Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
Introduction to Next-Generation Amide Synthesis
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries, particularly regarding the construction of robust amide bonds. A significant breakthrough in this domain is documented in Chinese Patent CN111978194B, which discloses a highly efficient preparation method for aryl acetamide compounds. This technology represents a paradigm shift from traditional amidation strategies by utilizing a palladium-catalyzed carbonylation approach that leverages benzyl formate as both a carbon monoxide source and a reactant. For R&D directors and process chemists, this methodology offers a compelling alternative to hazardous gas handling, providing a streamlined pathway to complex molecular architectures. The ability to utilize tertiary amines directly as amine sources, coupled with the avoidance of external oxidants, underscores the practical utility and green chemistry credentials of this invention. As a leading manufacturer, we recognize the immense potential of this route to enhance the production of high-purity pharmaceutical intermediates while mitigating operational risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamide compounds has relied heavily on the direct amidation of phenylacetic acid and its derivatives, a process that often necessitates harsh reaction conditions and generates significant stoichiometric waste. Alternatively, transition metal-catalyzed carbonylation reactions have emerged as a powerful tool; however, conventional protocols frequently depend on primary or secondary amines, leaving the activation of tertiary amines as a formidable challenge due to the strength of their C-N bonds. Furthermore, existing methods for tertiary amine carbonylation often require the use of explosive carbon monoxide and oxygen mixtures or excessive amounts of oxidants, creating severe safety hazards and complicating the purification process. These limitations not only increase the cost of goods sold but also pose substantial barriers to commercial scale-up, particularly for facilities lacking specialized high-pressure infrastructure. The reliance on unstable reagents and the generation of complex impurity profiles further strain quality control resources, making these traditional routes less attractive for modern, compliant manufacturing environments.
The Novel Approach
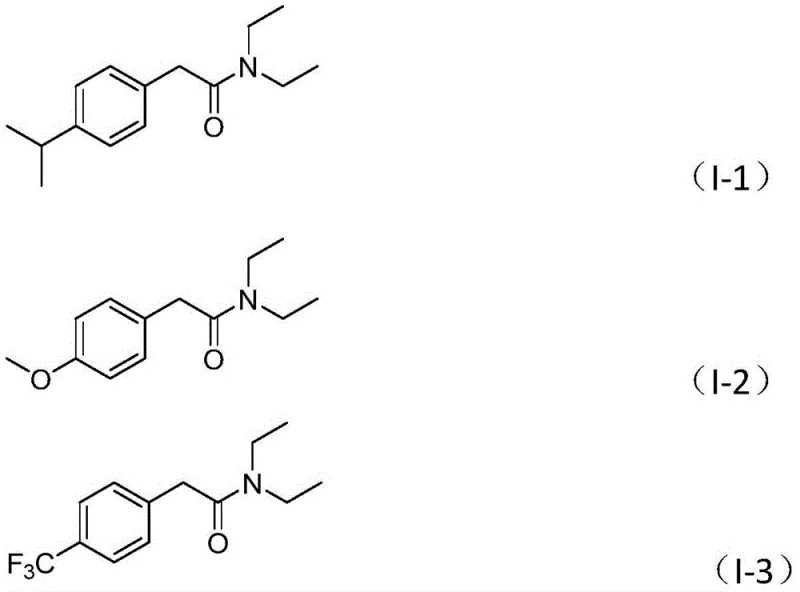
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a sophisticated palladium-catalyzed system that operates under relatively mild and safe conditions. By employing benzyl formate as a liquid carbon monoxide surrogate, the process eliminates the need for handling toxic CO gas, thereby drastically improving workplace safety and reducing equipment costs. The reaction proceeds efficiently at 130°C in acetonitrile, utilizing triethylamine or similar tertiary amines as the nitrogen source without requiring additional oxidants for C-N bond cleavage. This oxidative carbonylation strategy is remarkably tolerant of various functional groups, including halogens, alkyls, and electron-withdrawing groups like trifluoromethyl and cyano moieties. The versatility of this method allows for the synthesis of a diverse array of aryl acetamide derivatives, as evidenced by the successful preparation of compounds I-1 through I-5 with high yields.
The generality of this transformation is further highlighted by the broad substrate scope, accommodating substituted phenyl rings, naphthyl, furyl, and thienyl groups. This flexibility is crucial for medicinal chemists seeking to rapidly iterate on lead compounds or for process teams optimizing final API intermediates. The use of commercially available and inexpensive starting materials, such as formic acid-derived benzyl formate and common tertiary amines, ensures that the raw material costs remain low, facilitating a more economical production model.
