational complexity, making conventional carbonylation less attractive for flexible multi-purpose production facilities that require rapid turnaround between different campaigns.
The Novel Approach
The methodology described in CN112239456B circumvents these issues by utilizing 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide substitute. This innovative substitution allows the reaction to proceed under standard atmospheric pressure conditions without the need for pressurized gas lines. The process involves reacting N-pyridine sulfonyl-o-iodoaniline with an olefin in the presence of a palladium catalyst and the solid CO source in an organic solvent like 1,4-dioxane. 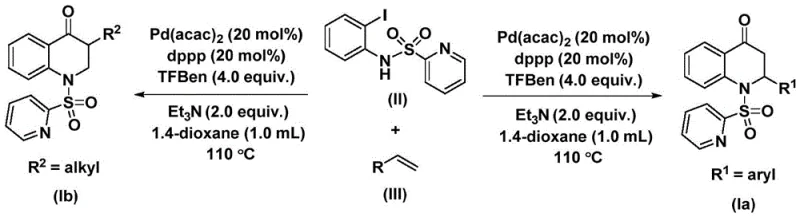 . This shift from gas to solid reagents drastically simplifies the reactor configuration, enabling the use of standard glass-lined or stainless steel vessels. The result is a process that is not only safer but also more amenable to precise dosing and reproducibility, which are critical parameters for maintaining batch-to-batch consistency in pharmaceutical intermediate production.
. This shift from gas to solid reagents drastically simplifies the reactor configuration, enabling the use of standard glass-lined or stainless steel vessels. The result is a process that is not only safer but also more amenable to precise dosing and reproducibility, which are critical parameters for maintaining batch-to-batch consistency in pharmaceutical intermediate production.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is the cornerstone of the subsequent transformation. Following this activation, the carbon monoxide molecule, released in situ from the thermal decomposition of the TFBen additive, inserts into the palladium-carbon bond. This insertion forms an acyl-palladium intermediate, effectively installing the carbonyl functionality required for the quinolone ring closure. The precision of this insertion step is vital, as it dictates the regioselectivity of the final product and ensures that the carbonyl group is positioned correctly within the heterocyclic framework.
Subsequently, the olefin substrate coordinates with the palladium center and undergoes migratory insertion into the acyl-palladium bond. This step creates a new carbon-carbon bond and extends the molecular skeleton, forming a palladium-alkyl intermediate. The cycle concludes with a reductive elimination step, which releases the final substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst. 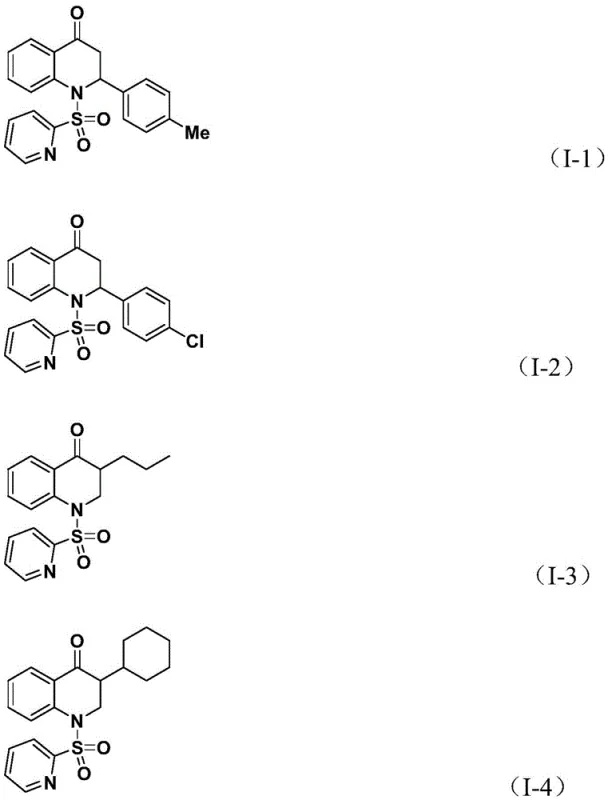 . This mechanism highlights the versatility of the system, as the nature of the olefin (whether aryl or alkyl substituted) directly influences the substitution pattern at the 2- or 3-position of the dihydroquinolone ring. The use of the pyridine sulfonyl group on the nitrogen atom serves a dual purpose: it acts as a protecting group during synthesis and remains part of the final structure, potentially influencing the biological activity or serving as a handle for further derivatization in downstream medicinal chemistry campaigns.
