Advanced Rhodium-Catalyzed Synthesis of Indolo Isoquinoline Intermediates for Commercial Scale
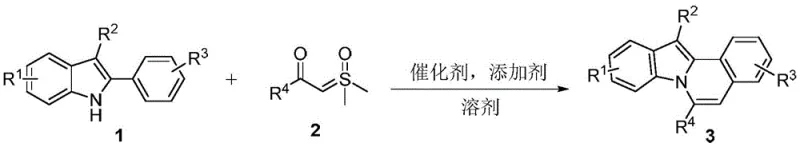
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex nitrogen-containing heterocyclic scaffolds, which serve as critical backbones for bioactive molecules. Patent CN108640917B introduces a groundbreaking synthetic approach for indolo[2,1-a]isoquinoline compounds, utilizing a rhodium-catalyzed one-pot tandem reaction between 2-aryl-3-alkylindole derivatives and sulfur ylides. This innovation addresses long-standing challenges in heterocyclic chemistry by offering a pathway that is not only operationally simple but also exhibits remarkable tolerance to diverse functional groups. For R&D directors and procurement specialists, this technology represents a significant leap forward in accessing high-value intermediates with improved atom economy and reduced environmental footprint. The ability to synthesize these tetracyclic fused systems under mild conditions opens new avenues for the development of agrochemicals, dyes, and potential pharmaceutical agents, positioning this method as a cornerstone for modern industrial organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolo isoquinoline skeleton has relied on multi-step sequences that often involve harsh reaction conditions and the use of hazardous reagents. Traditional pathways frequently suffer from poor atom economy, requiring extensive protection and deprotection strategies that inflate production costs and generate substantial chemical waste. Furthermore, the availability of specific starting materials for these older methods can be restricted, leading to supply chain bottlenecks and inconsistent quality in the final active pharmaceutical ingredients. The necessity to isolate and purify reactive intermediates in stepwise processes not only prolongs the manufacturing timeline but also increases the risk of product degradation and safety hazards associated with handling unstable species. These inefficiencies create significant barriers for scaling up production to meet the demands of the global market, necessitating a paradigm shift towards more streamlined and sustainable synthetic technologies.
The Novel Approach
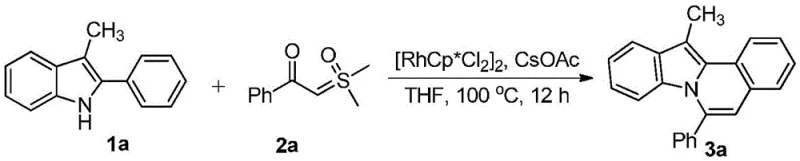
In stark contrast to legacy techniques, the disclosed invention leverages a sophisticated rhodium-catalyzed C-H activation strategy to achieve direct cyclization in a single vessel. This novel approach employs readily available 2-aryl-3-alkylindoles and sulfur ylides as building blocks, reacting them in the presence of a dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer catalyst and a cesium acetate additive. The reaction proceeds smoothly at temperatures ranging from 80°C to 140°C, with optimal results observed around 100°C in solvents like tetrahydrofuran. By bypassing the need for intermediate isolation, this one-pot protocol drastically simplifies the operational workflow and enhances overall process safety. The broad substrate scope allows for the introduction of various electronic and steric modifiers, enabling the rapid generation of diverse chemical libraries for drug discovery programs without compromising on yield or purity standards.
Mechanistic Insights into Rh-Catalyzed Cyclization
The core of this technological advancement lies in the precise mechanistic orchestration facilitated by the rhodium catalyst. The reaction initiates with the coordination of the rhodium species to the indole substrate, promoting a selective C-H bond activation at the strategic position required for ring closure. Subsequent insertion of the sulfur ylide into the metal-carbon bond generates a key organometallic intermediate, which then undergoes intramolecular cyclization to form the fused isoquinoline ring system. This catalytic cycle is meticulously balanced by the cesium acetate additive, which acts as a base to facilitate proton transfer and regenerate the active catalyst species. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters, as it highlights the importance of maintaining strict stoichiometric ratios between the catalyst, additive, and substrates to maximize turnover numbers and minimize metal contamination in the final product.
From an impurity control perspective, the mild nature of this catalytic system plays a pivotal role in ensuring high product fidelity. Unlike aggressive acid or base-mediated cyclizations that can lead to polymerization or decomposition of sensitive functional groups, this rhodium-catalyzed pathway operates under neutral to slightly basic conditions. This gentleness preserves the integrity of labile substituents such as halogens and esters, which are often present in advanced intermediates. Furthermore, the high selectivity of the C-H activation step reduces the formation of regioisomers and side products, thereby simplifying downstream purification processes. For quality assurance teams, this means a cleaner crude reaction profile and a more consistent impurity spectrum, which are critical factors for regulatory compliance and successful technology transfer to commercial manufacturing sites.
