Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Substituted 2,3-Dihydroquinolone Intermediates for Commercial Scale-Up
Introduction to High-Value Heterocyclic Scaffolds
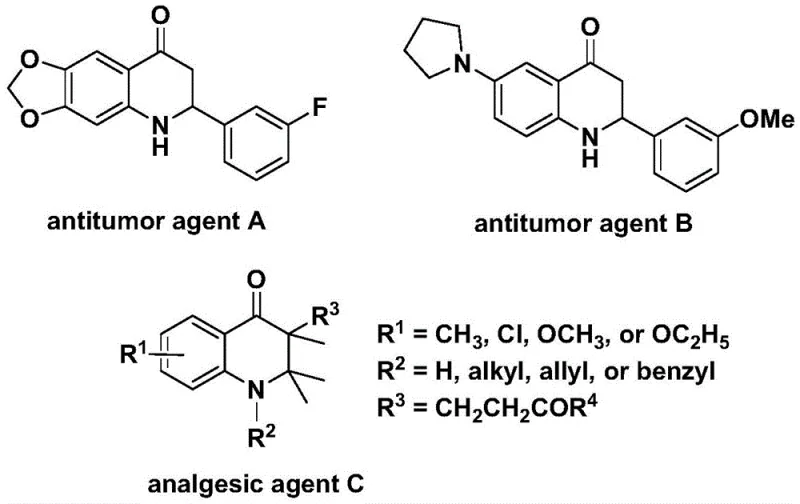
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. As detailed in patent CN112239456B, the substituted 2,3-dihydroquinolone core represents a critical structural motif found in numerous therapeutic agents, including those with demonstrated antitumor and analgesic properties. The biological significance of this scaffold is underscored by prior art referencing compounds with significant anti-cancer activity against human cell lines and potent pain-relieving capabilities, highlighting the urgent demand for robust manufacturing processes. This patent introduces a transformative preparation method that leverages transition metal catalysis to construct these complex skeletons with high efficiency and substrate versatility. By utilizing a palladium-catalyzed carbonylation strategy, the technology addresses long-standing challenges in heterocycle synthesis, offering a pathway to high-purity pharmaceutical intermediates that meet the rigorous standards of global regulatory bodies. For R&D directors and procurement specialists, understanding the nuances of this innovation is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering complex molecules at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for constructing the 2,3-dihydroquinolone ring system often rely on classical cyclization reactions that may suffer from harsh reaction conditions, limited substrate scope, or the requirement for hazardous reagents. Conventional carbonylation processes frequently necessitate the use of high-pressure carbon monoxide gas, which introduces significant safety hazards and requires specialized, expensive infrastructure that can hinder cost reduction in pharma manufacturing. Furthermore, older synthetic routes may struggle with regioselectivity issues or require multi-step sequences that erode overall yield and increase waste generation. The reliance on stoichiometric amounts of toxic reagents or precious metals without efficient recovery systems can also escalate production costs and environmental compliance burdens. These factors collectively create bottlenecks in the supply chain, leading to extended lead times and potential inconsistencies in batch-to-batch quality, which are critical concerns for supply chain heads managing the commercial scale-up of complex pharmaceutical additives.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a solid carbon monoxide surrogate, specifically 1,3,5-trimesic acid phenol ester (TFBen), to drive the carbonylation reaction under much milder and safer conditions. This innovation allows the reaction to proceed at atmospheric pressure equivalents without the need for handling toxic CO gas cylinders, thereby drastically simplifying the reactor setup and enhancing operational safety profiles. The method employs a palladium catalyst system combined with a specific phosphine ligand to facilitate the coupling of N-pyridine sulfonyl-o-iodoaniline with various olefins, achieving high conversion rates within a reasonable timeframe of 24 to 48 hours. This streamlined process not only improves the atom economy of the transformation but also enhances the functional group tolerance, allowing for the introduction of diverse substituents such as halogens, alkyls, and silyl groups without compromising yield. The ability to synthesize both 2-aryl and 3-alkyl substituted variants through simple substrate design provides unparalleled flexibility for medicinal chemists optimizing structure-activity relationships.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
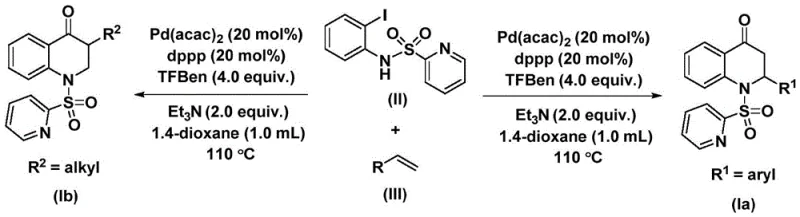