Mechanistic Insights into Pd-Catalyzed Carbonylation
The mechanistic underpinning of this transformation involves a carefully orchestrated catalytic cycle driven by palladium species, likely initiated by the in situ generation of a reactive Pd(0) complex from the Pd(OAc)2 precursor and the DPEphos ligand. The benzyl formate acts as a dual-purpose reagent, undergoing decarbonylation or decomposition to release carbon monoxide in a controlled manner within the reaction vessel, which then coordinates to the palladium center. Simultaneously, the tertiary amine undergoes a unique C-N bond activation step, a process that is typically energetically demanding but is facilitated here by the specific electronic environment created by the catalyst system and the presence of trifluoroacetic anhydride (TFAA). The TFAA likely serves as a dehydrating agent or activator, promoting the formation of the acyl-palladium intermediate without the need for external oxidants, which distinguishes this mechanism from traditional oxidative carbonylations. This intricate interplay between the catalyst, ligand, and additives ensures high turnover numbers and minimizes side reactions, resulting in the clean formation of the desired amide bond.
Impurity control is inherently built into this mechanism due to the high selectivity of the palladium catalyst for the specific C-H and C-N activation sites required for the transformation. The absence of aggressive oxidants reduces the likelihood of over-oxidation byproducts, while the use of a homogeneous catalyst system allows for precise tuning of reaction parameters to suppress dimerization or polymerization side reactions. For R&D teams, understanding this mechanism provides a roadmap for troubleshooting and optimization, ensuring that the process remains robust even when scaling to larger batches. The compatibility with sensitive functional groups implies that protecting group strategies can often be minimized, further streamlining the synthetic route and improving overall atom economy.
How to Synthesize Aryl Acetamide Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and thermal profiles to maximize yield and purity. The protocol involves charging a sealed tube or reactor with the palladium catalyst, the bidentate phosphine ligand, the benzyl formate substrate, the tertiary amine, and the trifluoroacetic anhydride additive in an aprotic solvent like acetonitrile. Maintaining the reaction temperature at 130°C for a duration of 24 hours is critical to drive the equilibrium towards product formation while ensuring complete consumption of the starting materials. Following the reaction period, the workup procedure is straightforward, involving filtration to remove any insoluble palladium residues, followed by silica gel treatment and column chromatography to isolate the pure aryl acetamide. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety compliance.
- Combine palladium catalyst (Pd(OAc)2), ligand (DPEphos), benzyl formate, tertiary amine, and trifluoroacetic anhydride (TFAA) in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C and maintain stirring for 24 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target aryl acetamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages that directly impact the bottom line and operational resilience. By shifting away from gaseous carbon monoxide and hazardous oxidants, manufacturers can significantly reduce capital expenditure on specialized high-pressure reactors and safety containment systems, leading to substantial cost savings in facility maintenance and insurance. The reliance on bench-stable, liquid reagents like benzyl formate simplifies logistics and storage, mitigating the risks associated with the transport and handling of compressed gases. Furthermore, the high functional group tolerance means that a single platform technology can be applied to synthesize a wide library of intermediates, reducing the need for multiple, disparate synthetic routes and consolidating the supply chain for diverse product portfolios.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reagents, combined with the use of cheap, commodity chemicals like benzyl formate and triethylamine, drives down the raw material cost per kilogram of product. Additionally, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps, contributing to a leaner manufacturing process. The high yields reported, ranging up to 87%, minimize material loss and waste disposal costs, enhancing the overall economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: Since all key reagents are commercially available and stable at room temperature, the risk of supply disruptions due to hazardous material shipping restrictions is markedly reduced. This stability ensures a consistent flow of materials, allowing for better production planning and shorter lead times for delivering high-purity pharmaceutical intermediates to clients. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, further securing the supply chain against upstream fluctuations.
- Scalability and Environmental Compliance: The absence of toxic gas emissions and the reduced need for stoichiometric oxidants align perfectly with modern environmental regulations and sustainability goals. Scaling this reaction from gram to ton scale is facilitated by the homogeneous nature of the catalysis and the manageable thermal profile, avoiding the exothermic runaways often seen in traditional oxidation reactions. This ease of scale-up ensures that commercial production can be ramped up quickly to meet market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the detailed experimental data and beneficial effects outlined in the patent documentation. Understanding these aspects helps stakeholders make informed decisions about adopting this technology for their specific manufacturing needs. The answers reflect the practical realities of running this reaction in a GMP-compliant environment.
Q: What is the primary advantage of using benzyl formate over carbon monoxide gas?
A: Benzyl formate serves as a safe, liquid surrogate for carbon monoxide, eliminating the need for high-pressure gas equipment and reducing safety risks associated with toxic CO handling in industrial settings.
Q: Does this method require additional oxidants for tertiary amine activation?
A: No, a key innovation of this patent is that the C-N bond cleavage of the tertiary amine occurs without the need for external oxidants, simplifying the reaction system and reducing waste.
Q: What is the typical yield range for this synthesis?
A: According to the patent data, the reaction demonstrates high efficiency with isolated yields ranging from 71% to 87% across various substituted aryl substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced technologies like the one described in CN111978194B to deliver superior chemical solutions to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of aryl acetamide meets the highest industry standards for pharmaceutical applications. Our state-of-the-art facilities are equipped to handle complex catalytic processes safely, providing you with a reliable source of high-quality intermediates.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this method for your supply chain. We are ready to provide specific COA data and route feasibility assessments to support your R&D and production goals, ensuring a partnership built on transparency, quality, and mutual success.
