Advanced Hypervalent Iodine Chemistry for Scalable Diazodifluoromethylation Reagent Production
Advanced Hypervalent Iodine Chemistry for Scalable Diazodifluoromethylation Reagent Production
The landscape of organofluorine chemistry is undergoing a significant transformation driven by the critical need for efficient methods to introduce fluorine-containing motifs into bioactive molecules. Patent CN114634482B, published in June 2023, introduces a groundbreaking methodology for the synthesis of novel diazodifluoromethylation reagents, addressing long-standing challenges in the field of pharmaceutical intermediate manufacturing. This technology leverages hypervalent iodine chemistry to generate stable yet reactive difluoromethyl radical precursors under remarkably mild conditions. The strategic importance of the difluoromethyl group cannot be overstated, as its unique electronic properties and ability to form hydrogen bonds significantly enhance the metabolic stability and binding affinity of drug candidates, exemplified by blockbuster medications such as Rosuvastatin Calcium and various PI3K inhibitors. By providing a robust pathway to access these valuable building blocks, this innovation positions itself as a cornerstone for the next generation of fluorinated API development.
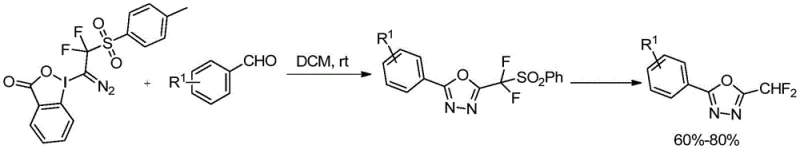
The practical utility of this technology is immediately evident in its application towards synthesizing complex heterocyclic systems. As illustrated in the reaction scheme, the novel reagents facilitate the one-step construction of 1,3,4-oxadiazole derivatives containing difluoromethyl groups directly from commercially available benzaldehydes. This transformation proceeds with impressive efficiency, yielding target compounds in the range of 60% to 80% without the need for exotic catalysts or extreme thermal conditions. For R&D directors and process chemists, this represents a paradigm shift from multi-step, low-yielding sequences to streamlined, high-atom-economy processes. The ability to functionalize diverse aromatic aldehydes with various substituents (R1 = H, F, Cl, Br, Me, OMe, tBu) underscores the broad substrate scope and functional group tolerance of this methodology, making it an indispensable tool for rapid library synthesis and lead optimization campaigns in modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of difluoromethyl groups into organic frameworks has been fraught with significant technical hurdles that impede scalable manufacturing. Traditional approaches often rely on the use of hazardous gaseous reagents such as chlorodifluoromethane (HCF2Cl) or sulfur tetrafluoride, which necessitate specialized high-pressure equipment and rigorous safety protocols to manage toxicity and corrosion risks. Furthermore, many established protocols require cryogenic conditions, typically operating at temperatures as low as -40°C, to control the high reactivity and instability of difluorocarbene or radical intermediates. These stringent thermal requirements not only inflate energy consumption but also limit the feasibility of batch processing in standard glass-lined reactors. Additionally, transition metal-catalyzed cross-coupling methods, while effective, often suffer from poor functional group compatibility and leave behind trace metal impurities that are notoriously difficult to remove to meet stringent pharmaceutical regulatory standards, thereby complicating the downstream purification process.
The Novel Approach
In stark contrast to these legacy methods, the hypervalent iodine-mediated strategy described in the patent offers a transformative solution that prioritizes operational simplicity and safety. By utilizing commercially available hypervalent iodine reagents in conjunction with TMSOTf and the Ps-DFA substrate, the reaction proceeds smoothly at room temperature under ambient air conditions. This elimination of cryogenic needs and inert atmosphere requirements drastically reduces the barrier to entry for implementation in both laboratory and pilot plant settings. The reaction times are equally impressive, typically completing within 3 to 5 hours, which enhances throughput and reduces reactor occupancy time. Moreover, the post-reaction workup is exceptionally straightforward, requiring only simple column chromatography with common solvent systems like petroleum ether and ethyl acetate to isolate high-purity products. This streamlined workflow minimizes solvent waste and labor costs, presenting a compelling case for adoption in cost-sensitive commercial manufacturing environments.
