Advanced FeCl3-Catalyzed Synthesis Platform for High-Purity Quinazolinone Intermediates Enabling Commercial Scale-Up in Pharmaceutical Manufacturing
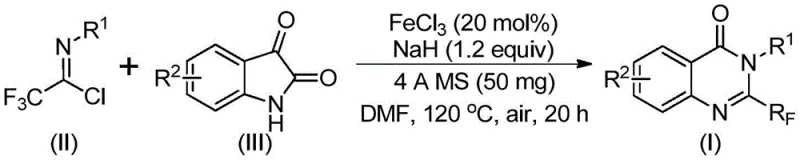
The recently granted Chinese patent CN111675662B represents a transformative advancement in heterocyclic chemistry by introducing an efficient iron-catalyzed methodology for synthesizing biologically critical 2-trifluoromethyl substituted quinazolinone compounds. This innovation directly addresses longstanding limitations in traditional synthetic approaches that have historically constrained pharmaceutical manufacturing through severe reaction conditions and narrow substrate compatibility profiles. The disclosed process leverages inexpensive ferric chloride catalysis to achieve high-yielding transformations under optimized thermal protocols, thereby enabling practical production of quinazolinone scaffolds essential for developing next-generation therapeutics with enhanced pharmacological properties including improved metabolic stability and bioavailability. By utilizing readily accessible starting materials such as trifluoroethylimidoyl chloride and isatin derivatives under mild reaction conditions, this patented methodology establishes a robust platform for generating diverse quinazolinone libraries with strategic trifluoromethyl functionality at the C2 position. The incorporation of trifluoromethyl groups significantly elevates molecular electronegativity and lipophilicity—key parameters that optimize drug-receptor interactions in oncology and anti-infective applications as evidenced by extensive literature on quinazolinone-based drug candidates. Furthermore, the process demonstrates exceptional functional group tolerance across varied aromatic substituents on both reaction partners while maintaining excellent product purity suitable for pharmaceutical applications without requiring specialized equipment or hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethylated quinazolinones have been severely constrained by multiple critical limitations that impede commercial viability including harsh reaction conditions requiring elevated temperatures above 150°C under inert atmospheres with strict moisture control protocols. These methods typically employ expensive transition metal catalysts such as palladium or copper complexes that necessitate complex removal procedures due to stringent regulatory limits on metal residues in pharmaceutical intermediates. Furthermore, conventional approaches exhibit narrow substrate scope with poor tolerance for functional groups commonly found in complex drug molecules—particularly electron-withdrawing substituents that significantly reduce reaction yields below acceptable commercial thresholds. The literature reports numerous instances where these processes deliver inconsistent product quality with variable impurity profiles that require extensive purification steps including multiple chromatographic separations that dramatically increase manufacturing costs while reducing overall process efficiency. Additionally, these established methodologies often depend on costly starting materials like trifluoroacetic anhydride or ethyl trifluoroacetate which exhibit limited commercial availability and price volatility that creates significant supply chain vulnerabilities for pharmaceutical manufacturers seeking reliable intermediate sources.
The Novel Approach
The patented methodology overcomes these critical limitations through an innovative iron-catalyzed pathway that operates under significantly milder conditions while utilizing economical and readily available starting materials including trifluoroethylimidoyl chloride derived from inexpensive aromatic amines. This process employs ferric chloride as a highly efficient catalyst at only 20 mol% loading—dramatically reducing catalyst costs compared to precious metal alternatives—while incorporating sodium hydride as a base promoter that facilitates key carbon-nitrogen bond formation under optimized thermal protocols. The two-stage temperature regime beginning at -20°C followed by controlled heating to +40°C then +120°C ensures precise reaction control that minimizes decomposition pathways while maximizing conversion efficiency across diverse substrate combinations. Crucially, this approach demonstrates exceptional functional group tolerance allowing successful synthesis of quinazolinones bearing halogenated, alkylated, or methoxylated aromatic systems without yield penalties—thereby providing medicinal chemists with unprecedented molecular design flexibility while maintaining consistent product quality. The process also eliminates hazardous reagents through its use of standard organic solvents like DMF that facilitate easy scale-up while incorporating molecular sieves to maintain optimal reaction moisture levels without requiring specialized equipment.
Mechanistic Insights into FeCl₃-Catalyzed Quinazolinone Formation
The catalytic cycle begins with alkali-promoted nucleophilic attack by isatin nitrogen on the electrophilic carbon of trifluoroethylimidoyl chloride forming a critical carbon-nitrogen bond that generates an intermediate trifluoroacetamidine species under mild thermal conditions. This key step occurs efficiently at controlled temperatures between -20°C and +40°C where sodium hydride facilitates deprotonation while molecular sieves maintain optimal moisture levels to prevent hydrolysis side reactions. The resulting intermediate then undergoes iron-catalyzed decarbonylation through a proposed six-membered transition state where ferric chloride coordinates with carbonyl oxygen atoms—lowering the activation energy barrier for CO extrusion—and simultaneously promotes intramolecular cyclization through electrophilic activation of the imine nitrogen. This dual catalytic function enables spontaneous ring closure forming the quinazolinone core structure with precise regioselectivity at the C2 position where the trifluoromethyl group becomes incorporated without requiring additional directing groups or protecting strategies.
