Advanced Pd-Catalyzed Synthesis of Polysubstituted Ketene Imines for Commercial Scale-Up
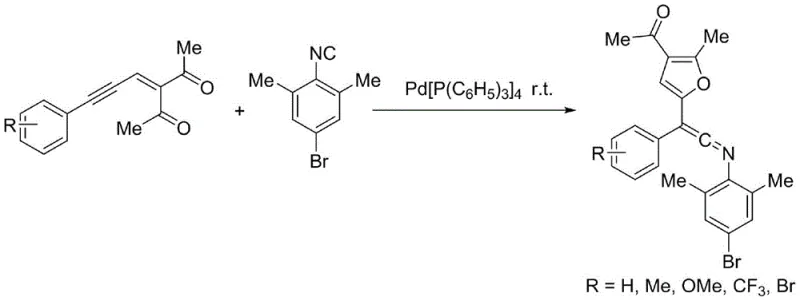
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular scaffolds efficiently. Patent CN110590717B introduces a groundbreaking approach to synthesizing polysubstituted ketene imines, a class of compounds renowned for their high reactivity in nucleophilic additions and cycloadditions. This technology leverages a palladium-catalyzed insertion reaction between 4-bromo-2,6-dimethylphenyl isonitrile and specific enynone derivatives. Unlike conventional methods that often struggle with harsh conditions or poor atom economy, this novel route operates under remarkably mild parameters, typically at room temperature in tetrahydrofuran. For R&D directors and procurement managers alike, this represents a significant opportunity to access high-purity pharmaceutical intermediates with streamlined processing. The ability to generate complex structures containing furan rings opens new avenues for synthesizing bioactive molecules targeting microbial infections and cardiovascular conditions, positioning this technology as a cornerstone for modern medicinal chemistry pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
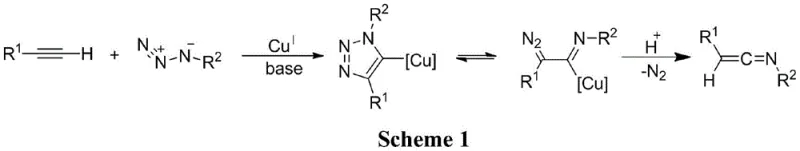
Historically, the construction of polysubstituted ketene imine skeletons has relied on methodologies that present significant operational challenges for industrial scale-up. For instance, earlier literature describes copper-catalyzed reactions involving sulfonyl azides and terminal alkynes, which often necessitate strict control over reaction parameters and can generate sulfonated byproducts that complicate downstream purification. Other approaches involving the dehydrohalogenation of chloroimines require strong bases and can suffer from limited substrate scope or stability issues with the resulting intermediates. Furthermore, many traditional pathways involve multi-step sequences with moderate yields, leading to increased waste generation and higher overall production costs. These limitations often result in extended lead times for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for drug manufacturers who require reliable access to complex building blocks for total synthesis projects.
The Novel Approach
The methodology disclosed in patent CN110590717B offers a transformative solution by utilizing a palladium-catalyzed migratory insertion strategy. This approach activates the alkynyl triple bond of the enynone substrate, facilitating an intramolecular nucleophilic attack by the carbonyl oxygen. This initial cyclization generates an active resonance-stabilized intermediate that subsequently undergoes insertion with the isonitrile component. The result is the direct formation of complex polysubstituted ketene imines bearing a furan moiety. This route is characterized by its exceptional atom economy and operational simplicity, eliminating the need for hazardous reagents or extreme temperatures. By merging the formation of the heterocyclic ring and the ketene imine functionality in a single catalytic cycle, this method drastically simplifies the synthetic workflow, offering a clear pathway for cost reduction in fine chemical manufacturing while ensuring the structural integrity required for downstream biological applications.
Mechanistic Insights into Palladium-Catalyzed Cyclization and Insertion
The core of this technological advancement lies in the unique activation mode of the palladium catalyst. Upon coordination with the electron-rich alkyne of the enynone substrate, the palladium center lowers the energy barrier for the subsequent intramolecular nucleophilic attack by the proximal carbonyl oxygen. This step is critical as it forms the furan ring system, a structural motif highly valued in medicinal chemistry for its metabolic stability and hydrogen-bonding capabilities. The resulting organopalladium species is resonance-stabilized, preventing premature decomposition and allowing for the precise insertion of the isonitrile molecule. This insertion step is regioselective, ensuring that the nitrogen atom of the isonitrile is correctly positioned to form the ketene imine double bond. For technical teams, understanding this mechanism is vital for optimizing reaction conditions, as the stability of the palladium intermediate directly correlates with the observed yields, which can reach up to 89% in optimized examples. This mechanistic clarity allows for rational tuning of ligand environments to further enhance efficiency.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature processes. The mild reaction conditions minimize the formation of thermal degradation products or polymerization byproducts that often plague ketene imine synthesis. The specificity of the palladium insertion ensures that side reactions, such as homocoupling of the isonitrile or oligomerization of the enynone, are significantly suppressed. This high level of chemoselectivity translates to a cleaner crude reaction profile, reducing the burden on purification units. For quality assurance teams, this means that achieving stringent purity specifications is more attainable with fewer recrystallization or chromatography steps. The ability to tolerate various functional groups on the aromatic rings of the substrates further demonstrates the robustness of this catalytic cycle, making it adaptable for synthesizing a diverse library of analogues for structure-activity relationship studies.
