Sustainable Rhodium-Catalyzed Aminocarbonylation for Scalable Acetamide Production
Sustainable Rhodium-Catalyzed Aminocarbonylation for Scalable Acetamide Production
The landscape of organic synthesis is continuously evolving towards greener, more efficient methodologies that align with modern sustainability goals while maintaining high economic viability. A groundbreaking development in this sector is detailed in Chinese Patent CN112812032A, which discloses a novel preparation method for acetamide compounds utilizing a rhodium-catalyzed aminocarbonylation strategy. This innovative approach distinguishes itself by employing dimethyl carbonate (DMC) not merely as a solvent, but critically as a dual-function reagent serving as both the green reaction medium and the essential C1 carbon source. For R&D directors and process chemists seeking robust pathways for constructing carbonyl-containing scaffolds, this technology represents a significant leap forward, replacing hazardous traditional reagents with a benign, biodegradable alternative that streamlines the synthetic workflow and minimizes environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of acetamide motifs and related carbonyl structures has relied heavily on classical methylation and carbonylation protocols that pose substantial safety and environmental challenges. Conventional methylating agents such as diazomethane, dimethyl sulfate, and methyl iodide are frequently employed, yet they are plagued by severe drawbacks including high toxicity, corrosiveness, and in the case of diazomethane, explosive instability. Furthermore, these traditional stoichiometric reactions typically necessitate the use of excessive amounts of base to drive the transformation, inevitably leading to the generation of large quantities of useless inorganic salt byproducts that require complex and costly disposal procedures. From a supply chain perspective, the reliance on such hazardous materials complicates logistics, increases regulatory compliance burdens, and introduces significant operational risks that can disrupt continuous manufacturing operations.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN112812032A introduces a transformative catalytic cycle that leverages the unique reactivity of dimethyl carbonate under rhodium catalysis. This novel route effectively bypasses the need for dangerous alkylating agents by utilizing DMC, a low-toxicity compound that can be derived from atmospheric carbon dioxide, thereby closing the carbon loop. The reaction operates under relatively mild thermal conditions, typically around 120°C, and demonstrates exceptional functional group tolerance, accommodating a wide array of substituents including electron-withdrawing trifluoromethyl groups and electron-donating alkoxy moieties without compromising yield. By integrating the solvent and the carbon source into a single reagent, this method drastically simplifies the reaction matrix, reduces the overall E-factor of the process, and offers a safer, more scalable alternative for the production of high-value acetamide intermediates.
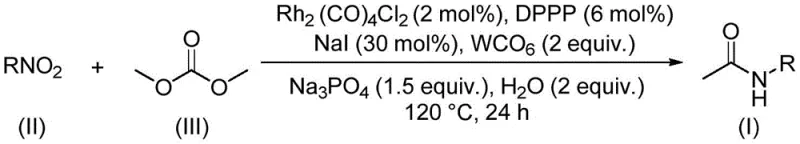
Mechanistic Insights into Rhodium-Catalyzed Aminocarbonylation
The core of this synthetic breakthrough lies in the sophisticated interplay between the rhodium catalyst system and the nitro compound substrate within the dimethyl carbonate medium. The catalytic cycle likely initiates with the activation of the nitro group by the rhodium species, potentially facilitated by the presence of tungsten carbonyl and iodide promoters which assist in the reduction and subsequent carbonylation steps. Unlike traditional carbonylations that require high-pressure carbon monoxide gas, this system ingeniously generates the necessary carbonyl equivalents in situ from the decomposition or activation of dimethyl carbonate. This mechanistic pathway allows for the direct insertion of the carbonyl group into the nitrogen-carbon bond, forming the stable acetamide linkage with high atom economy. The presence of ligands such as 1,3-bis(diphenylphosphino)propane (DPPP) further stabilizes the active rhodium species, ensuring sustained catalytic turnover and preventing catalyst deactivation over the extended reaction period of 24 hours.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions commonly associated with free radical pathways or harsh acidic conditions. The specificity of the rhodium-catalyzed transformation ensures that sensitive functional groups on the aromatic ring, such as halogens or esters, remain intact, resulting in a cleaner crude reaction profile. This high selectivity reduces the burden on downstream purification processes, as fewer structurally similar byproducts are generated compared to non-catalytic thermal methods. For quality assurance teams, this translates to a more predictable impurity spectrum, facilitating easier method validation and ensuring that the final acetamide products meet stringent purity specifications required for pharmaceutical applications without the need for extensive recrystallization or chromatographic separation.
