Advanced Rhodium-Catalyzed Synthesis of Isocoumarin Derivatives for Commercial Pharmaceutical Manufacturing
Advanced Rhodium-Catalyzed Synthesis of Isocoumarin Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access biologically active scaffolds, and the recent disclosure in patent CN112625020B presents a significant advancement in this domain. This patent details a novel methodology for the preparation of isocoumarin derivatives, a class of compounds renowned for their diverse physiological activities including antibacterial, anti-inflammatory, and anticancer properties. The core innovation lies in the utilization of a Rhodium(III)-catalyzed carbon-hydrogen (C-H) bond activation strategy, which fundamentally alters the synthetic approach to these valuable heterocyclic systems. By leveraging sulfoxide sulfur ylides as carbene precursors and oxazoline moieties as intrinsic directing groups, this technology offers a streamlined route that bypasses many of the limitations associated with traditional multistep syntheses. For procurement and R&D leaders, understanding the mechanistic depth and operational simplicity of this patent is crucial for evaluating its potential impact on supply chain resilience and cost structures in the manufacturing of high-purity pharmaceutical intermediates.
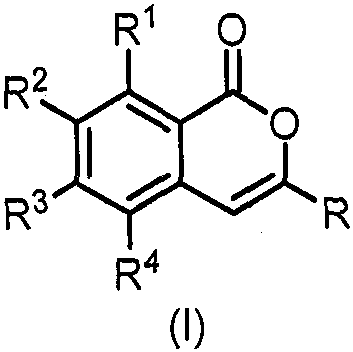
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isocoumarin skeleton has relied heavily on classical condensation reactions or cyclization strategies that often necessitate the use of pre-functionalized starting materials. These conventional pathways typically involve multiple synthetic steps, including the installation and subsequent removal of protecting groups or temporary directing groups to achieve the desired regioselectivity. Such processes inherently suffer from poor atom economy and generate substantial amounts of chemical waste, which poses significant challenges for environmental compliance and cost management in large-scale operations. Furthermore, the reliance on harsh reaction conditions or unstable intermediates in traditional methods can lead to inconsistent yields and difficulties in impurity control, complicating the purification process and ultimately affecting the quality of the final active pharmaceutical ingredient. The need for stoichiometric amounts of reagents and the generation of byproducts also drive up the raw material costs, making these older methodologies less attractive for modern, sustainability-focused manufacturing environments.
The Novel Approach
In stark contrast, the methodology described in CN112625020B introduces a paradigm shift by employing transition metal-catalyzed C-H activation, which allows for the direct functionalization of unactivated C-H bonds. This approach utilizes readily available oxazoline derivatives and sulfoxide sulfur ylides as key building blocks, eliminating the need for pre-halogenated substrates that are often expensive and hazardous to handle. The use of the oxazoline group is particularly strategic; it acts as a powerful directing group that guides the rhodium catalyst to the specific ortho-C-H bond, ensuring high regioselectivity without the need for post-reaction removal, as it becomes part of the final scaffold or transforms efficiently. This tandem reaction sequence not only reduces the total number of synthetic steps but also significantly improves the overall atom economy of the process. By integrating the carbene insertion and cyclization into a single catalytic cycle, this novel route minimizes waste generation and simplifies the downstream processing, offering a clear pathway to cost reduction in fine chemical manufacturing.
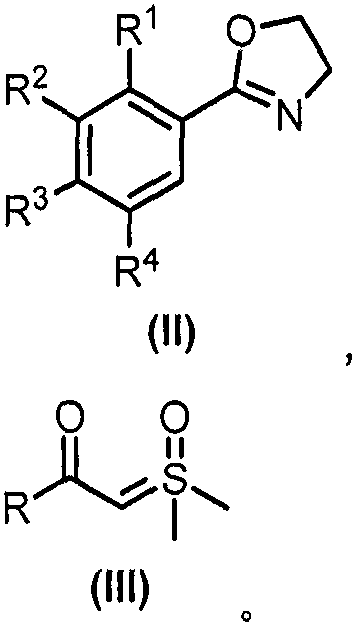
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The catalytic cycle underpinning this transformation is a sophisticated interplay of organometallic steps initiated by the activation of the Rhodium(III) precursor. The reaction typically employs a dimeric catalyst such as [Cp*RhCl2]2 in conjunction with a silver salt additive like AgNTf2, which serves to abstract the chloride ligands and generate the active cationic Rhodium species. This electrophilic metal center then coordinates with the nitrogen atom of the oxazoline directing group, facilitating the cleavage of the proximal aromatic C-H bond through a concerted metalation-deprotonation (CMD) mechanism. This step is critical as it forms a stable five-membered rhodacycle intermediate, which sets the stage for the subsequent carbon-carbon bond formation. The presence of additives such as sodium acetate and benzoic acid further modulates the acidity and basicity of the medium, optimizing the kinetics of the C-H activation step and ensuring the stability of the catalytic species throughout the prolonged reaction time at elevated temperatures.
Following the C-H activation, the sulfoxide sulfur ylide enters the catalytic cycle as a carbene precursor, reacting with the rhodacycle to form a metal-carbene intermediate via ligand exchange and nitrogen extrusion or similar pathways depending on the specific ylide structure. A unique feature of this specific patent is the nucleophilic attack by the oxygen atom of the oxazoline ring on the carbene carbon, rather than the nitrogen atom which is more commonly observed in similar quinolinone syntheses. This oxygen-directed cyclization leads to the formation of the lactone ring characteristic of isocoumarins, followed by protonolysis to release the final product and regenerate the active Rhodium(III) catalyst. This mechanistic nuance is vital for R&D directors as it explains the high selectivity for isocoumarin over other potential isomers. The robust nature of this catalytic cycle allows for a broad substrate scope, tolerating various electronic and steric environments on both the oxazoline and the ylide components, which is essential for generating diverse libraries of analogs for structure-activity relationship studies.
