Advanced Palladium-Catalyzed Carbonylation for Commercial Scale-up of Complex 2,3-Dihydroquinolone Intermediates
Advanced Palladium-Catalyzed Carbonylation for Commercial Scale-up of Complex 2,3-Dihydroquinolone Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies to access nitrogen-containing heterocycles, particularly the 2,3-dihydroquinolone scaffold, which serves as a critical core structure in numerous bioactive molecules. As disclosed in patent CN112239456B, a novel preparation method has been developed that utilizes a transition metal palladium-catalyzed carbonylation reaction to efficiently construct these valuable skeletons. This technology represents a significant leap forward in synthetic organic chemistry, addressing long-standing challenges regarding safety and substrate tolerance. The biological relevance of this scaffold cannot be overstated, as derivatives such as antitumor agent A, antitumor agent B, and analgesic agent C demonstrate potent activities against human cancer cells and pain pathways, highlighting the urgent need for reliable production methods.
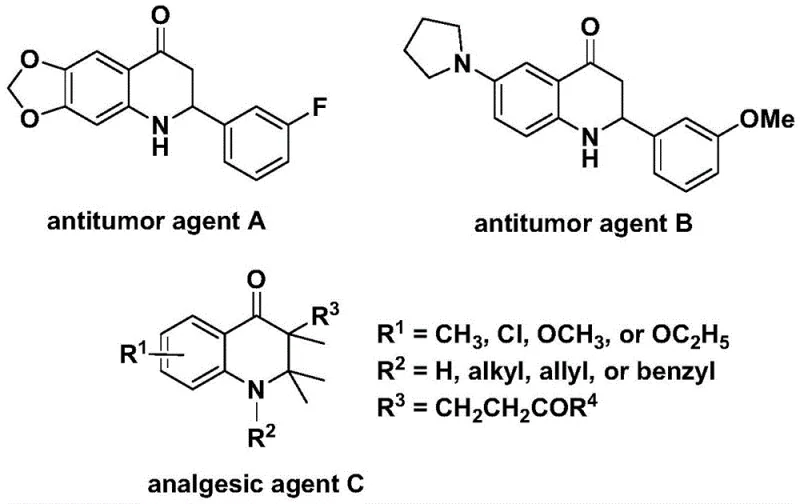
For procurement managers and supply chain directors, the implications of this patent extend far beyond the laboratory bench. The ability to synthesize 2-aryl and 3-alkyl substituted 2,3-dihydroquinolone compounds through rational substrate design opens up vast possibilities for library generation and process optimization. By leveraging this technology, a reliable pharmaceutical intermediate supplier can offer clients a pathway to complex molecules that was previously hindered by the logistical nightmares of handling gaseous carbon monoxide. The method described herein not only simplifies the operational workflow but also ensures that the initial raw materials are cheap and easy to obtain, which is a fundamental requirement for cost-effective manufacturing in the competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has relied heavily on traditional carbonylation reactions that utilize carbon monoxide gas directly. While chemically effective, these conventional methods present severe drawbacks for industrial application, primarily centered around safety and infrastructure costs. Handling high-pressure cylinders of toxic CO gas requires specialized equipment, rigorous safety protocols, and often limits the reaction to facilities with specific hazard ratings, thereby constraining supply chain flexibility. Furthermore, the direct use of gaseous CO can lead to uncontrolled reaction rates, resulting in the formation of polymeric byproducts and difficult-to-remove impurities that compromise the purity profile of the final active pharmaceutical ingredient. These factors collectively drive up the cost of goods sold and introduce significant risks to production continuity.
The Novel Approach
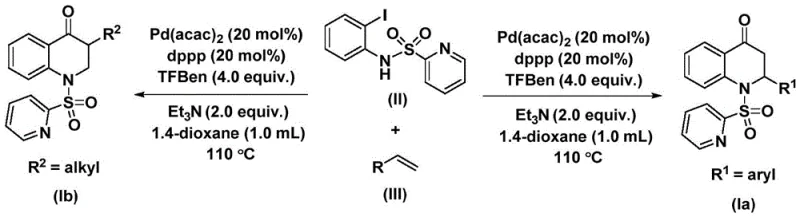
The innovative approach detailed in the patent data overcomes these barriers by employing 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide surrogate. This strategic substitution transforms a hazardous gas-phase reaction into a manageable liquid-phase process, drastically reducing the safety burden on the manufacturing facility. The reaction proceeds under relatively mild thermal conditions, typically around 110 °C, using a palladium catalyst system that exhibits remarkable tolerance for diverse functional groups. As illustrated in the reaction scheme, the process allows for the seamless integration of various olefin substrates, ranging from electron-rich aryl alkenes to simple alkyl chains, without the need for protecting group strategies. This versatility ensures that a reliable agrochemical intermediate supplier or pharma partner can rapidly adapt the process to produce a wide array of structural analogues.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific targets. The catalytic cycle initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is facilitated by the bidentate phosphine ligand, 1,3-bis(diphenylphosphino)propane (dppp), which stabilizes the metal center and promotes the subsequent migratory insertion steps. The presence of the pyridine sulfonyl group on the nitrogen atom plays a dual role: it acts as a directing group to facilitate cyclization and serves as a leaving group precursor, although in this specific carbonylative manifold, it primarily modulates the electronic properties of the aniline nitrogen to favor the desired reactivity pattern.