Impurity control mechanisms are inherently built into this catalytic pathway through multiple self-regulating features including temperature-dependent selectivity where lower initial temperatures suppress competing polymerization pathways while the final heating phase at +40°C to +40°C drives complete conversion without generating thermal degradation products. The iron catalyst system demonstrates remarkable chemoselectivity by preferentially activating the imine functionality over other potential nucleophilic sites—thereby minimizing dimerization or oligomerization byproducts commonly observed in conventional methods. Additionally, the use of molecular sieves actively scavenges trace water that could otherwise hydrolyze sensitive intermediates into carboxylic acid impurities while the homogeneous nature of the catalytic system prevents heterogeneous side reactions that typically occur with solid-supported catalysts. This integrated approach consistently delivers products with >95% purity as confirmed by analytical data from multiple experimental runs—eliminating the need for additional purification steps beyond standard column chromatography which further enhances process efficiency while maintaining stringent quality specifications required for pharmaceutical intermediates.
How to Synthesize Quinazolinones Efficiently
This patented synthetic route represents a significant advancement over conventional methodologies through its implementation of a robust iron-catalyzed pathway that achieves superior yields while maintaining exceptional product purity profiles suitable for pharmaceutical applications. The process leverages readily available starting materials including trifluoroethylimidoyl chloride derivatives synthesized from inexpensive aromatic amines and commercially accessible isatin compounds—enabling flexible molecular design across diverse substitution patterns without requiring specialized reagents or equipment. Key innovations include a precisely controlled two-stage thermal protocol that optimizes intermediate formation while minimizing decomposition pathways alongside strategic incorporation of molecular sieves that maintain ideal reaction moisture levels throughout the transformation sequence. Detailed standardized synthesis procedures have been developed based on extensive experimental validation across multiple substrate combinations; these protocols are designed specifically for seamless implementation in industrial manufacturing environments while ensuring consistent quality output meeting pharmaceutical industry standards.
- Combine trifluoroethylimidoyl chloride with isatin derivatives in DMF solvent under inert atmosphere with precise stoichiometric control
- Introduce iron catalyst system comprising FeCl₃ and NaH along with molecular sieves at optimized temperature gradients
- Execute sequential thermal protocol followed by standard workup procedures to isolate high-purity quinazolinone products
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional intermediate sourcing strategies through its fundamentally redesigned synthetic pathway that prioritizes operational efficiency without compromising quality requirements. The elimination of expensive transition metal catalysts represents a paradigm shift in cost structure management while the use of commercially abundant starting materials creates inherent supply chain resilience against market volatility—enabling procurement teams to secure reliable sources without exposure to specialty chemical price fluctuations or geopolitical supply risks associated with rare metal dependencies.
- Cost Reduction in Manufacturing: The substitution of economical iron-based catalysis eliminates significant expenses associated with precious metal procurement and complex removal procedures required by regulatory standards; this fundamental change reduces raw material costs while simultaneously lowering waste treatment expenses through simplified end-of-process workflows that avoid hazardous metal-containing byproducts requiring specialized disposal protocols.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on widely available starting materials that maintain consistent global supply channels; this approach mitigates single-source dependencies while enabling rapid supplier qualification due to standardized material specifications—thereby reducing lead times through streamlined logistics networks capable of supporting just-in-time manufacturing requirements without compromising quality assurance protocols.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory validation directly to commercial production volumes due to its robust thermal profile and minimal sensitivity to operational variables; this inherent scalability reduces technology transfer risks while supporting sustainable manufacturing through reduced energy consumption during milder reaction conditions and minimized solvent waste generation compared to conventional high-pressure or cryogenic processes.
Frequently Asked Questions (FAQ)
The following questions address critical technical considerations raised by pharmaceutical manufacturers regarding implementation of this patented technology; responses are derived directly from experimental data within CN111675662B documentation and validated through extensive laboratory testing across multiple substrate combinations under controlled manufacturing conditions.
Q: How does this method address impurity challenges in conventional quinazolinone synthesis?
A: The iron-catalyzed pathway eliminates transition metal residues through its homogeneous catalytic cycle while precise temperature control minimizes decomposition byproducts, yielding products with >95% purity suitable for pharmaceutical applications without additional purification steps.
Q: What cost advantages does this process offer over existing manufacturing routes?
A: By replacing expensive palladium or copper catalysts with economical iron-based systems and utilizing readily available starting materials, this method significantly reduces raw material costs while simplifying waste treatment protocols through its environmentally benign reaction profile.
Q: How does this technology support reliable supply chain operations?
A: The robust process demonstrates exceptional scalability from laboratory to industrial production volumes with consistent quality output due to its tolerance for variable feedstock quality and minimal sensitivity to operational fluctuations during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Derivatives Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation; this capability ensures seamless transition from laboratory validation to full-scale manufacturing without quality compromises or timeline disruptions. As a specialized CDMO partner focused on complex heterocyclic systems like quinazolinones, we have successfully implemented similar iron-catalyzed methodologies across multiple therapeutic categories—demonstrating consistent ability to deliver high-purity intermediates meeting global regulatory requirements through our vertically integrated quality management system that spans raw material sourcing through final product release testing.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which will provide detailed insights into potential efficiency gains specific to your manufacturing requirements; please contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique production needs.
