Advanced Silver-Catalyzed Synthesis of Isocoumarin Derivatives for Scalable Pharmaceutical Manufacturing
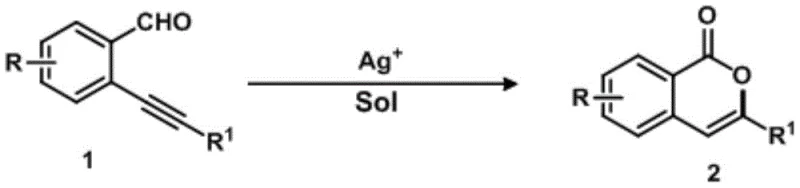
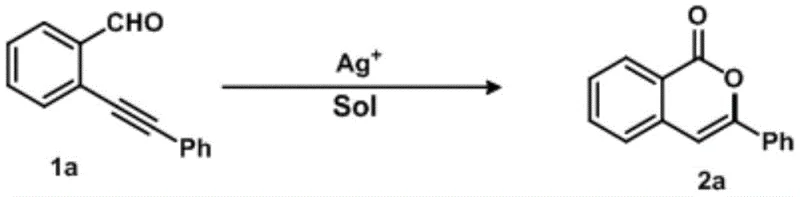
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN112778261B, which discloses a highly efficient method for synthesizing isocoumarin derivatives through silver catalysis. Isocoumarins are privileged structures found in numerous natural products and therapeutic agents, exhibiting diverse biological activities ranging from anti-ulcer and anti-allergic properties to potential treatments for Alzheimer's disease. However, traditional synthetic routes have often been plagued by limitations such as harsh reaction conditions, poor regioselectivity, and the generation of toxic waste. The technology described in this patent addresses these critical pain points by introducing a mild, one-step cyclization strategy using 2-(phenylethynyl)benzaldehyde as a model substrate. This innovation represents a paradigm shift for manufacturers looking to optimize their production lines for high-purity pharmaceutical intermediates, offering a pathway that balances chemical efficiency with economic viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isocoumarin scaffold has relied heavily on transition metal-catalyzed intramolecular cyclizations of alkynyl-substituted carboxylic acids or esters. While effective in academic settings, these conventional strategies present substantial hurdles for industrial application. A primary concern is the reliance on expensive and sometimes scarce transition metal catalysts that drive up the overall cost of goods sold (COGS). Furthermore, many existing protocols necessitate the use of strong oxidants to facilitate the cyclization process, which not only introduces safety hazards regarding thermal runaway but also generates significant amounts of toxic by-products that complicate downstream purification. Another critical bottleneck is the availability of starting materials; traditional routes often require multi-step synthesis to prepare the necessary precursors, leading to cumulative yield losses and extended lead times. Additionally, issues with regioselectivity frequently result in complex product mixtures, requiring resource-intensive chromatographic separations that are difficult to translate from gram-scale laboratory experiments to ton-scale commercial production.
The Novel Approach
In stark contrast to these legacy methods, the silver-catalyzed protocol outlined in the patent data offers a streamlined and economically attractive alternative. By utilizing readily available 2-(phenylethynyl)benzaldehyde derivatives as direct starting materials, the process eliminates the need for pre-functionalization steps, thereby drastically simplifying the synthetic sequence. The core of this innovation lies in the use of monovalent silver salts, such as silver tetrafluoroborate (AgBF4), which act as efficient π-acid catalysts to activate the alkyne moiety under remarkably mild conditions. The reaction proceeds smoothly at temperatures as low as 30°C to 60°C, avoiding the energy-intensive heating required by older methodologies. This approach not only enhances the safety profile of the manufacturing process but also significantly reduces the formation of degradation products, resulting in cleaner reaction profiles. The versatility of this method is further demonstrated by its compatibility with a wide range of solvents, including toluene and DMF, allowing process chemists to select conditions that optimize both yield and environmental impact.
Mechanistic Insights into Silver-Catalyzed Cyclization
The efficacy of this synthetic route is rooted in the unique electronic properties of the monovalent silver cation (Ag+). In the proposed mechanistic pathway, the silver ion coordinates with the triple bond of the phenylethynyl group, increasing its electrophilicity and facilitating a nucleophilic attack by the carbonyl oxygen of the aldehyde group. This interaction triggers a 6-endo-dig cyclization, a process that is kinetically favored under the specific reaction conditions provided. Unlike gold or platinum catalysts which might be overly reactive or difficult to remove from the final product due to strict residual metal limits in APIs, silver salts offer a balanced reactivity profile. The mild Lewis acidity of Ag+ is sufficient to promote the ring closure without inducing unwanted side reactions such as polymerization or over-oxidation. Furthermore, the use of non-coordinating anions like tetrafluoroborate (BF4-) ensures that the silver center remains accessible for substrate activation throughout the catalytic cycle. This precise control over the reaction mechanism allows for high conversion rates, with experimental data showing yields reaching up to 88% when optimized conditions are employed.
From an impurity control perspective, the mildness of the silver-catalyzed system is a distinct advantage for R&D teams focused on quality by design (QbD). Harsh oxidative conditions often lead to the formation of radical species that can attack the aromatic rings or the newly formed lactone structure, generating hard-to-remove impurities. By operating at moderate temperatures (e.g., 40°C) and avoiding strong oxidants, this method minimizes thermal stress on the molecule. The result is a crude reaction mixture with a significantly simplified impurity profile, which facilitates easier purification via standard crystallization or chromatography techniques. This level of purity is essential for pharmaceutical intermediates intended for subsequent coupling reactions or biological testing, where trace contaminants can skew results or compromise the safety of the final drug substance. The robustness of the catalyst system also means that it tolerates various electronic environments on the aromatic rings, maintaining consistent performance across a broad substrate scope.
