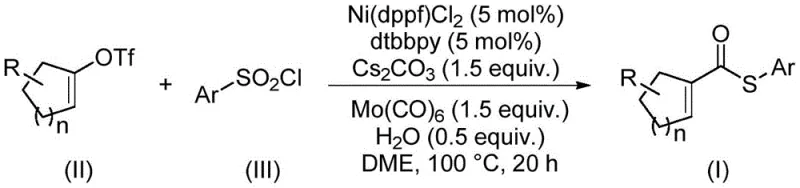
Advanced Nickel-Catalyzed Synthesis of Alpha Beta Unsaturated Thioesters for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex molecular architectures, particularly those containing sulfur motifs which are prevalent in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN114773242A, which discloses a highly efficient preparation method for α,β-unsaturated thioester compounds. These compounds serve as critical building blocks in organic synthesis, participating in pivotal transformations such as Diels-Alder reactions, conjugate additions, and cascade cyclizations. The patented technology addresses long-standing challenges in thioester synthesis by replacing expensive precious metal catalysts with a cost-effective nickel-based system and eliminating the use of malodorous thiols. For R&D directors and procurement strategists, this innovation represents a paradigm shift towards more sustainable and economically viable manufacturing processes for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α,β-unsaturated thioesters has relied heavily on condensation reactions or transition metal-catalyzed thiocarbonylation using precious metals such as rhodium, platinum, and palladium. While these traditional methods often exhibit good catalytic efficiency, they suffer from severe economic and operational drawbacks that hinder large-scale adoption. The primary constraint is the exorbitant cost of precious metal catalysts, which significantly inflates the raw material expenditure for bulk production. Furthermore, conventional thiocarbonylation protocols frequently necessitate the use of thiols as the sulfur source. Thiols are characterized by their extremely unpleasant and penetrating odors, posing significant occupational health and safety challenges in a plant environment. Additionally, thiols have a strong propensity to coordinate with and poison transition metal catalysts, leading to reduced turnover numbers and inconsistent reaction outcomes. These factors collectively create a bottleneck for the commercial scale-up of complex polymer additives or agrochemical intermediates that require thioester functionalities.
The Novel Approach
The methodology outlined in the patent data introduces a transformative alternative that circumvents these legacy issues through a clever redesign of the catalytic system and reagent selection. By utilizing nickel, an earth-abundant and inexpensive base metal, the process drastically reduces the catalyst cost burden compared to palladium or rhodium systems. Crucially, the invention employs arylsulfonyl chlorides as the sulfur source instead of thiols. This substitution not only eliminates the foul odors associated with thiol handling but also prevents catalyst poisoning, thereby enhancing the overall reaction efficiency and reproducibility. Moreover, the process utilizes molybdenum carbonyl [Mo(CO)6] which serves a dual function as both the carbonyl source and the reducing agent. This dual role simplifies the reagent list and avoids the need for handling high-pressure carbon monoxide gas cylinders, which are typically required for carbonylation reactions and pose significant safety risks. The result is a streamlined, safer, and more cost-effective pathway for producing high-purity OLED materials or specialty chemicals.
Mechanistic Insights into Nickel-Catalyzed Thiocarbonylation
The core of this technological advancement lies in the intricate interplay between the nickel catalyst and the unique reagent combination. The reaction initiates with the oxidative addition of the alkenyl trifluoromethanesulfonate to the active nickel(0) species generated in situ. Unlike traditional carbonylations where gaseous CO is used, here the carbonyl moiety is delivered via the decomposition of molybdenum hexacarbonyl under thermal conditions. This controlled release of CO prevents the formation of inactive and highly toxic nickel tetracarbonyl [Ni(CO)4] species, which is a common pitfall in nickel-catalyzed carbonylations that leads to catalyst deactivation. The arylsulfonyl chloride then participates in the cycle, likely undergoing reduction to generate the necessary sulfur nucleophile or interacting directly with the acyl-nickel intermediate. The presence of water and cesium carbonate plays a vital role in facilitating the reduction of the sulfonyl chloride and neutralizing acidic byproducts, ensuring the catalytic cycle proceeds smoothly without stalling.