How to Synthesize Isocoumarin Derivatives Efficiently
The practical execution of this synthesis is designed to be straightforward yet precise, leveraging standard laboratory equipment that can be easily scaled to pilot and production reactors. The process begins with the careful weighing and mixing of the oxazoline substrate and the sulfoxide sulfur ylide in a molar ratio that typically favors the ylide to drive the reaction to completion, often around 1:2.0. These solids are suspended in a chlorinated solvent such as 1,2-dichloroethane (DCE), which provides the necessary solubility and thermal stability for the high-temperature conditions required. The addition of the catalyst system, comprising the rhodium dimer and the silver activator, along with the acid and base additives, must be performed under an inert atmosphere to prevent oxidation of the sensitive metal centers. Once the mixture is homogenized, it is heated to approximately 90°C and maintained under stirring for an extended period, typically around 18 hours, to ensure full conversion of the starting materials. Detailed standardized synthesis steps follow below.
- Prepare the reaction mixture by combining oxazoline substrate, sulfoxide sulfur ylide, [Cp*RhCl2]2 catalyst, AgNTf2 additive, NaOAc, and PhCOOH in DCE solvent under inert atmosphere.
- Heat the reaction mixture to 90°C and maintain stirring for approximately 18 hours to ensure complete conversion via C-H activation and cyclization.
- Upon completion, remove the solvent under reduced pressure and purify the crude residue using silica gel column chromatography to isolate the target isocoumarin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rhodium-catalyzed technology presents several compelling advantages that directly address common pain points in the sourcing of complex pharmaceutical intermediates. Firstly, the simplification of the synthetic route translates into a significant reduction in the number of unit operations required, which inherently lowers the operational expenditure associated with labor, energy, and equipment usage. By eliminating the need for separate steps to install and remove directing groups, the overall process time is drastically shortened, leading to faster turnaround times for custom synthesis projects and improved responsiveness to market demands. Furthermore, the high atom economy of the C-H activation strategy means that a larger proportion of the raw material mass is incorporated into the final product, reducing the volume of waste that needs to be treated and disposed of, thereby aligning with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive pre-functionalized starting materials and the reduction in waste disposal costs. While Rhodium is a precious metal, the catalyst loading is kept relatively low at around 2.5 mol%, and the ability to recycle or recover the metal from the reaction residue can further mitigate the raw material cost impact. Additionally, the use of commercially available oxazolines and sulfoxide sulfur ylides as starting blocks ensures a stable and competitive supply base, avoiding the price volatility often associated with specialized halogenated intermediates. The streamlined purification process, often requiring only a single column chromatography step or crystallization, further reduces the consumption of silica gel and solvents, contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this reaction conditions enhances reliability by reducing the risk of batch failures due to sensitive reagents. The starting materials are generally stable and can be sourced from multiple suppliers, reducing the risk of single-source dependency. The reaction tolerance to various functional groups means that a single platform technology can be used to produce a wide range of derivatives, allowing for greater flexibility in production planning and inventory management. This versatility is crucial for maintaining continuity of supply for key drug candidates, as the same reactor setup can be quickly adapted to synthesize different analogs without extensive requalification or retooling, ensuring that production schedules are met even when molecular requirements change.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of standard organic solvents and moderate temperatures that are well within the operating limits of typical stainless steel reactors used in the fine chemical industry. The absence of highly hazardous reagents or extreme pressure conditions simplifies the safety assessment and regulatory approval for scale-up. Moreover, the improved atom economy and reduced waste generation directly contribute to a lower environmental footprint, making it easier to comply with green chemistry principles and local environmental protection laws. This alignment with sustainability standards not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing partner, which is increasingly important for global pharmaceutical clients.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled a set of frequently asked questions based on the detailed specifications and experimental data provided in the documentation. These answers address common concerns regarding the feasibility, scope, and operational parameters of the Rhodium-catalyzed synthesis, providing a quick reference for technical teams evaluating this technology for their specific projects. The information is derived directly from the patent examples and mechanistic descriptions to ensure accuracy and relevance.
Q: What is the primary advantage of using oxazoline as a directing group in this synthesis?
A: Oxazoline serves as an intrinsic directing group that controls regioselectivity during C-H activation without requiring removal post-reaction, thereby enhancing atom and step economy compared to traditional removable directing groups.
Q: How does this Rhodium-catalyzed method improve upon conventional isocoumarin synthesis?
A: This method utilizes direct C-H activation and sulfoxide sulfur ylides as carbene precursors, avoiding the need for pre-functionalized substrates and enabling the construction of complex heterocyclic skeletons with higher efficiency and fewer synthetic steps.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction conditions involve standard organic solvents like DCE and moderate temperatures (90°C), making it adaptable for scale-up from laboratory to commercial production with appropriate engineering controls for solvent handling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isocoumarin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating the development of next-generation therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be seamlessly translated into reliable industrial manufacturing. We are committed to delivering high-purity isocoumarin derivatives that meet the rigorous quality standards required by the global pharmaceutical industry. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch, minimizing the risk of downstream processing issues for our clients.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your project's unique requirements, helping you optimize your budget without compromising on quality. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of fine chemical synthesis and secure a stable, cost-effective supply of critical intermediates for your pipeline.