How to Synthesize Indolo[2,1-a]isoquinoline Efficiently
Implementing this synthesis route requires careful attention to the preparation of the reaction mixture and the control of thermal parameters. The standard protocol involves dissolving the 2-aryl-3-alkylindole starting material in a dry aprotic solvent, followed by the sequential addition of the sulfur ylide, the rhodium catalyst dimer, and the cesium acetate additive. The reaction vessel is then sealed and heated to the target temperature, typically maintained for a duration sufficient to ensure complete conversion as monitored by chromatographic methods. Upon completion, the mixture is cooled and subjected to a standard aqueous workup involving extraction with ethyl acetate, washing with brine, and drying over anhydrous sodium sulfate. The detailed standardized synthesis steps see the guide below.
- Dissolve 2-aryl-3-alkylindole compound in a suitable solvent such as tetrahydrofuran.
- Add sulfur ylide, dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer catalyst, and cesium acetate additive.
- Stir and react at 80-140°C, preferably 100°C, followed by standard extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of multiple isolation steps inherently reduces the consumption of solvents and consumables, leading to a leaner manufacturing process with a lower environmental burden. This efficiency translates directly into cost optimization, as fewer unit operations mean reduced labor hours, lower energy consumption for heating and cooling cycles, and decreased waste disposal fees. Moreover, the use of commercially available and stable starting materials mitigates the risk of raw material shortages, ensuring a more resilient supply chain capable of withstanding market fluctuations. The robustness of the reaction conditions also implies a higher success rate in scale-up trials, reducing the time and capital investment required to bring new intermediates from the laboratory to pilot and commercial production scales.
- Cost Reduction in Manufacturing: The one-pot nature of this reaction significantly lowers the operational expenditure by removing the need for intermediate purification stages. By avoiding the isolation of potentially unstable intermediates, manufacturers save on chromatography media, filtration equipment, and solvent recovery costs. The high atom economy of the tandem reaction ensures that a greater proportion of raw materials are converted into the desired product, minimizing waste generation. Additionally, the catalyst loading is optimized to be low, reducing the expense associated with precious metal usage while maintaining high catalytic efficiency. These factors collectively contribute to a substantially reduced cost of goods sold, enhancing the competitiveness of the final pharmaceutical or agrochemical product in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on readily accessible starting materials such as substituted indoles and sulfur ylides ensures a stable and predictable supply chain. Unlike specialized reagents that may have limited suppliers or long lead times, these precursors are commonly produced by multiple chemical vendors, reducing the risk of single-source dependency. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, eliminating the need for specialized corrosion-resistant equipment. This compatibility with existing infrastructure accelerates the timeline for technology transfer and commercialization, allowing companies to respond more quickly to market demands and reduce lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process is facilitated by its straightforward operational requirements and the absence of hazardous reagents or extreme conditions. The reaction generates minimal byproducts, simplifying effluent treatment and helping facilities meet stringent environmental regulations. The ability to run the reaction in common solvents like tetrahydrofuran or toluene further aids in solvent recovery and recycling initiatives. As regulatory pressures on chemical manufacturing increase, adopting greener synthetic routes like this becomes a critical component of corporate sustainability strategies. The process demonstrates excellent potential for continuous flow adaptation, which could further enhance safety and throughput in large-scale production environments, ensuring long-term viability and compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this indolo isoquinoline synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on catalyst performance, substrate flexibility, and process robustness. Understanding these aspects is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the objective capabilities of the method as demonstrated in the provided examples, ensuring accurate expectations for project planning and resource allocation.
Q: What catalyst system is required for this indolo isoquinoline synthesis?
A: The process utilizes a dichloro(pentamethylcyclopentadienyl)rhodium(III) dimer catalyst with cesium acetate as an additive, enabling mild reaction conditions.
Q: Does this method support diverse substrate scopes?
A: Yes, the protocol accommodates various substituents including halogens, alkyl, and alkoxy groups on both the indole and phenyl rings, ensuring broad applicability.
Q: How does this route improve manufacturing efficiency?
A: By employing a one-pot tandem reaction strategy, it eliminates the need for isolating unstable intermediates, thereby simplifying the workflow and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[2,1-a]isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics and fine chemicals. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN108640917B can be successfully translated into industrial reality. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and consistency makes us a trusted partner for multinational corporations seeking to secure their supply chains for complex heterocyclic building blocks.
We invite you to collaborate with us to explore the full potential of this rhodium-catalyzed technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain. Together, we can accelerate the development of life-saving medicines and advanced materials through superior chemical manufacturing solutions.