How to Synthesize Isocoumarin Derivatives Efficiently
Implementing this silver-catalyzed methodology in a production setting requires careful attention to reaction parameters to maximize yield and reproducibility. The patent provides clear guidance on the optimal stoichiometry and conditions, highlighting that a molar ratio of substrate to catalyst around 10:1 is effective for driving the reaction to completion. The choice of solvent plays a pivotal role, with toluene emerging as a preferred medium due to its ability to dissolve the organic substrates while maintaining a favorable boiling point for the reaction temperature window. The process is designed to be operationally simple, involving the dissolution of the starting material, addition of the catalyst, and a controlled heating period followed by a standard aqueous workup. This simplicity makes it an ideal candidate for technology transfer from R&D to pilot plant and eventually to full-scale manufacturing.
- Dissolve 2-(phenylethynyl)benzaldehyde substrate in a suitable solvent such as toluene or DMF.
- Add a monovalent silver salt catalyst, preferably AgBF4, to the reaction mixture at a molar ratio of approximately 10: 1.
- Heat the mixture to 40°C for 6 hours, then perform standard aqueous workup and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silver-catalyzed synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Since the method utilizes 2-(phenylethynyl)benzaldehydes, which are themselves accessible via standard Sonogashira couplings of commercially abundant iodobenzaldehydes and terminal alkynes, the dependency on exotic or custom-synthesized precursors is eliminated. This accessibility translates directly into enhanced supply security and reduced vulnerability to market fluctuations for niche starting materials. Moreover, the elimination of strong oxidants and toxic reagents simplifies the waste management infrastructure required at the manufacturing site, leading to substantial cost savings in environmental compliance and hazardous waste disposal. The ability to run the reaction at near-ambient temperatures also reduces the energy load on the production facility, contributing to a lower carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The economic impact of switching to this silver-catalyzed protocol is profound, primarily driven by the reduction in process steps and reagent costs. Traditional methods often require expensive palladium or gold catalysts, whereas silver salts like AgBF4 are significantly more cost-effective and widely available. Furthermore, the high atom economy of this one-step cyclization means that less material is wasted during the transformation, improving the overall mass balance of the process. By avoiding the need for protective group strategies or multi-step functionalization sequences often seen in older routes, manufacturers can achieve a leaner production workflow. This streamlining reduces labor hours, reactor occupancy time, and solvent consumption, all of which are key drivers in the final cost per kilogram of the active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of this synthetic method. The tolerance of the reaction to various substituents on the aromatic rings means that a single standardized protocol can be used to produce a diverse library of isocoumarin derivatives. This flexibility allows manufacturers to respond quickly to changing demands for different analogues without needing to revalidate entirely new processes for each variation. Additionally, the use of common solvents like toluene and DMF ensures that there are no bottlenecks related to specialized solvent availability. The mild reaction conditions also reduce the risk of batch failures due to thermal excursions, ensuring a more consistent and reliable output of high-quality material that meets strict delivery schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, but this silver-catalyzed route is inherently scalable. The exotherm of the reaction is manageable due to the mild temperatures involved, reducing the need for complex cooling systems that can be limiting factors in large reactors. From an environmental standpoint, the absence of heavy metal oxidants and the potential for silver recovery align with modern green chemistry principles. This compliance with stringent environmental regulations minimizes the risk of regulatory delays and enhances the company's reputation as a sustainable manufacturer. The simplified purification process further reduces the volume of silica gel and solvents required for chromatography, lowering the overall environmental burden of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this silver-catalyzed synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering practical guidance for process development teams evaluating this route for commercial adoption. Understanding these nuances is critical for ensuring successful technology transfer and maximizing the yield and purity of the final isocoumarin products.
Q: What is the optimal catalyst for this isocoumarin synthesis?
A: According to patent CN112778261B, silver tetrafluoroborate (AgBF4) is the preferred catalyst, yielding up to 88% conversion in toluene, outperforming silver nitrate and silver carbonate.
Q: Can this method tolerate various substituents on the benzaldehyde ring?
A: Yes, the method demonstrates excellent functional group tolerance, successfully processing substrates with electron-withdrawing groups like fluorine and electron-donating groups like methyl without significant yield loss.
Q: Why is this silver-catalyzed route superior to traditional methods?
A: Traditional methods often require harsh oxidants or complex multi-step substrate preparation. This novel approach utilizes readily available starting materials under mild conditions (30-60°C) without toxic by-products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isocoumarin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the silver-mediated cyclization described in CN112778261B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle sensitive silver-catalyzed reactions with precision, maintaining stringent purity specifications through our rigorous QC labs. We understand that consistency and quality are paramount for our global clients, and our dedicated technical team is prepared to optimize every parameter of this synthesis to meet your specific project requirements.
We invite you to collaborate with us to leverage this cutting-edge methodology for your next drug development program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific silver-catalyzed route can optimize your budget without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data for our isocoumarin portfolio and to discuss detailed route feasibility assessments tailored to your unique molecular targets. Let us help you accelerate your timeline to market with a reliable, scalable, and cost-effective supply solution.
