Revolutionizing Difluoromethylation: A Scalable Hypervalent Iodine Strategy for Pharmaceutical Intermediates
Revolutionizing Difluoromethylation: A Scalable Hypervalent Iodine Strategy for Pharmaceutical Intermediates
The landscape of organofluorine chemistry is undergoing a significant transformation, driven by the critical need for efficient methods to introduce fluorine-containing motifs into bioactive molecules. As detailed in the recent patent CN114634482B, a breakthrough has been achieved in the synthesis of novel diazodifluoromethylation reagents, addressing long-standing challenges in the field of pharmaceutical intermediate manufacturing. This technology leverages the unique reactivity of hypervalent iodine species to facilitate the introduction of difluoromethyl groups under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift away from hazardous gaseous reagents and extreme thermal requirements, offering a safer and more controllable pathway for late-stage functionalization. The ability to generate these reactive species at room temperature not only enhances operational safety but also opens new avenues for the synthesis of complex drug candidates where thermal sensitivity is a major concern.
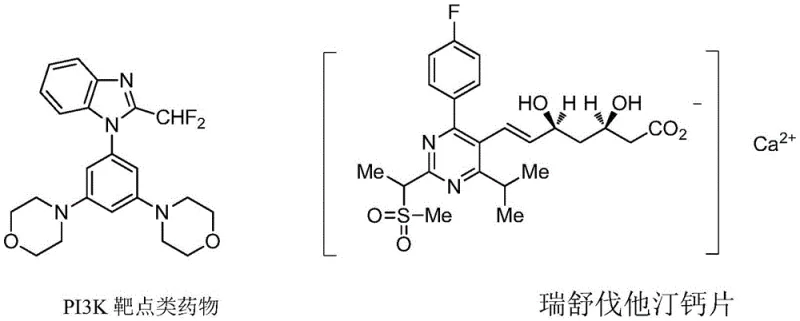
The strategic importance of the difluoromethyl group cannot be overstated in modern medicinal chemistry. As highlighted in the background of the patent, approximately 15-20% of newly approved drugs contain fluorine atoms, with the difluoromethyl moiety playing a pivotal role due to its unique electronic and steric properties. The weak acidity of the hydrogen atom in the difluoromethyl group allows for the formation of hydrogen bonds with electron-rich systems in biological macromolecules, thereby enhancing binding affinity and metabolic stability. This patent provides a robust solution for accessing these valuable structural units, positioning it as a key enabling technology for the development of next-generation therapeutics, including lipid-lowering agents like Rosuvastatin and PI3K inhibitors. By streamlining the synthesis of the reagents themselves, the overall timeline for drug discovery projects can be significantly compressed.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoromethyl groups into organic frameworks has been fraught with significant technical hurdles and safety risks. Traditional methodologies often rely on the use of gaseous reagents such as chlorodifluoromethane (HCF2Cl) or sulfur tetrafluoride, which require specialized handling equipment and strict safety protocols due to their toxicity and volatility. Furthermore, many established protocols necessitate cryogenic conditions, frequently operating at temperatures as low as -40°C to control the high reactivity and poor stability of difluorocarbene intermediates. These harsh conditions impose a heavy burden on manufacturing infrastructure, requiring expensive cooling systems and limiting the scalability of the processes. Additionally, the functional group compatibility of these older methods is often poor, leading to side reactions and reduced yields when applied to complex molecular scaffolds common in pharmaceutical research. The reliance on transition metal catalysts in some variations further complicates the purification process, necessitating rigorous metal scavenging steps to meet regulatory standards for residual impurities in active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN114634482B introduces a highly efficient, one-step synthesis of diazodifluoromethylation reagents using readily available hypervalent iodine precursors. The core innovation lies in the reaction between commercially available hypervalent iodine reagents, such as 1-methoxy-1,2-benzoiodo-3(H)one or iodobenzene diacetate, and the substrate (2-diazo-1,1-difluoroethyl)-sulfonylbenzene (Ps-DFA), activated by trimethylsilyl trifluoromethanesulfonate (TMSOTf). This reaction proceeds smoothly at room temperature under ambient air conditions, eliminating the need for inert atmospheres or cryogenic cooling. The operational simplicity is a major advantage, allowing for rapid screening and scale-up without specialized equipment. The resulting reagents are stable enough to be isolated and stored, yet reactive enough to serve as potent sources of difluoromethyl radicals for downstream transformations. This approach effectively decouples the difficulty of reagent synthesis from the application, providing chemists with a reliable "fluorine block" that can be deployed on demand.
Mechanistic Insights into Hypervalent Iodine-Mediated Fluorination
The mechanistic elegance of this transformation relies on the Lewis acid activation of the hypervalent iodine center by TMSOTf. In the proposed pathway, the trimethylsilyl cation coordinates with the oxygen or acetate ligands on the iodine(III) species, increasing the electrophilicity of the iodine center. This activation facilitates a ligand exchange reaction with the Ps-DFA substrate, displacing the original ligands and forming a new carbon-iodine bond. The resulting hypervalent iodine(III) intermediate is unstable and undergoes reductive elimination or homolytic cleavage to generate the desired diazodifluoromethyl radical species or the stable diazo compound depending on the specific conditions and substituents. The presence of the sulfone group in the Ps-DFA precursor plays a crucial role in stabilizing the adjacent carbanion character during the initial nucleophilic attack, ensuring high regioselectivity. Understanding this mechanism is vital for process optimization, as it highlights the stoichiometric balance required between the iodine source, the activator, and the difluoro-substrate to maximize yield and minimize the formation of iodobenzene byproducts.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed routes. Since the reaction does not involve palladium, copper, or iron catalysts, the risk of heavy metal contamination in the final product is virtually eliminated. This is a critical consideration for pharmaceutical manufacturers who must adhere to strict ICH guidelines regarding elemental impurities. The primary byproducts of this reaction are typically silyl ethers and reduced iodine species, which are generally easier to separate via standard aqueous workups or silica gel chromatography compared to organometallic complexes. Furthermore, the reaction demonstrates excellent functional group tolerance, meaning that sensitive moieties such as esters, amides, or halides present on the aromatic rings of the hypervalent iodine precursor remain intact. This chemoselectivity ensures that the impurity profile of the final reagent is clean and predictable, facilitating faster regulatory approval for downstream drug substances derived from these intermediates.
