Scalable Manufacturing of High-Purity Bempedoic Acid Intermediates via Novel Trityl Protection Strategy
Scalable Manufacturing of High-Purity Bempedoic Acid Intermediates via Novel Trityl Protection Strategy
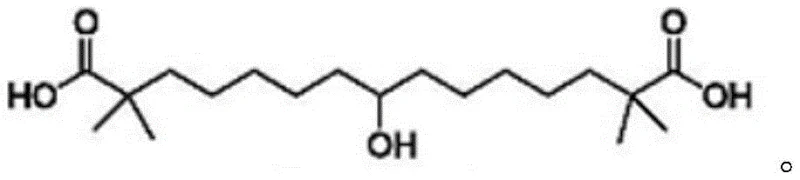
The global demand for effective lipid-lowering therapies has intensified the focus on Bempedoic Acid, a first-in-class ATP citrate lyase inhibitor approved for treating hypercholesterolemia. As supply chains for cardiovascular active pharmaceutical ingredients (APIs) face increasing pressure, the efficiency and safety of synthetic routes for key intermediates become paramount. Patent CN115611739A introduces a groundbreaking preparation method for Bempedoic Acid intermediates that addresses critical bottlenecks in traditional manufacturing. This technical insight report analyzes the proprietary trityl-protection strategy detailed in the patent, offering a pathway to high-purity pharmaceutical intermediates with enhanced operational safety. By leveraging triphenylmethyl (trityl) groups, this methodology circumvents the use of hazardous reagents found in legacy processes, thereby facilitating cost reduction in API manufacturing while ensuring robust quality control suitable for regulatory submission.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Bempedoic Acid backbone has relied on routes fraught with significant safety and environmental hazards. For instance, the methodology disclosed in WO2004067489 utilizes p-toluenesulfonylmethyl isocyanide, a reagent known for its high toxicity and poor atom economy. Furthermore, this legacy process necessitates the use of sodium hydride (NaH), a pyrophoric substance that poses severe explosion risks upon contact with moisture or air, complicating industrial scale-up and requiring specialized containment infrastructure. Beyond safety concerns, these conventional pathways often generate potential genotoxic impurities, such as p-toluenesulfonyl derivatives, which are notoriously difficult to purge to acceptable limits. Additionally, the purification of key intermediates in these older routes frequently demands column chromatography, a technique that is economically unviable and operationally cumbersome for multi-kilogram or ton-scale production, leading to lower overall yields and inconsistent batch quality.
The Novel Approach
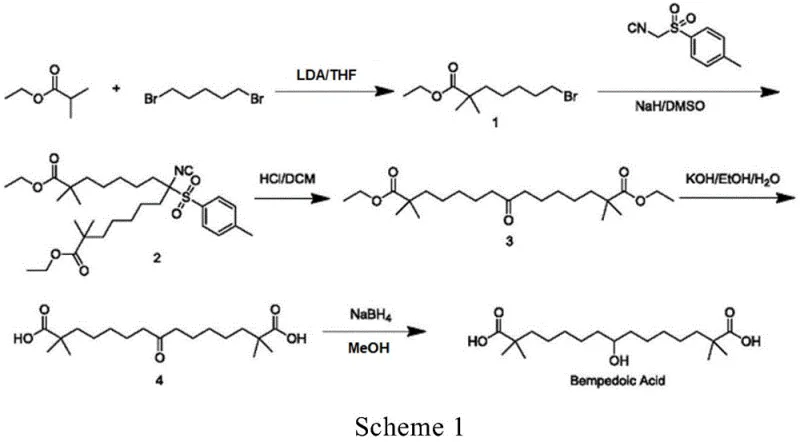
In stark contrast, the innovative route described in CN115611739A employs a strategic trityl protection mechanism that fundamentally alters the reaction landscape. By protecting the isobutyric acid moiety as a triphenylmethyl ester early in the synthesis, the process enables the use of milder and safer alkylation conditions. The core transformation involves the coupling of a trityl-protected bromo-ester with diethyl acetonedicarboxylate, a reaction that proceeds with high selectivity and minimal side-product formation. A distinct advantage of this approach is the physical properties of the resulting intermediate (Compound 3); the bulky trityl groups impart crystallinity that allows for purification via simple recrystallization from solvent mixtures like acetone and n-hexane. This eliminates the need for silica gel chromatography entirely, drastically reducing solvent consumption and processing time. The result is a streamlined workflow that delivers superior yields and purity profiles, making it an ideal candidate for a reliable pharmaceutical intermediate supplier aiming for commercial viability.