How to Synthesize Acetamide Compounds Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and reproducibility. The protocol involves charging a sealed vessel with the nitro compound substrate, dimethyl carbonate, and a specific cocktail of catalysts and additives including dirhodium tetracarbonyl dichloride, DPPP, tungsten carbonyl, sodium phosphate, sodium iodide, and water. The mixture is then heated to 120°C for approximately 24 hours, allowing the aminocarbonylation to proceed to completion. Following the reaction, the workup is straightforward, involving filtration and silica gel treatment followed by standard column chromatography.
- Charge a sealed reactor with dirhodium tetracarbonyl dichloride, DPPP ligand, tungsten carbonyl, sodium phosphate, sodium iodide, water, the specific nitro compound substrate, and dimethyl carbonate.
- Heat the reaction mixture to a temperature range of 110-130°C (optimally 120°C) and maintain stirring for a duration of 20 to 28 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix the residue with silica gel, and purify the crude product via column chromatography to isolate the high-purity acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this rhodium-catalyzed methodology presents a compelling value proposition centered on cost optimization and risk mitigation. The shift away from hazardous, regulated reagents like dimethyl sulfate towards commodity-grade dimethyl carbonate significantly lowers the barrier to entry for raw material sourcing. Since dimethyl carbonate is produced on a massive industrial scale for other applications, its availability is high and its price point is stable, insulating the manufacturing process from the volatility often seen with specialty fine chemicals. Furthermore, the elimination of stoichiometric bases and the reduction of inorganic waste streams directly correlate to reduced waste disposal costs, a critical factor in maintaining competitive margins in the fine chemical sector.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the dual role of dimethyl carbonate, which eliminates the need to purchase separate solvents and carbon sources, thereby reducing material inventory costs. Additionally, the high efficiency of the rhodium catalyst system means that lower loadings of expensive metal catalysts can be utilized while still achieving excellent conversion rates, optimizing the cost-per-kilogram of the final product. The simplified post-treatment procedure, which avoids complex neutralization steps and extensive aqueous workups, further reduces labor and utility consumption, leading to substantial overall cost savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are commercially available and widely produced, such as nitro compounds and dimethyl carbonate, manufacturers can secure a more resilient supply chain less prone to disruptions. The robustness of the reaction conditions, which tolerate a wide range of substrates without requiring custom-synthesized precursors, allows for greater flexibility in sourcing raw materials from multiple vendors. This diversification of the supply base ensures continuity of supply even if one vendor faces production issues, thereby safeguarding the production schedule and ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with increasingly strict environmental regulations, future-proofing the manufacturing process against potential legislative changes regarding solvent use and waste generation. The absence of toxic gases like carbon monoxide and the use of a biodegradable solvent simplify the safety infrastructure required for scale-up, allowing for smoother technology transfer from pilot plant to commercial production scales. This ease of scalability ensures that the process can readily adapt to fluctuating market demands, supporting both small-batch clinical supply and large-scale commercial manufacturing without the need for significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction conditions and substrate compatibility. Understanding these details is crucial for process engineers evaluating the feasibility of adopting this technology for their specific production needs.
Q: What are the primary advantages of using dimethyl carbonate in this synthesis?
A: Dimethyl carbonate acts as both a green solvent and a C1 source, eliminating the need for toxic methylating agents like diazomethane and reducing inorganic salt waste generation significantly.
Q: What is the functional group tolerance of this rhodium-catalyzed method?
A: The method exhibits broad substrate compatibility, successfully tolerating substituents such as alkoxy, trifluoromethyl, halogens (F, Cl, Br), methyl, thiomethyl, and acetyl groups on the aryl ring.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of commercially available starting materials, mild reaction conditions (120°C), and simple post-treatment procedures makes this route highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the rhodium-mediated aminocarbonylation described in CN112812032A for producing high-quality acetamide derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of acetamide intermediate delivered meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our existing acetamide portfolio or to discuss route feasibility assessments for your custom synthesis needs, ensuring a partnership built on transparency, quality, and mutual success.