. This mechanism highlights the versatility of the system, as the nature of the olefin (whether aryl or alkyl substituted) directly influences the substitution pattern at the 2- or 3-position of the dihydroquinolone ring. The use of the pyridine sulfonyl group on the nitrogen atom serves a dual purpose: it acts as a protecting group during synthesis and remains part of the final structure, potentially influencing the biological activity or serving as a handle for further derivatization in downstream medicinal chemistry campaigns.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The synthesis protocol outlined in the patent provides a clear pathway for producing these valuable intermediates with high purity. The procedure involves combining the palladium catalyst, ligand, solid CO source, and substrates in a solvent, followed by heating. This standardized approach minimizes variability and ensures that the reaction proceeds to completion within a predictable timeframe. For detailed operational parameters and specific stoichiometric ratios required to achieve optimal yields, please refer to the structured guide below.
- Combine bis(acetylacetone)palladium, dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester (TFBen), N-pyridine sulfonyl-o-iodoaniline, and the specific olefin substrate in 1,4-dioxane within a Schlenk tube.
- Heat the reaction mixture to a temperature range of 100-120 °C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the crude sample with silica gel, and purify the target substituted 2,3-dihydroquinolone compound via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this solid-state carbonylation technology offers transformative benefits regarding cost structure and logistical reliability. The elimination of high-pressure carbon monoxide gas removes a significant bottleneck in facility utilization, allowing manufacturers to run campaigns in standard equipment without lengthy safety certifications for high-pressure systems. This flexibility translates directly into reduced lead times for campaign startup and increased overall plant throughput. Furthermore, the reliance on commercially available starting materials, such as simple olefins and iodoanilines, ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague complex synthetic routes dependent on exotic reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of specialized high-pressure infrastructure. By replacing toxic CO gas with a solid surrogate, the need for expensive pressure-rated reactors and associated safety monitoring systems is eliminated. This significantly lowers the barrier to entry for production and reduces the amortized cost per kilogram of the final intermediate. Additionally, the use of a palladium catalyst system, while requiring metal recovery, operates efficiently at relatively low loadings, and the simplified workup procedure involving filtration and chromatography reduces solvent consumption and waste disposal costs compared to more cumbersome gas-handling protocols.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. The tolerance for various functional groups on the olefin substrate means that a single platform technology can be used to produce a wide library of analogs. This versatility allows suppliers to respond rapidly to changing demand for different derivatives without retooling the entire production line. The use of stable, solid reagents like TFBen also simplifies storage and transportation logistics, as there are no regulatory hurdles associated with shipping compressed toxic gases, ensuring uninterrupted material flow even in regions with strict hazardous material transport regulations.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram levels is straightforward due to the homogeneous nature of the reaction and the absence of gas-liquid mass transfer limitations. The reaction can be easily intensified in larger vessels without the engineering challenges of sparging gas efficiently. From an environmental standpoint, the contained generation of carbon monoxide from the solid surrogate minimizes fugitive emissions, aligning with increasingly stringent environmental, health, and safety (EHS) standards. The ability to recycle the palladium catalyst further enhances the green chemistry profile of the process, reducing the heavy metal burden in the final waste streams and supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What are the safety advantages of using 1,3,5-trimesic acid phenol ester (TFBen) over carbon monoxide gas?
A: Using TFBen as a solid carbon monoxide surrogate eliminates the need for high-pressure gas cylinders and specialized containment equipment required for toxic CO gas. This significantly reduces operational hazards and capital expenditure for reactor infrastructure, making the process safer and more accessible for standard laboratory and pilot plant settings.
Q: What is the substrate compatibility of this carbonylation reaction?
A: The method demonstrates excellent substrate tolerance, successfully accommodating both aryl-substituted olefins (yielding 2-aryl derivatives) and alkyl- or silyl-substituted olefins (yielding 3-alkyl derivatives). Functional groups such as methyl, chloro, fluoro, and tert-butyl on the aryl ring are well-tolerated, allowing for diverse structural modifications.
Q: What yields can be expected from this synthetic protocol?
A: Experimental data indicates isolated yields ranging from approximately 59% to 88%, depending on the specific olefin substrate used. Electron-rich and sterically hindered aryl olefins generally provide higher yields compared to simple alkyl chains, demonstrating the robustness of the catalytic system across different electronic environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we are uniquely positioned to manufacture these complex 2,3-dihydroquinolone intermediates. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with stringent purity specifications, ensuring that every batch meets the rigorous quality standards required by global pharmaceutical clients. Our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of the final product, guaranteeing consistency and reliability for your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can accelerate your timeline to market while maximizing value.
