Advanced Synthesis of Bempedoic Acid Intermediates for Commercial Scale Manufacturing
The pharmaceutical landscape for lipid-lowering therapies has been significantly reshaped by the introduction of Bempedoic acid, a potent ATP citrate lyase (ACL) inhibitor. As demand for this active pharmaceutical ingredient (API) grows, the efficiency of its supply chain becomes paramount for global health outcomes. Patent CN112521282A introduces a transformative synthesis method for a critical Bempedoic acid intermediate, specifically 7-chloro-2,2-dimethylheptanoate. This innovation addresses long-standing bottlenecks in the manufacturing process, offering a pathway to higher purity and reduced production costs. The core of this technology lies in a strategic modification of the alkylation reagents, shifting away from traditional dibromo-alkanes to a mixed halo-alkane system. 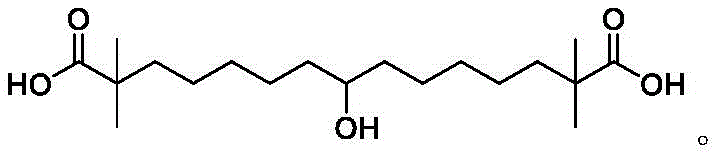 This structural optimization not only streamlines the reaction sequence but also fundamentally alters the impurity profile, ensuring that the resulting high-purity pharmaceutical intermediates meet the rigorous standards required for cardiovascular drug manufacturing.
This structural optimization not only streamlines the reaction sequence but also fundamentally alters the impurity profile, ensuring that the resulting high-purity pharmaceutical intermediates meet the rigorous standards required for cardiovascular drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key heptanoate backbone for Bempedoic acid has relied heavily on the alkylation of isobutyrate esters using 1,5-dibromopentane. While chemically straightforward in theory, this approach suffers from a critical lack of selectivity. Because both terminal positions on the pentane chain possess highly reactive bromine leaving groups, the reaction environment often promotes double alkylation events. This results in the formation of significant quantities of disubstituted byproducts, where two ester molecules attach to a single pentane chain. 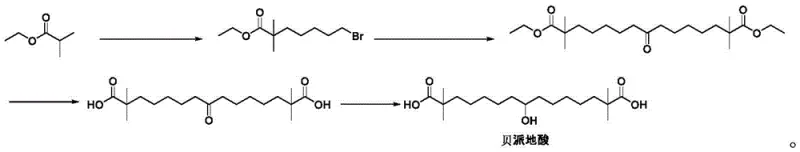 These dimeric impurities are structurally similar to the desired product, making their removal via standard distillation extremely difficult and often necessitating expensive chromatographic purification. Furthermore, the consumption of valuable starting materials to form these waste byproducts drastically reduces the overall atom economy and yield of the process, creating a substantial burden on cost reduction in pharmaceutical intermediates manufacturing.
These dimeric impurities are structurally similar to the desired product, making their removal via standard distillation extremely difficult and often necessitating expensive chromatographic purification. Furthermore, the consumption of valuable starting materials to form these waste byproducts drastically reduces the overall atom economy and yield of the process, creating a substantial burden on cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by introducing 1-bromo-5-chloropentane as the alkylating agent. This seemingly minor change exploits the difference in leaving group ability between bromine and chlorine. Under the specific basic conditions employed, the bromine end of the molecule reacts selectively with the enolate of the isobutyrate, while the chlorine end remains inert. This orthogonality effectively acts as a chemical "stopper," preventing the second alkylation event that plagues the conventional route. Consequently, the formation of the disubstituted dimer is suppressed to negligible levels, often below 1%. This selectivity allows for the isolation of the target 7-chloro-2,2-dimethylheptanoate through simple fractional distillation, bypassing the need for complex purification trains. The result is a robust, scalable process that delivers superior quality intermediates ready for downstream conversion into the final API.
Mechanistic Insights into Selective Enolate Alkylation
The success of this synthesis hinges on the precise control of nucleophilic substitution dynamics. The process begins with the generation of a lithium enolate from ethyl isobutyrate using a strong, non-nucleophilic base such as Lithium Diisopropylamide (LDA) at cryogenic temperatures, typically around -70°C. This ensures the quantitative formation of the kinetic enolate without competing side reactions. When 1-bromo-5-chloropentane is introduced, the soft nucleophilic character of the enolate preferentially attacks the carbon bearing the bromine atom. Bromine is a superior leaving group compared to chlorine due to its larger atomic radius and weaker bond strength with carbon. This kinetic preference ensures that the substitution occurs exclusively at the bromo-terminus. The chlorine atom, being a poorer leaving group under these specific conditions, survives the reaction intact. This preserved chloro-functionality is crucial, as it serves as the handle for the subsequent chain-extension step involving p-toluenesulfonylmethylisocyanide (TosMIC). By maintaining this functional group integrity, the synthesis avoids the dead-end pathways associated with double alkylation.