From an impurity control perspective, this mechanism offers distinct advantages for manufacturing high-purity electronic chemical or pharmaceutical intermediates. The use of dtbbpy (4,4'-di-tert-butyl-2,2'-bipyridine) as a ligand stabilizes the nickel center and enhances its selectivity, minimizing side reactions such as homocoupling of the alkenyl substrate. The broad functional group tolerance observed in the experimental data—accommodating halogens, alkyl chains, and ether groups—suggests that the catalytic cycle is robust against steric and electronic variations. This robustness is critical for maintaining a clean impurity profile, as it reduces the formation of difficult-to-remove byproducts that often plague complex syntheses. Consequently, the downstream purification burden is lessened, allowing for higher overall yields and purity levels essential for regulatory compliance in drug substance manufacturing.
How to Synthesize Alpha,Beta-Unsaturated Thioesters Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to reduce lead time for high-purity pharmaceutical intermediates. The protocol involves charging a sealed tube with the nickel catalyst, ligand, molybdenum carbonyl, base, and substrates in a glyme solvent, followed by heating at 100°C for 20 hours. This single-pot operation eliminates the need for multiple isolation steps or hazardous gas handling infrastructure. The detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures derived from the patent examples, are provided below to facilitate immediate technology transfer and pilot plant evaluation.
- Combine nickel catalyst (Ni(dppf)Cl2), ligand (dtbbpy), molybdenum carbonyl, cesium carbonate, water, alkenyl trifluoromethanesulfonate, and arylsulfonyl chloride in a sealed tube.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture thoroughly to ensure homogeneity.
- Heat the reaction mixture to 100°C and maintain this temperature for 20 hours to allow the thiocarbonylation to proceed to completion.
- Upon completion, filter the reaction mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target thioester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed protocol offers substantial strategic benefits beyond mere technical feasibility. The shift from precious metals to base metals fundamentally alters the cost structure of the synthesis, decoupling production economics from the volatile pricing of rhodium and palladium markets. Furthermore, the replacement of thiols with stable, odorless arylsulfonyl chlorides significantly improves the working environment and reduces the costs associated with specialized scrubbing systems and personal protective equipment. The use of commercially available starting materials ensures a reliable supply chain, mitigating the risk of raw material shortages that can disrupt production schedules for critical API intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive precious metal catalysts with nickel results in a direct and significant decrease in catalyst expenditure. Additionally, the use of molybdenum carbonyl as a solid CO source removes the need for high-pressure gas infrastructure and the associated safety compliance costs. The simplified workup procedure, involving basic filtration and chromatography, further reduces processing time and solvent consumption, leading to substantial cost savings in the overall manufacturing budget without compromising yield.
- Enhanced Supply Chain Reliability: The starting materials, including alkenyl trifluoromethanesulfonates and arylsulfonyl chlorides, are widely available from global chemical suppliers, ensuring a stable and continuous supply stream. Unlike specialized thiols which may have limited vendors and long lead times, these reagents are commodity chemicals. This availability allows for flexible sourcing strategies and reduces the risk of supply chain bottlenecks, ensuring consistent delivery of the final thioester products to downstream customers.
- Scalability and Environmental Compliance: The reaction conditions are mild and utilize standard laboratory equipment that translates easily to industrial reactors. The absence of toxic gases and foul-smelling reagents simplifies waste treatment and emissions control, aligning with increasingly stringent environmental regulations. The high atom economy and efficient conversion rates minimize waste generation, supporting green chemistry initiatives and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed thiocarbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the process capabilities and limitations for potential adopters.
Q: Why is nickel preferred over palladium or rhodium for this thiocarbonylation?
A: Nickel is an abundant and inexpensive base metal compared to precious metals like palladium, rhodium, or platinum. While precious metals show high activity, their cost limits large-scale industrial application. Furthermore, this specific nickel system avoids the formation of toxic Ni(CO)4 species often associated with nickel carbonylations by using molybdenum carbonyl as a controlled CO source.
Q: What are the safety advantages of using arylsulfonyl chloride over thiols?
A: Traditional thioester synthesis often relies on thiols, which are notorious for their unpleasant, pervasive odors and tendency to poison transition metal catalysts. Using arylsulfonyl chloride as the sulfur source eliminates these olfactory hazards and prevents catalyst deactivation, leading to a cleaner reaction profile and easier handling in a manufacturing environment.
Q: Can this method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates broad substrate scope and excellent functional group tolerance. It successfully accommodates various substituents on the aryl ring, including alkyl groups (methyl, isopropyl, tert-butyl), halogens (chlorine), and electron-donating or withdrawing groups (methoxy, trifluoromethoxy), making it highly versatile for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed synthesis route for the production of high-value sulfur-containing intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha,beta-unsaturated thioester delivered meets the highest quality standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this novel route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline while maximizing cost efficiency.
