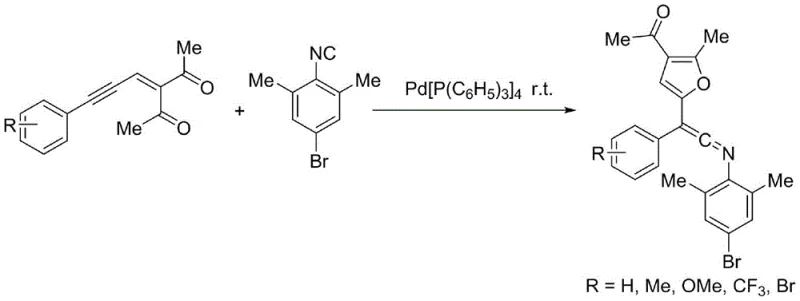
Advanced Palladium-Catalyzed Synthesis of Polysubstituted Ketene Imines for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex molecular scaffolds with high efficiency and minimal environmental impact. Patent CN110590717B introduces a groundbreaking approach for the synthesis of polysubstituted ketene imines, a class of compounds renowned for their high reactivity in nucleophilic additions and cycloadditions. These structures serve as pivotal intermediates in the total synthesis of natural products and therapeutic agents, such as the calcium channel blocker (S)-Verapamil. The disclosed technology leverages a palladium-catalyzed insertion reaction between 4-bromo-2,6-dimethylphenyl isonitrile and enynones, offering a pathway that is not only operationally simple but also exhibits exceptional atom economy. By activating the alkynyl triple bond through palladium coordination, the method facilitates an intramolecular nucleophilic attack by the carbonyl oxygen, followed by cyclization and subsequent isonitrile insertion. This sequence allows for the construction of highly functionalized architectures under remarkably mild conditions, specifically at room temperature in tetrahydrofuran, addressing the critical industry demand for sustainable and scalable synthetic solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of ketene imine skeletons has relied on methodologies that often present significant logistical and economic challenges for industrial application. For instance, earlier strategies developed by researchers such as Prof. Chang utilized copper-catalyzed reactions between sulfonyl azides and terminal alkynes, which frequently necessitate strict control over reaction parameters and can suffer from issues related to catalyst cost and stability. Other approaches, such as those reported by Katagiri involving the dehydrohalogenation of chloroimines, often require strong bases and generate stoichiometric amounts of salt waste, complicating downstream purification and waste management. Furthermore, palladium-catalyzed migratory insertions involving alpha-halo phosphonates, while effective, may involve substrates that are less readily available or require multi-step preparation, thereby increasing the overall cost of goods. These conventional routes often struggle with balancing reactivity and selectivity, sometimes requiring elevated temperatures or extended reaction times that degrade sensitive functional groups, ultimately limiting their utility in the synthesis of complex, multifunctional drug candidates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN110590717B offers a streamlined and elegant solution that bypasses many of these traditional bottlenecks. The core innovation lies in the direct coupling of readily accessible enynones with isonitriles using a tetrakis(triphenylphosphine)palladium catalyst system. This novel pathway proceeds through a unique mechanism where the palladium center activates the alkyne moiety, triggering a cascade that forms a resonance-stabilized intermediate capable of undergoing efficient insertion. The result is the rapid assembly of polysubstituted ketene imines bearing diverse functional groups, including furan rings, which are valuable motifs in medicinal chemistry. The reaction conditions are exceptionally benign, occurring at room temperature without the need for exotic ligands or harsh additives. This simplicity translates directly into operational ease, allowing for rapid reaction monitoring via thin-layer chromatography and straightforward isolation of the target products through standard column chromatography techniques.
Mechanistic Insights into Palladium-Catalyzed Cyclization and Insertion
The mechanistic elegance of this transformation provides R&D teams with a deep understanding of how complex molecular complexity is generated from simple precursors. The cycle initiates with the oxidative addition or coordination of the zero-valent palladium species to the electron-rich alkynyl triple bond of the enynone substrate. This activation significantly enhances the electrophilicity of the alkyne, facilitating an intramolecular nucleophilic attack by the proximal carbonyl oxygen atom. This step is crucial as it drives the formation of a cyclic oxonium or similar activated intermediate, which serves as the reactive species for the subsequent step. The presence of the isonitrile then allows for a migratory insertion event, where the carbon-nitrogen triple bond of the isonitrile inserts into the metal-carbon or metal-oxygen bond, effectively expanding the molecular framework. This sequence culminates in the formation of the stable ketene imine product, regenerating the active palladium catalyst to continue the cycle. Understanding this detailed pathway is essential for optimizing reaction parameters and predicting the compatibility of various substituents on the starting materials.
From an impurity control perspective, the specificity of this palladium-mediated mechanism offers significant advantages over non-catalytic or radical-based alternatives. Because the reaction proceeds through a well-defined organometallic cycle, side reactions such as polymerization of the enynone or uncontrolled oligomerization of the isonitrile are minimized. The mild room temperature conditions further suppress thermal degradation pathways that often lead to the formation of tarry byproducts in high-temperature syntheses. Additionally, the use of triphenylphosphine ligands helps stabilize the palladium center, preventing the formation of palladium black which can sequester the catalyst and reduce yield. The high atom economy of the process means that nearly all atoms from the starting materials are incorporated into the final product, reducing the burden on purification systems and ensuring a cleaner crude reaction profile. This level of control is paramount for pharmaceutical applications where strict limits on impurities and heavy metal residues must be maintained.