From an impurity control perspective, this mechanism provides a definitive solution to the "dimer problem." In the traditional dibromo route, once the first alkylation occurs, the remaining bromine on the product is still reactive enough to undergo a second alkylation with another equivalent of enolate, especially if local concentrations or temperatures fluctuate. In the novel chloro-bromo route, even if a second equivalent of enolate were present, the remaining chlorine terminus is sufficiently unreactive to prevent further substitution under the controlled reaction parameters. This inherent chemical stability translates directly into a cleaner crude reaction mixture. The reduction in structural analogs simplifies the downstream processing significantly, allowing manufacturers to achieve high purity specifications without resorting to preparative HPLC or silica gel chromatography, which are notoriously difficult to scale for multi-ton production.
How to Synthesize 7-Chloro-2,2-dimethylheptanoate Efficiently
The implementation of this synthesis route requires careful attention to temperature control and reagent addition rates to maximize the benefits of the selective alkylation mechanism. The process is designed to be operationally simple, utilizing common organic solvents like tetrahydrofuran (THF) and standard workup procedures involving aqueous quenching and extraction. The following guide outlines the critical stages of the transformation, emphasizing the conditions necessary to maintain high selectivity and yield. For detailed operational parameters and safety protocols, please refer to the standardized synthesis steps provided below.
- Perform selective alkylation of ethyl isobutyrate with 1-bromo-5-chloropentane using LDA base at low temperature to form ethyl 7-chloro-2,2-dimethylheptanoate.
- React the chloro-ester intermediate with p-toluenesulfonylmethylisocyanide (TosMIC) under basic conditions to extend the carbon chain and introduce the isonitrile functionality.
- Execute acid-catalyzed hydrolysis and cyclization of the isonitrile diester to yield the final keto-diester precursor for Bempedoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification process. By eliminating the formation of hard-to-remove dimeric impurities, the technology removes the necessity for column chromatography, a unit operation that is often a bottleneck in fine chemical manufacturing due to high solvent consumption and low throughput. This shift to distillation-based purification significantly lowers the operational expenditure (OPEX) associated with solvent recovery and waste disposal. Furthermore, the high selectivity of the reaction improves the overall material throughput, meaning less raw material is wasted on byproduct formation. This efficiency gain contributes to substantial cost savings in the final price of the intermediate, enhancing the competitiveness of the supply chain.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps results in a leaner manufacturing process with lower solvent usage and reduced labor hours. The ability to purify the intermediate via fractional distillation allows for continuous or large-batch processing, which is far more economically viable than batch-wise column loading. Additionally, the high yield of the alkylation step ensures that expensive starting materials like specialized bases and esters are utilized efficiently, minimizing the cost per kilogram of the final product. These factors combine to create a highly cost-effective production model suitable for generic drug markets.
- Enhanced Supply Chain Reliability: The raw materials required for this process, specifically 1-bromo-5-chloropentane and ethyl isobutyrate, are commodity chemicals with stable global supply chains. Unlike some exotic reagents that may face sourcing volatility, these inputs are readily available from multiple vendors, reducing the risk of supply disruption. The robustness of the reaction conditions, which tolerate slight variations without catastrophic loss of selectivity, further ensures consistent production output. This reliability is critical for maintaining the continuity of API supply for essential cardiovascular medications, safeguarding against market shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving seamlessly from laboratory glassware to industrial reactors. The avoidance of silica gel and the reduction in solvent volume align with green chemistry principles, lowering the environmental footprint of the manufacturing site. Reduced solvent waste translates to lower disposal costs and easier compliance with increasingly stringent environmental regulations. The simplicity of the workup—typically involving water quenching and phase separation—facilitates rapid turnover of reactor vessels, increasing the overall capacity of the production facility without the need for significant capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Bempedoic acid intermediates using this patented methodology. These answers are derived directly from the experimental data and technical disclosures found in the source patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio.
Q: Why is 1-bromo-5-chloropentane preferred over 1,5-dibromopentane for this synthesis?
A: Using 1,5-dibromopentane often leads to double alkylation (disubstitution) because both bromine atoms are highly reactive leaving groups. By using 1-bromo-5-chloropentane, the chlorine atom acts as a dormant group during the initial alkylation, preventing dimerization and significantly improving the purity of the mono-alkylated intermediate.
Q: Does this process require expensive column chromatography for purification?
A: No. One of the primary advantages of this novel method is that the target intermediate can be purified via fractional distillation. This eliminates the need for costly and time-consuming column chromatography, making the process much more suitable for large-scale industrial production.
Q: What are the typical yields for the initial alkylation step?
A: According to the experimental data in patent CN112521282A, the alkylation step using LDA and 1-bromo-5-chloropentane can achieve yields exceeding 90%, with the disubstituted byproduct content reduced to as low as 0.3%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bempedoic Acid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence ensures that the 7-chloro-2,2-dimethylheptanoate we supply meets the exacting standards required for GMP-grade API synthesis, providing a solid foundation for your drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your total cost of ownership. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable source of high-quality intermediates that combine cutting-edge chemistry with commercial pragmatism, securing your position in the competitive lipid-lowering drug market.