The mechanistic pathway of this transformation involves a sophisticated catalytic cycle initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which is subsequently trapped by carbon monoxide released in situ from the thermal decomposition of the TFBen surrogate. The insertion of carbon monoxide into the palladium-carbon bond forms an acyl-palladium species, a crucial intermediate that dictates the formation of the carbonyl functionality within the final heterocyclic ring. Following this, the coordinated olefin undergoes migratory insertion into the acyl-palladium bond, extending the carbon chain and setting the stage for ring closure. The cycle is completed by a reductive elimination step that releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst for further turnover. This precise control over the catalytic cycle ensures high reaction efficiency and minimizes the formation of undesired byproducts, which is vital for maintaining stringent purity specifications in API intermediate production.
Impurity control is further enhanced by the choice of ligands and additives, such as 1,3-bis(diphenylphosphino)propane (dppp) and triethylamine, which stabilize the palladium center and promote the desired reductive elimination pathway. The use of aprotic solvents like 1,4-dioxane ensures that all starting materials are fully dissolved, facilitating homogeneous catalysis and consistent heat transfer throughout the reaction vessel. This homogeneity is critical for preventing localized hot spots that could lead to decomposition or polymerization of sensitive olefin substrates. Moreover, the compatibility of the system with electron-rich and electron-deficient aryl olefins demonstrates the robustness of the catalytic manifold against electronic variations in the substrate. For quality assurance teams, this mechanistic stability translates to a more predictable impurity profile, simplifying the validation process and reducing the risk of unexpected genotoxic impurities that often plague complex organic syntheses.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and temperature controls as outlined in the patent examples. The process begins with the careful weighing of palladium bis(acetylacetonate) and the dppp ligand, ensuring a molar ratio that maximizes catalytic turnover while minimizing metal residue in the final product. The reaction mixture, containing the iodoaniline derivative, olefin, base, and CO surrogate in dioxane, is heated to 110°C, a temperature optimized to balance reaction rate with reagent stability. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by adding Pd(acac)2, dppp ligand, Et3N, TFBen CO surrogate, N-pyridine sulfonyl-o-iodoaniline, and olefin into 1,4-dioxane solvent.
- Heat the reaction mixture to 110°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion of starting materials.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the high-purity substituted 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial strategic advantages for procurement managers and supply chain leaders seeking to optimize their sourcing strategies for high-value intermediates. The elimination of high-pressure gas infrastructure significantly reduces capital expenditure requirements for manufacturing facilities, allowing for more flexible production scheduling and lower overhead costs. Additionally, the use of commercially available starting materials, such as simple olefins and readily synthesized iodoanilines, ensures a stable and resilient supply chain that is less susceptible to raw material shortages or price volatility. The robustness of the reaction conditions means that the process can be scaled from gram-level laboratory experiments to multi-ton commercial production with minimal re-optimization, thereby reducing lead time for high-purity pharmaceutical intermediates entering the market. This scalability is crucial for meeting the fluctuating demands of downstream drug manufacturers without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The adoption of a solid CO surrogate eliminates the need for specialized high-pressure reactors and associated safety monitoring systems, leading to significant operational cost savings. Furthermore, the high reaction efficiency and yield reduce the amount of raw material waste, directly lowering the cost of goods sold (COGS) per kilogram of product. The simplified post-treatment process, involving standard filtration and chromatography, minimizes labor hours and solvent consumption compared to more cumbersome traditional methods. These cumulative efficiencies result in a more competitive pricing structure for the final intermediate, providing buyers with better value without sacrificing quality standards.