How to Synthesize Polysubstituted Ketene Imines Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale discovery and pilot-scale production. The protocol utilizes common laboratory solvents and reagents, eliminating the need for specialized equipment beyond standard pressure tubes and rotary evaporators. The reaction is typically conducted by dissolving the enynone and the 4-bromo-2,6-dimethylphenyl isonitrile in tetrahydrofuran, followed by the addition of the palladium catalyst. The mixture is then stirred at ambient temperature, allowing the transformation to proceed to completion as monitored by standard analytical techniques. Upon completion, the solvent is removed under reduced pressure, and the residue is purified using silica gel chromatography. This straightforward workflow minimizes the technical barriers to adoption, enabling process chemists to rapidly generate libraries of novel ketene imine derivatives for biological evaluation or further synthetic elaboration.
- Dissolve 4-bromo-2,6-dimethylphenyl isonitrile, enynone substrate, and tetrakis(triphenylphosphine)palladium catalyst in tetrahydrofuran solvent at a optimized molar ratio.
- Stir the reaction mixture at room temperature in a sealed pressure tube, monitoring progress via TLC until the starting materials are fully consumed.
- Remove the solvent under vacuum conditions and purify the resulting concentrate using column chromatography with a petroleum ether and ethyl acetate eluent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology presents a compelling value proposition centered on cost efficiency and supply reliability. The elimination of cryogenic cooling systems and the ability to run reactions at room temperature significantly reduces the energy consumption associated with the manufacturing process. Furthermore, the use of commercially available starting materials such as enynones and substituted isonitriles ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized precursors. The high atom economy and clean reaction profile reduce the volume of solvent and silica required for purification, leading to substantial savings in waste disposal costs and raw material usage. These factors collectively contribute to a lower cost of goods sold (COGS), making the final intermediates more competitive in the global market.
- Cost Reduction in Manufacturing: The operational simplicity of this process drives significant cost optimization by removing the need for expensive specialized ligands or extreme reaction conditions. By utilizing a robust palladium catalyst system that functions efficiently at room temperature, the process eliminates the capital and operational expenditures associated with heating or cooling infrastructure. The high yields observed across various substrates mean that less raw material is wasted, directly improving the material balance and reducing the cost per kilogram of the active intermediate. Additionally, the straightforward workup procedure minimizes labor hours and processing time, further enhancing the overall economic viability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like tetrahydrofuran and triphenylphosphine ensures that production schedules are not disrupted by the degradation of sensitive catalysts or reagents. The broad substrate scope demonstrated in the patent indicates that the process is tolerant to various functional groups, allowing for flexibility in sourcing different substituted enynones without needing to re-optimize the entire process. This flexibility is crucial for maintaining continuity of supply in the face of fluctuating raw material markets. The scalability of the reaction from milligram to multi-kilogram scales without loss of efficiency provides confidence that supply demands can be met consistently as project needs evolve from clinical trials to commercial launch.
- Scalability and Environmental Compliance: The green chemistry metrics of this process align well with modern environmental, health, and safety (EHS) standards. The absence of toxic heavy metal salts or hazardous oxidants simplifies the effluent treatment process, reducing the environmental footprint of the manufacturing site. The ability to conduct the reaction in a closed pressure tube at ambient temperature enhances operator safety by minimizing exposure to volatile solvents or exothermic events. As regulatory pressures on chemical manufacturing intensify, adopting a process with inherent safety and environmental advantages positions the supply chain for long-term compliance and sustainability, avoiding potential future liabilities associated with older, dirtier technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and benefits of the technology. Understanding these details is essential for stakeholders evaluating the integration of this method into their existing production workflows or R&D pipelines.
Q: What are the primary advantages of this Pd-catalyzed method over traditional Cu-catalyzed routes?
A: This method operates at room temperature with significantly shorter reaction times and avoids the use of harsh bases or cryogenic conditions often required in copper-catalyzed azide-alkyne cycloadditions, leading to improved safety and energy efficiency.
Q: What is the structural complexity achievable with this synthetic route?
A: The process efficiently constructs polysubstituted ketene imines containing five distinct functional groups, including furan moieties, which are critical scaffolds for bioactive molecules and chiral ligands.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the use of common solvents like THF, ambient temperature conditions, and straightforward workup procedures involving rotary evaporation and column chromatography makes this route highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Ketene Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110590717B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted ketene imine delivered meets the highest international standards for pharmaceutical intermediates. We are committed to leveraging our technical expertise to optimize this palladium-catalyzed route for your specific needs, ensuring maximum yield and minimal impurity profiles.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating exactly how this technology can improve your bottom line. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us be your trusted partner in delivering high-quality chemical solutions that drive your business forward.