Mechanistic Insights into Trityl-Mediated Alkylation and Condensation
The chemical elegance of this synthesis lies in the sequential construction of the carbon backbone using robust protecting group chemistry. The process initiates with the esterification of isobutyric acid and triphenylmethanol using DIC (1,3-diisopropylcarbodiimide) in dichloromethane, forming Compound 1 with exceptional efficiency. Subsequently, Compound 1 undergoes deprotonation by lithium diisopropylamide (LDA) in tetrahydrofuran at cryogenic temperatures (-10°C to 0°C) to generate a stable enolate. This nucleophile attacks 1,4-dibromobutane in a controlled SN2 reaction to install the necessary alkyl chain, yielding Compound 2. The pivotal step involves the double alkylation of diethyl acetonedicarboxylate with Compound 2. Here, the presence of an iodide salt, such as potassium iodide, acts as a catalytic promoter, likely facilitating a Finkelstein-type halogen exchange to enhance the electrophilicity of the bromo-ester. The reaction is conducted in ethanol with a carbonate base (e.g., cesium or potassium carbonate) at moderate temperatures (60-70°C), ensuring complete conversion while preserving the integrity of the ester functionalities.
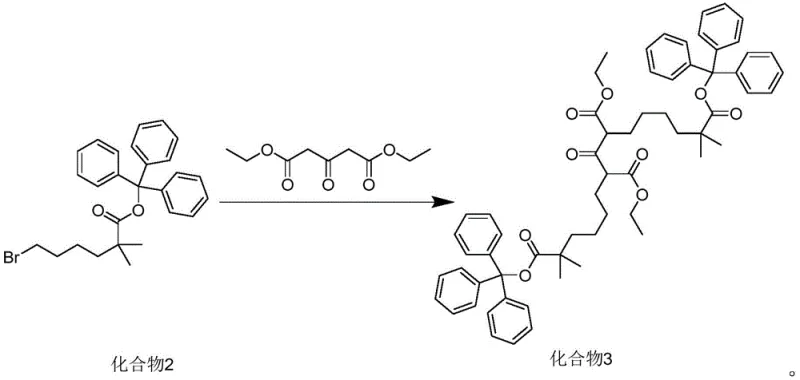
Impurity control is intrinsically built into this mechanistic design. The steric bulk of the trityl group prevents unwanted poly-alkylation or self-condensation side reactions that often plague linear syntheses. Furthermore, the final hydrolysis step serves a dual purpose: it cleaves the ethyl esters to reveal the carboxylic acids and simultaneously removes the trityl protecting groups under acidic workup conditions. The triphenylmethanol byproduct generated during deprotection is lipophilic and can be efficiently extracted into non-polar solvents like n-hexane, leaving the polar diacid product in the aqueous phase. This phase-separation capability ensures that the final product, 8-keto-2,2,14,14-tetramethylpentadecanedioic acid (Compound 4), is obtained with HPLC purity exceeding 99%, effectively mitigating the risk of carrying over organic impurities into the final API synthesis.
How to Synthesize Bempedoic Acid Intermediate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process is divided into four distinct operational stages: protection, chain extension, condensation, and deprotection. Each stage utilizes commercially available raw materials and standard reactor equipment, avoiding the need for exotic catalysts or high-pressure vessels. The following guide outlines the critical operational windows defined in the patent data, emphasizing the importance of temperature control during the LDA addition and the specific solvent ratios required for optimal recrystallization. For detailed standard operating procedures and safety data sheets regarding these specific transformations, please refer to the technical documentation provided below.
- Protect isobutyric acid with triphenylmethanol using DIC in DCM to form Compound 1.
- Alkylate Compound 1 with 1,4-dibromobutane using LDA in THF at low temperature to yield Compound 2.