How to Synthesize Polysubstituted Ketene Imine Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and safety. The process begins with the precise weighing of the 4-bromo-2,6-dimethylphenyl isonitrile and the enynone derivative, which are then dissolved in anhydrous tetrahydrofuran. The addition of the tetrakis(triphenylphosphine)palladium catalyst initiates the reaction immediately upon stirring at room temperature. Monitoring the reaction progress via thin-layer chromatography (TLC) is essential to determine the optimal endpoint, preventing over-reaction or catalyst decomposition. Once the starting materials are consumed, the solvent is removed under reduced pressure, and the residue is purified using standard column chromatography techniques. Detailed standardized synthesis steps for this procedure are provided in the guide below.
- Dissolve 4-bromo-2,6-dimethylphenyl isonitrile, enynone, and tetrakis(triphenylphosphine)palladium catalyst in tetrahydrofuran (THF) solvent.
- Maintain the reaction mixture at room temperature and stir until TLC analysis indicates complete consumption of starting materials.
- Remove the solvent under vacuum and purify the resulting concentrate via column chromatography using a PE: EA (50:1) eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed technology offers substantial strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the need for cryogenic conditions or high-pressure reactors, facilities can utilize existing general-purpose equipment, thereby lowering capital investment requirements. Furthermore, the use of common solvents like THF and commercially available catalysts ensures a stable supply chain for raw materials, mitigating the risk of production delays caused by specialty reagent shortages. This reliability is crucial for maintaining continuous production schedules for high-purity pharmaceutical intermediates, ensuring that downstream drug development timelines are met without interruption.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by its high atom economy and mild operating conditions. Traditional methods often require stoichiometric amounts of expensive activators or generate significant waste streams that incur disposal costs. In contrast, this catalytic process utilizes substoichiometric amounts of palladium, and the high yields observed (up to 89%) mean less raw material is wasted per kilogram of product. Additionally, the simplified workup procedure reduces the consumption of silica gel and solvents during purification. These factors combine to create a leaner manufacturing process that significantly lowers the cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Because the reaction proceeds efficiently at room temperature, it is less susceptible to fluctuations in utility supplies, such as steam or chilled water, which can disrupt more sensitive processes. The substrates involved, such as substituted phenyl isonitriles and enynones, are derived from readily available commodity chemicals, reducing dependency on single-source suppliers. This diversification of the raw material base ensures that production can be scaled up rapidly to meet surging demand without the long lead times associated with custom synthesis of exotic starting materials. Consequently, partners can rely on a consistent flow of material, supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by its exothermic profile, which is manageable under standard cooling conditions. The absence of hazardous gases or highly toxic reagents simplifies compliance with environmental, health, and safety (EHS) regulations. The high selectivity of the reaction minimizes the generation of complex waste mixtures, making waste treatment more straightforward and cost-effective. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the manufacturing process against tightening regulatory standards. For companies aiming to enhance their sustainability credentials, adopting this efficient synthetic route demonstrates a commitment to responsible chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows. The answers provided reflect the current state of the art as defined by the intellectual property, ensuring accuracy and relevance for decision-making processes.
Q: What are the key advantages of this Pd-catalyzed method over traditional Cu-catalyzed routes?
A: Unlike traditional Cu-catalyzed methods which often require harsh conditions or produce sulfonated byproducts, this Pd-catalyzed insertion operates at room temperature with excellent atom economy and generates a furan-containing scaffold valuable for drug synthesis.
Q: What is the structural significance of the resulting ketene imine products?
A: The products feature a polysubstituted ketene imine backbone with a furan ring, serving as critical intermediates for synthesizing bioactive molecules used in treating microbial infections, ulcers, and arrhythmias.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction utilizes mild conditions (room temperature), common solvents (THF), and avoids expensive or toxic reagents, making it highly scalable for commercial production of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Ketene Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced intermediates like polysubstituted ketene imines play in the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of material delivered meets the exacting standards required for pharmaceutical applications. We understand that consistency and quality are non-negotiable in the life sciences sector, and our integrated approach to process development ensures that potential scale-up issues are identified and resolved early.
We invite you to collaborate with us to leverage this innovative palladium-catalyzed technology for your specific drug discovery programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only reliable and compliant but also driven by a commitment to scientific excellence and commercial value.
