Advanced Manufacturing of Flurbiprofen: A Technical Breakthrough in Green Catalysis and Commercial Scalability
Introduction to Next-Generation Flurbiprofen Manufacturing
The global demand for non-steroidal anti-inflammatory drugs (NSAIDs) continues to drive innovation in the synthesis of key active pharmaceutical ingredients, with Flurbiprofen standing out as a critical therapeutic agent for rheumatic arthritis and osteoarthritis. Recent intellectual property developments, specifically patent CN112225657B, have introduced a transformative preparation method that addresses long-standing inefficiencies in biphenyl fragment construction. This novel approach leverages a palladium-carbon (Pd/C) catalyzed coupling reaction within an aqueous solvent system, marking a significant departure from traditional methods that rely on harsh conditions and toxic reagents. By shifting the paradigm towards green chemistry principles, this technology not only enhances the environmental profile of the manufacturing process but also establishes a robust foundation for high-purity production essential for regulatory compliance. 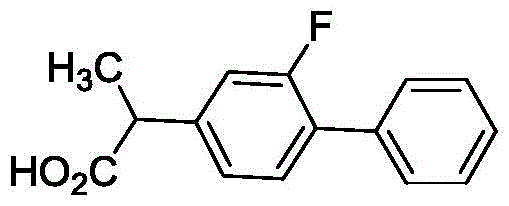
For R&D directors and procurement strategists, understanding the nuances of this patented route is vital for securing a competitive edge in the pharmaceutical intermediate market. The method utilizes 2-(3-fluoro-4-bromophenyl) propionic acid and phenylboronic acid as key precursors, reacting them under alkaline conditions to forge the critical carbon-carbon bond. This strategic selection of starting materials avoids the use of carcinogenic benzene solvents found in legacy processes, thereby mitigating significant health and safety risks associated with industrial scale-up. Furthermore, the ability to recycle the heterogeneous catalyst multiple times without substantial loss of activity presents a compelling economic argument for adoption, promising substantial reductions in raw material costs and waste disposal expenses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Flurbiprofen has been plagued by operational complexities and safety hazards inherent in early patent literature such as US3755427 and US4266069. Traditional routes often necessitated the use of Ullmann reactions at excessively high temperatures ranging from 180°C to 200°C, resulting in low yields and difficult-to-control exothermic profiles that pose risks during commercial production. Moreover, the reliance on benzene as a solvent in certain established protocols introduces severe toxicity concerns, requiring specialized containment equipment and rigorous monitoring to protect personnel and the environment. These legacy methods also frequently employ homogeneous metal ligand catalysts which are not only expensive but also difficult to remove from the final product, leading to potential heavy metal contamination issues that complicate downstream purification and regulatory approval processes.
The Novel Approach
In stark contrast, the methodology disclosed in CN112225657B offers a streamlined and safer alternative by employing a heterogeneous Pd/C catalyst in a predominantly aqueous medium. This innovation effectively eliminates the need for nitrogen protection and complex ligand systems, drastically simplifying the operational requirements and reducing the barrier to entry for large-scale manufacturing. The use of water as a primary solvent not only aligns with green chemistry initiatives but also facilitates easier product isolation through simple pH adjustment and crystallization, bypassing the need for energy-intensive distillation of volatile organic compounds. Additionally, the replacement of toxic brominating agents and carcinogenic solvents with safer alternatives ensures a cleaner impurity profile, directly addressing the quality concerns of R&D teams focused on delivering high-purity API intermediates to the market.
Mechanistic Insights into Pd/C-Catalyzed Suzuki Coupling
The core of this technological advancement lies in the efficient execution of the Suzuki-Miyaura cross-coupling reaction using a heterogeneous palladium catalyst. Unlike homogeneous systems where palladium complexes are dissolved in the reaction medium, the Pd/C catalyst provides a solid surface for the oxidative addition of the aryl bromide and the subsequent transmetallation with the phenylboronic acid species. This heterogeneous nature allows for the facile separation of the catalyst via simple filtration post-reaction, enabling its recovery and reuse for multiple cycles without significant degradation in performance. The mechanistic pathway proceeds through the activation of the carbon-bromine bond on the catalyst surface, followed by the coupling with the boronate complex formed under alkaline conditions, ultimately yielding the biphenyl skeleton of Flurbiprofen with high regioselectivity and minimal side reactions.
From an impurity control perspective, the aqueous alkaline environment plays a crucial role in suppressing the formation of homocoupling byproducts and deboronated species that often plague Suzuki reactions in organic solvents. The specific stoichiometry employed, with a molar ratio of 2-(3-fluoro-4-bromophenyl) propionic acid to phenylboronic acid optimized between 1:1 and 1:1.5, ensures complete conversion of the limiting reagent while minimizing excess reagent waste. Furthermore, the crystallization step utilizing an ethanol-water solvent system exploits the differential solubility of the product versus potential impurities, effectively purging residual palladium and organic byproducts to achieve HPLC purity levels exceeding 99.5%. This rigorous control over the chemical landscape ensures that the final material meets the stringent specifications required for pharmaceutical applications.