Mechanistic Insights into Hypervalent Iodine-Mediated Difluoromethylation
The core of this technological advancement lies in the unique reactivity of the hypervalent iodine center, which acts as a powerful electrophilic activator for the difluoromethyl diazo species. The mechanism likely involves the initial activation of the hypervalent iodine reagent by trimethylsilyl trifluoromethanesulfonate (TMSOTf), generating a highly reactive cationic iodine species in situ. This activated complex then undergoes a ligand exchange or nucleophilic attack by the diazo carbon of the Ps-DFA precursor, facilitating the transfer of the difluoromethyl diazo group to the iodine center. The resulting hypervalent iodine(III) intermediate is stabilized by the electron-withdrawing sulfonyl and fluoro groups, preventing premature decomposition while retaining sufficient energy to act as a radical precursor upon subsequent activation or thermal input. This delicate balance between stability and reactivity is crucial for the reagent's shelf-life and its controlled release of difluoromethyl radicals during downstream transformations.
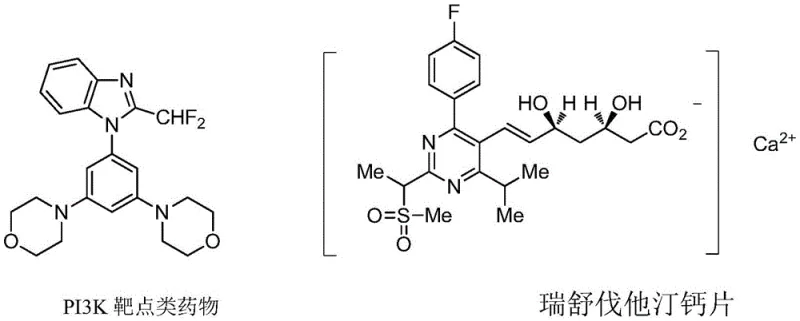
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring product quality. Unlike harsh fluorination methods that can lead to extensive side reactions such as polymerization or non-selective fluorination of sensitive functional groups, this room-temperature protocol preserves the integrity of the molecular scaffold. The use of stoichiometric amounts of reagents (molar ratio 1:1:2 to 1:1:2.5) ensures complete conversion of the valuable hypervalent iodine starting material, minimizing the presence of unreacted precursors in the crude mixture. Furthermore, the byproducts generated during the reaction, primarily derived from the iodine leaving group and silyl species, are generally polar and easily separated from the desired neutral organic product during the aqueous workup and chromatographic purification. This inherent selectivity reduces the burden on analytical teams to identify and quantify complex impurity profiles, accelerating the timeline from synthesis to biological evaluation.
How to Synthesize Diazodifluoromethylation Reagents Efficiently
The synthesis of these high-value reagents is designed to be accessible to any well-equipped organic synthesis laboratory, leveraging off-the-shelf chemicals and standard operating procedures. The process begins with the preparation of the key Ps-DFA precursor, which involves a sequential transformation of p-methylthiophenol through alkylation, reduction, oxidation, and diazotization steps, each optimized for high yield and purity. Once the Ps-DFA is secured, the final assembly of the diazodifluoromethylation reagent is a single-pot operation that requires minimal technical oversight. The detailed standardized synthetic steps, including precise stoichiometry, solvent choices, and isolation techniques, are outlined in the comprehensive guide below to ensure reproducibility and safety during scale-up operations.
- Combine commercially available hypervalent iodine reagents (such as iodobenzene diacetate or 1-methoxy-1,2-benzoiodo-3(H)one) with trimethylsilyl trifluoromethanesulfonate (TMSOTf) in an appropriate solvent like dichloromethane.
- Add the synthesized substrate (2-diazo-1,1-difluoroethyl)-sulfonylbenzene (Ps-DFA) to the reaction mixture under air conditions at room temperature.