- Enhanced Supply Chain Reliability: By relying on stable, shelf-stable reagents rather than hazardous gases, the manufacturing process becomes inherently safer and less prone to regulatory shutdowns or logistics disruptions. The broad substrate compatibility allows for the rapid switching between different analogues using the same core process platform, enhancing the agility of the supply chain to respond to new drug development pipelines. This flexibility ensures that partners can secure a continuous supply of diverse intermediates from a single reliable pharmaceutical intermediate supplier, mitigating the risks associated with multi-vendor sourcing strategies. Consequently, inventory management becomes more streamlined, and the risk of stockouts due to specialized reagent unavailability is drastically reduced.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing catalytic amounts of palladium and generating fewer hazardous byproducts compared to stoichiometric alternatives. The ability to run the reaction in common organic solvents like dioxane, which can be recovered and recycled, further aligns with increasingly strict environmental regulations governing chemical manufacturing. As production scales up, the consistent performance of the catalyst system ensures that waste streams remain manageable and predictable, facilitating easier permitting and compliance reporting. This environmental stewardship not only protects the ecosystem but also safeguards the brand reputation of downstream pharmaceutical clients who prioritize sustainable sourcing in their vendor audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific drug substance manufacturing workflow.
Q: What are the primary advantages of using TFBen as a CO surrogate in this synthesis?
A: Using 1,3,5-trimesic acid phenol ester (TFBen) eliminates the need for handling toxic carbon monoxide gas directly, significantly improving operational safety and simplifying the equipment requirements for industrial scale-up.
Q: What is the substrate compatibility of this palladium-catalyzed method?
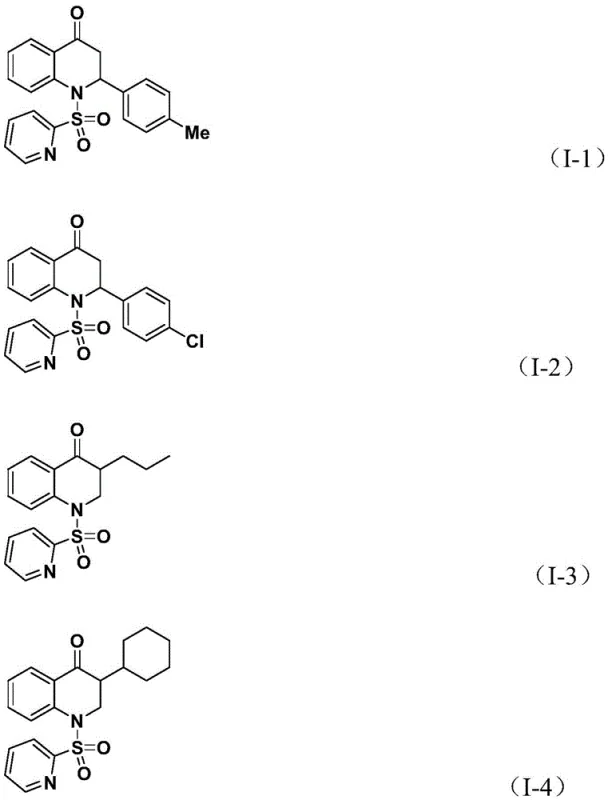
A: The method demonstrates excellent functional group tolerance, successfully accommodating various aryl substituents including methyl, chloro, fluoro, and tert-butyl groups, as well as alkyl and silyl chains on the olefin component.
Q: How does this method impact the purity profile of the final pharmaceutical intermediate?
A: The specific ligand system (dppp) and mild reaction conditions minimize side reactions and byproduct formation, resulting in a cleaner crude reaction mixture that facilitates easier purification and higher final assay purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of novel drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering substituted 2,3-dihydroquinolone compounds with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our dedication to technical excellence means that we do not just supply chemicals; we provide solutions that accelerate your timeline to market while maintaining the highest standards of safety and quality.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate our capability as your trusted partner in fine chemical manufacturing. Let us help you secure a stable supply of these vital building blocks for your next generation of therapeutic agents.