Following the formation of the aryl-palladium species, the carbon monoxide molecule, released in situ from the thermal decomposition of the trimesic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This acyl species then undergoes coordination and migratory insertion with the olefin substrate, creating a new carbon-carbon bond and extending the molecular framework. The cycle concludes with an intramolecular nucleophilic attack or reductive elimination step that closes the six-membered ring, releasing the substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst. This elegant mechanism ensures high atom economy and minimizes waste, aligning with modern green chemistry principles while delivering high-purity intermediates suitable for downstream drug development.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical execution of this synthesis is designed to be straightforward, requiring standard laboratory glassware and commonly available reagents, which facilitates easy technology transfer from R&D to pilot plant operations. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, iodoaniline derivative, and olefin in an aprotic solvent such as 1,4-dioxane. The mixture is then heated to promote the catalytic cycle, after which standard workup procedures involving filtration and silica gel chromatography yield the pure product. For detailed operational parameters and specific stoichiometric ratios required to achieve optimal yields across different substrate classes, please refer to the standardized synthesis steps outlined below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and the specific olefin substrate in 1,4-dioxane solvent within a Schlenk tube.
- Heat the reaction mixture to 110 °C and maintain stirring for a duration of 48 hours to ensure complete conversion of the starting materials into the target dihydroquinolone scaffold.
- Upon completion, filter the reaction mixture, load the crude residue onto silica gel, and perform column chromatography purification to isolate the high-purity substituted 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly address the pain points of procurement managers and supply chain heads. The shift from gaseous CO to a solid surrogate eliminates the need for expensive high-pressure reactors and specialized gas handling infrastructure, leading to significant capital expenditure savings and reduced operational complexity. This simplification allows for greater flexibility in manufacturing site selection and reduces the regulatory burden associated with hazardous material storage. Furthermore, the use of commercially available starting materials, such as palladium bis(acetylacetonate) and various olefins, ensures a stable and resilient supply chain that is less susceptible to geopolitical disruptions or raw material shortages.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling requirements translates directly into lower operational costs, as facilities do not need to invest in specialized scrubbing systems or high-pressure containment units. Additionally, the high reaction efficiency and broad substrate scope minimize the need for extensive purification steps, reducing solvent consumption and waste disposal costs. By streamlining the synthetic route, manufacturers can achieve a more favorable cost structure, making the final intermediates more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like 1,3,5-trimesic acid phenol ester enhances supply chain security by removing the logistical challenges associated with transporting and storing compressed gases. This stability ensures consistent production schedules and reduces the risk of unplanned downtime due to supply interruptions. Moreover, the robustness of the catalytic system means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply of critical pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work effectively from gram-scale experiments to potential multi-kilogram production runs. The use of a closed system with a solid CO source significantly reduces the environmental footprint by preventing the release of volatile organic compounds and toxic gases into the atmosphere. This alignment with environmental, social, and governance (ESG) goals makes the process attractive for companies looking to reduce their carbon footprint and adhere to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method, providing clarity for technical teams evaluating its adoption. These answers are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, ensuring accuracy and relevance for process development scientists.
Q: What is the primary advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: The use of 1,3,5-trimesic acid phenol ester serves as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous gaseous CO, thereby significantly enhancing operational safety and simplifying reactor requirements for industrial scale-up.
Q: What types of olefin substrates are compatible with this palladium-catalyzed protocol?
A: The method demonstrates excellent substrate compatibility, successfully accommodating both substituted aryl olefins containing groups like methyl, chloro, fluoro, and tert-butyl, as well as various alkyl olefins including linear, branched, and silyl-substituted variants.
Q: How does this method improve impurity control compared to traditional carbonylation?
A: By utilizing a controlled release of carbon monoxide from a solid surrogate rather than high-pressure gas, the reaction kinetics are moderated, which minimizes side reactions and polymerization of the olefin, leading to a cleaner crude profile and easier downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality heterocyclic building blocks for the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from discovery to market. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2,3-dihydroquinolone intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced carbonylation technology for your specific drug development programs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how this efficient synthesis method can accelerate your timeline and optimize your budget for high-purity pharmaceutical intermediates.