- Stir the reaction for 3-5 hours until completion, followed by simple purification via silica gel column chromatography to isolate the pure diazodifluoromethylation reagent derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this hypervalent iodine-based technology offers tangible benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing infrastructure required. By eliminating the need for specialized cryogenic reactors, gas handling systems for toxic fluorinated gases, and expensive transition metal catalysts, the capital expenditure (CAPEX) for setting up production lines is significantly reduced. This lower barrier to entry allows for more flexible manufacturing networks, potentially enabling decentralized production closer to key R&D hubs. Furthermore, the reliance on commercially available starting materials such as iodobenzene diacetate and common solvents like dichloromethane ensures a robust and resilient supply chain, mitigating the risks associated with sourcing obscure or single-source specialty chemicals that often plague complex fluorination projects.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of costly unit operations and consumables. Traditional methods often incur high costs due to the need for continuous low-temperature cooling and the disposal of hazardous metal waste streams. In this new protocol, the absence of transition metals removes the expensive and time-consuming heavy metal scavenging steps typically required to meet ppm-level specifications for API intermediates. Additionally, the high atom economy and simplified workup reduce solvent consumption and waste treatment costs, leading to substantial overall cost savings per kilogram of produced reagent. The ability to run reactions at room temperature also translates to lower energy bills, contributing to a more sustainable and cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any commercial chemical operation, and this methodology excels by utilizing a feedstock base that is deeply integrated into the global chemical market. The hypervalent iodine reagents and sulfone precursors are produced by multiple vendors worldwide, reducing the risk of supply disruptions caused by geopolitical issues or single-vendor bottlenecks. The stability of the final diazodifluoromethylation reagents under ambient conditions further enhances logistics; they can be stored and transported without the need for specialized cold-chain logistics, simplifying inventory management and reducing shipping costs. This reliability ensures that downstream drug development programs remain on schedule, avoiding costly delays associated with material shortages.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but the linear scalability of this room-temperature reaction makes it highly attractive for commercial expansion. The exothermic profile is manageable without aggressive cooling, allowing for larger batch sizes in standard reactors. From an environmental standpoint, the process aligns well with green chemistry principles by avoiding toxic gases and minimizing heavy metal contamination. The waste stream is predominantly organic and iodine-based, which is easier to treat and dispose of compared to the complex mixed waste generated by traditional fluorination methods. This compliance with increasingly stringent environmental regulations future-proofs the manufacturing process against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled a set of frequently asked questions based on the specific technical details and experimental data provided in the patent documentation. These answers address common concerns regarding reaction safety, substrate compatibility, and purification strategies, offering a clear picture of what to expect during process implementation. Understanding these nuances is essential for risk assessment and resource planning prior to initiating pilot studies.
Q: What are the advantages of this hypervalent iodine method over traditional difluoromethylation?
A: Traditional methods often require harsh conditions, toxic gases like HCF2Cl2, or cryogenic temperatures (-40°C). This novel approach operates at room temperature under air, significantly simplifying operational safety and equipment requirements while maintaining high yields of 60%-80%.
Q: Is the Ps-DFA precursor difficult to source for large-scale production?
A: The precursor Ps-DFA is synthesized from readily available starting materials such as p-methylthiophenol and ethyl difluorobromoacetate through a robust multi-step sequence involving oxidation and diazotization, ensuring a reliable supply chain for the final reagent.
Q: What types of chemical structures can be modified using these reagents?
A: These reagents serve as versatile sources of difluoromethyl radicals, enabling the efficient introduction of difluoromethyl groups into various organic molecules, including the synthesis of 1,3,4-oxadiazole derivatives which are critical scaffolds in medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diazodifluoromethylation Reagent Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced fluorination technologies in accelerating the delivery of life-saving medicines to the market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel fluorinated building blocks or the scale-up of established routes, our infrastructure is designed to support your most challenging projects with speed and precision.
We invite you to collaborate with us to unlock the full commercial potential of this hypervalent iodine chemistry for your specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current manufacturing bottlenecks. We encourage you to reach out today to obtain specific COA data for our available fluorinated reagents and to discuss detailed route feasibility assessments that can optimize your supply chain and reduce time-to-market for your critical API candidates.