- Condense Compound 2 with diethyl acetonedicarboxylate using carbonate base and iodide catalyst in ethanol to form Compound 3.
- Hydrolyze Compound 3 with aqueous alkali followed by acidification and recrystallization to obtain the final keto-diacid Compound 4.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of the trityl-protection route offers substantial advantages over legacy methodologies, directly impacting the bottom line and supply continuity. The elimination of hazardous reagents like sodium hydride removes the requirement for specialized storage facilities and expensive hazard mitigation protocols, thereby lowering the overhead costs associated with manufacturing. Moreover, the ability to purify intermediates through crystallization rather than chromatography significantly reduces the consumption of high-grade silica and large volumes of elution solvents, leading to a greener and more cost-effective process. This efficiency translates into shorter cycle times and higher throughput capacity, allowing suppliers to respond more agilely to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the simplification of downstream processing. By replacing column chromatography with recrystallization, the process drastically cuts down on solvent usage and waste disposal costs. The high yield of the protection step (over 95%) and the condensation step (over 80%) ensures that raw material utilization is maximized, minimizing the cost of goods sold (COGS). Additionally, the recovery of triphenylmethanol during the hydrolysis workup offers a potential avenue for recycling valuable starting materials, further enhancing the overall atom economy and financial viability of the production campaign.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as triphenylmethanol, isobutyric acid, and diethyl acetonedicarboxylate ensures a stable and diversified supply base, reducing the risk of raw material shortages. Unlike routes dependent on custom-synthesized toxic isocyanides, this method utilizes reagents that are readily available from multiple global vendors. The robustness of the reaction conditions, which tolerate standard industrial equipment materials, also means that production can be easily transferred between different manufacturing sites or scaled up from pilot to commercial scale without significant re-engineering, guaranteeing consistent supply continuity for long-term contracts.
- Scalability and Environmental Compliance: This synthesis pathway aligns perfectly with modern environmental, health, and safety (EHS) regulations. The avoidance of genotoxic reagents simplifies the regulatory filing process and reduces the burden of extensive impurity qualification studies. The aqueous workup and solvent recovery systems are straightforward to implement, minimizing the environmental footprint of the manufacturing process. The inherent safety of the reagents reduces the risk of plant shutdowns due to safety incidents, ensuring that production schedules are met reliably. This makes the process highly attractive for commercial scale-up of complex pharmaceutical intermediates where regulatory compliance and operational safety are non-negotiable prerequisites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating technology transfer and procurement officers assessing supplier capabilities. The responses highlight the specific advantages of the trityl route in terms of purity, safety, and operational simplicity.
Q: Why is the trityl protection group advantageous for Bempedoic Acid synthesis?
A: The trityl (triphenylmethyl) group provides robust protection for the carboxylic acid during harsh alkylation conditions and can be easily removed during hydrolysis. Crucially, it allows the intermediate (Compound 3) to be purified via simple recrystallization rather than column chromatography, significantly improving scalability and reducing production costs.
Q: How does this method improve safety compared to prior art routes?
A: Unlike previous methods that utilize highly toxic p-toluenesulfonylmethyl isocyanide or pyrophoric sodium hydride (NaH), this novel route employs safer reagents such as lithium diisopropylamide (LDA) under controlled conditions and standard carbonate bases. This eliminates the risk of spontaneous combustion and reduces the formation of potential genotoxic impurities.
Q: What is the expected purity of the final intermediate using this process?
A: The patent data indicates that through optimized recrystallization steps, particularly for Compound 3 and the final Compound 4, HPLC purity exceeding 98% to 99.6% can be consistently achieved, meeting the stringent quality requirements for pharmaceutical API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bempedoic Acid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising trityl-protection chemistry described in CN115611739A can be seamlessly translated into reliable supply. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Bempedoic Acid intermediate meets the exacting standards required for global pharmaceutical markets. Our commitment to quality assurance ensures that potential genotoxic impurities are monitored and controlled at every stage of the synthesis.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains for cardiovascular therapeutics. By leveraging our advanced synthesis platforms, you can secure a stable source of high-purity pharmaceutical intermediates while achieving significant operational efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can support your drug development and commercialization goals.