How to Synthesize Diazodifluoromethylation Reagents Efficiently
The synthesis protocol outlined in the patent is designed for maximum operational ease and reproducibility, making it ideal for both laboratory-scale discovery and pilot-plant production. The process begins with the precise weighing of the hypervalent iodine precursor, such as 1-methoxy-1,2-benzoiodo-3(H)one, and mixing it with TMSOTf and Ps-DFA in a molar ratio of approximately 1:1:2 to 1:1:2.5. The choice of solvent is flexible, with dichloromethane, acetonitrile, or methanol proving effective, though dichloromethane is often preferred for its solubility profile. The reaction mixture is simply stirred at room temperature for 3 to 5 hours, after which the progress can be monitored via TLC or HPLC. Detailed standardized synthesis steps and specific troubleshooting guides for scaling this reaction are provided in the technical documentation below.
- Prepare the reaction mixture by combining commercially available hypervalent iodine reagents (such as 1-methoxy-1,2-benzoiodo-3(H)one or iodobenzene diacetate) with trimethylsilyl trifluoromethanesulfonate (TMSOTf) and the substrate Ps-DFA in an appropriate solvent like dichloromethane.
- Maintain the reaction under air conditions at room temperature, stirring continuously for a duration of 3 to 5 hours to ensure complete conversion of the starting materials into the desired diazo derivatives.
- Upon completion, perform a simplified workup involving silica gel column chromatography using a petroleum ether and ethyl acetate mixture to isolate the high-purity diazodifluoromethylation reagent with yields ranging from 60% to 80%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The shift from cryogenic, gas-phase reactions to a ambient temperature, solution-phase process fundamentally alters the cost structure of manufacturing fluorine-containing intermediates. By removing the dependency on specialized low-temperature reactors and hazardous gas handling infrastructure, capital expenditure for new production lines can be drastically reduced. Furthermore, the use of commercially available starting materials ensures a robust and diversified supply base, mitigating the risk of raw material shortages that often plague niche fluorination chemistries. The simplified workup procedure, which relies on standard column chromatography rather than complex distillation or metal scavenging, translates directly into reduced labor costs and shorter batch cycle times, enhancing overall throughput.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of cryogenic cooling systems result in substantial operational cost savings. The process utilizes commodity chemicals like TMSOTf and simple hypervalent iodine salts, which are produced at scale globally, ensuring stable pricing and preventing cost volatility associated with proprietary reagents. Additionally, the high atom economy of the ligand exchange mechanism minimizes waste generation, reducing the costs associated with solvent recovery and waste disposal. The ability to run the reaction under air conditions further reduces the consumption of inert gases like nitrogen or argon, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly strengthened by the use of non-proprietary, off-the-shelf reagents. Unlike custom-synthesized fluorinating agents that may have long lead times and single-source dependencies, the precursors for this method are widely available from multiple global suppliers. This redundancy ensures business continuity and protects against supply disruptions. The stability of the final diazodifluoromethylation reagents also allows for inventory buffering, enabling manufacturers to produce in larger batches and store for future use without significant degradation, thus smoothing out production schedules and improving responsiveness to customer demand fluctuations.
- Scalability and Environmental Compliance: The benign nature of the reaction conditions facilitates seamless scale-up from gram to kilogram scales without the need for extensive re-optimization. The absence of toxic gases and heavy metals simplifies environmental compliance and permits acquisition, as the effluent streams are easier to treat and monitor. This aligns with the growing industry emphasis on green chemistry principles, allowing companies to reduce their carbon footprint by eliminating energy-intensive cooling steps. The robustness of the process ensures consistent quality across different batch sizes, which is essential for maintaining supply continuity during the transition from clinical trials to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this new diazodifluoromethylation method over traditional techniques?
A: Unlike conventional methods that require harsh gases like HCF2Cl2 or cryogenic temperatures as low as -40°C, this novel approach operates efficiently at room temperature under air conditions, significantly reducing energy consumption and equipment complexity while maintaining high functional group tolerance.
Q: What is the expected yield and purity profile for the synthesized reagents?
A: The patent data indicates that the reaction consistently achieves yields between 60% and 80%. The post-reaction processing is straightforward, utilizing standard column chromatography to deliver high-purity reagents suitable for sensitive downstream nucleophilic reactions.
Q: Can these reagents be applied to complex pharmaceutical scaffolds?
A: Yes, the generated diazodifluoromethylation reagents serve as versatile radical sources capable of introducing difluoromethyl structural units into various organic molecules, including complex heterocycles like 1,3,4-oxadiazoles, which are prevalent in modern drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazodifluoromethylation Reagent Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced fluorination technologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114634482B can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped to handle hypervalent iodine chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the highest quality intermediates for your critical projects. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to delivering solutions that meet your exacting standards.
We invite you to explore how our expertise in fluorine chemistry can optimize your synthesis routes and reduce your time to market. Contact our Customized Cost-Saving Analysis team today to discuss your specific requirements. We encourage potential partners to reach out to our technical procurement team to request specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to bring your next-generation fluorinated therapeutics from concept to commercial success with speed and precision.