How to Synthesize Flurbiprofen Efficiently
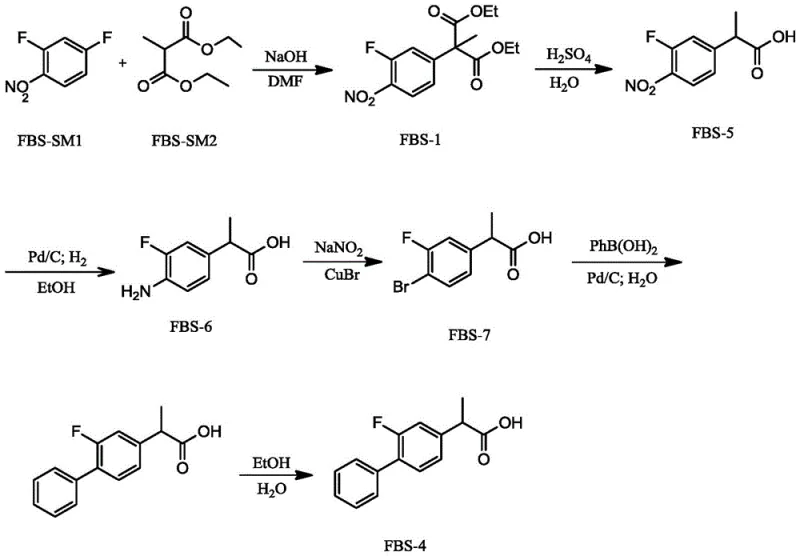
The synthesis protocol outlined in the patent provides a clear roadmap for producing Flurbiprofen with high efficiency and reproducibility. The process begins with the nucleophilic substitution of 2,4-difluoronitrobenzene, followed by a series of functional group transformations including hydrolysis, decarboxylation, and reduction to generate the key bromo-intermediate. The culmination of the sequence is the palladium-catalyzed coupling step, which serves as the pivotal moment for constructing the biaryl framework. Detailed operational parameters regarding temperature, pressure, and reagent ratios are critical for maximizing yield and ensuring consistent quality across batches. For a comprehensive breakdown of the standardized synthetic steps and specific reaction conditions, please refer to the technical guide below.
- Perform nucleophilic substitution of 2,4-difluoronitrobenzene with diethyl methylmalonate under alkaline conditions to form the nitro-intermediate.
- Execute acid hydrolysis and thermal decarboxylation to obtain 3-fluoro-4-nitro-alpha-methyl phenylacetic acid, followed by catalytic hydrogenation to reduce the nitro group.
- Conduct diazotization and bromination to generate the bromo-intermediate, then perform the key Pd/C-catalyzed Suzuki coupling with phenylboronic acid in water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical feasibility. The shift towards a water-based system significantly reduces the dependency on volatile organic solvents, which are subject to fluctuating market prices and increasingly stringent environmental regulations. By minimizing the use of hazardous materials, manufacturers can lower their insurance premiums and waste disposal costs, contributing to a more sustainable and economically viable production model. The robustness of the process also implies a reduction in batch failures and downtime, ensuring a more reliable supply of critical intermediates to downstream API manufacturers who depend on consistent delivery schedules to meet their own production targets.
- Cost Reduction in Manufacturing: The implementation of a recyclable Pd/C catalyst fundamentally alters the cost structure of Flurbiprofen production by eliminating the recurring expense of expensive phosphine ligands and homogeneous palladium salts. Since the catalyst can be recovered and reused for multiple cycles while maintaining high activity, the effective cost per kilogram of catalyst consumption is drastically reduced. Furthermore, the simplified work-up procedure involving filtration and crystallization reduces the need for complex chromatography or extensive solvent exchanges, leading to significant savings in utility costs and labor hours associated with purification.
- Enhanced Supply Chain Reliability: The use of readily available and stable starting materials such as phenylboronic acid and 2,4-difluoronitrobenzene ensures a secure supply chain that is less susceptible to disruptions caused by the scarcity of exotic reagents. The aqueous nature of the reaction medium also simplifies logistics and storage requirements, as there is no need for specialized infrastructure to handle large volumes of flammable or toxic organic solvents. This operational simplicity translates into shorter lead times and greater flexibility in scaling production up or down in response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the heterogeneous catalyst system allowing for easy adaptation to larger reactor volumes without the mass transfer limitations often encountered in homogeneous catalysis. The reduction in toxic waste generation aligns with global trends towards greener manufacturing practices, facilitating easier compliance with environmental permits and reducing the risk of regulatory penalties. This forward-thinking approach not only future-proofs the manufacturing facility against tightening regulations but also enhances the corporate social responsibility profile of the organization.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of this synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the Pd/C catalyst improve cost efficiency compared to traditional ligand systems?
A: The patented process utilizes heterogeneous Pd/C which can be recycled more than 5 times while maintaining yields above 88%, eliminating the need for expensive, single-use phosphine ligands and complex metal scavenging steps.
Q: What represents the primary environmental advantage of this synthesis route?
A: By replacing toxic organic solvents like benzene with a water-based system and avoiding carcinogenic reagents, the process significantly reduces hazardous waste generation and improves operator safety profiles.
Q: What purity levels can be achieved with the described crystallization method?
A: The purification protocol involving ethanol-water crystallization consistently achieves HPLC purity levels exceeding 99.5%, meeting stringent requirements for pharmaceutical active ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flurbiprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering high-purity Flurbiprofen intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the Pd/C catalyzed aqueous coupling process positions us as a strategic partner capable of meeting the evolving needs of the industry.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits applicable to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
