Advanced Palladium-Catalyzed Synthesis of 2-Aminoquinoline Dihydrofuran Compounds for Commercial Scale-Up
Introduction to Next-Generation Quinofuran Synthesis
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, particularly for constructing privileged structures found in bioactive natural products and pharmaceutical agents. Patent CN108409747B introduces a groundbreaking methodology for the synthesis of 2-aminoquinoline dihydrofuran compounds, leveraging a sophisticated palladium-catalyzed tandem cyclization strategy. This innovation addresses critical bottlenecks in the production of quinofuran derivatives, which are increasingly valued for their unique biological activities and potential applications in drug discovery pipelines. By utilizing readily available o-aminophenyl homopropargyl alcohols and isonitriles as primary building blocks, this technology enables the direct construction of the polycyclic framework in a single operational step. The significance of this approach lies not only in its chemical elegance but also in its practical viability for industrial manufacturing, offering a robust alternative to legacy methods that often suffer from poor atom economy and safety concerns.
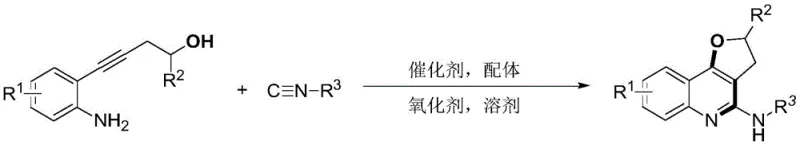
For research directors and process chemists seeking reliable pharmaceutical intermediate supplier partnerships, understanding the mechanistic underpinnings of such transformations is vital for assessing scalability and impurity profiles. The described method employs a synergistic combination of a palladium salt catalyst, specifically palladium tetratriphenylphosphine, and a bidentate phosphine ligand to drive the reaction forward under oxidative conditions. This catalytic system facilitates the activation of the alkyne moiety and the subsequent insertion of the isonitrile, leading to the formation of the fused quinoline-dihydrofuran core with high regioselectivity. The ability to tolerate a wide array of substituents on the aromatic ring and the isonitrile component underscores the versatility of this platform, making it an attractive candidate for the rapid generation of diverse compound libraries for biological screening.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinofuran skeletons has been plagued by significant operational complexities and safety hazards that hinder efficient commercial production. Traditional routes frequently rely on the pre-formation of quinoline intermediates, necessitating multiple synthetic steps that accumulate waste and reduce overall yield. Furthermore, many established protocols utilize explosive azide compounds as key reagents for ring closure, posing severe safety risks during scale-up and requiring specialized containment infrastructure that drives up capital expenditure. These legacy methods often demand harsh reaction conditions, including extreme temperatures or strong acidic environments, which can lead to the degradation of sensitive functional groups and the formation of difficult-to-remove impurities. Consequently, the cost reduction in API manufacturing is severely compromised by the need for extensive purification processes and the handling of hazardous materials, creating supply chain vulnerabilities for downstream pharmaceutical applications.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data revolutionizes this landscape by employing a direct, one-pot tandem cyclization that bypasses the need for unstable intermediates. By reacting o-aminophenyl homopropargyl alcohol directly with isonitriles in the presence of a mild copper salt oxidant, the process achieves the construction of the target polycyclic system with remarkable efficiency. The reaction proceeds smoothly at moderate temperatures ranging from 80 to 100°C, eliminating the energy-intensive requirements of cryogenic cooling or high-pressure reactors. This methodological shift not only enhances the safety profile of the operation by removing explosive reagents but also significantly streamlines the workflow, allowing for the direct isolation of the target compound after simple extraction and chromatography. For procurement managers, this translates to a more resilient supply chain capable of delivering high-purity OLED material or pharmaceutical precursors with reduced lead times and lower operational overhead.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The success of this synthetic strategy hinges on a meticulously orchestrated catalytic cycle involving palladium-mediated C-H activation and migratory insertion events. The reaction initiates with the oxidative addition of the palladium catalyst to the substrate, followed by intramolecular palladium oxide oxidation which activates the alkyne functionality for nucleophilic attack. Subsequently, the isonitrile molecule undergoes migratory insertion into the organopalladium species, extending the carbon framework and setting the stage for ring closure. A crucial step involves the intramolecular capture of the generated alkyl-carbon-palladium bond by the pendant amino group, which acts as an internal nucleophile to form the dihydrofuran ring. Finally, reductive elimination releases the desired 2-aminoquinoline dihydrofuran product and regenerates the active palladium catalyst, completing the cycle. This mechanism ensures high atom economy, as nearly all atoms from the starting materials are incorporated into the final product, minimizing waste generation.
From a quality control perspective, the mild nature of this catalytic system plays a pivotal role in impurity control and product consistency. The use of specific ligands like 1,3-bis(diphenylphosphino)propane helps stabilize the palladium center, preventing the formation of palladium black and other inactive species that could contaminate the product stream. Furthermore, the compatibility of the reaction with various functional groups, including halogens and esters, allows for the synthesis of complex derivatives without the need for protecting group strategies, which often introduce additional steps and potential sources of impurity. The resulting products exhibit clean spectral profiles, as evidenced by the characterization data in the patent, facilitating easier purification and ensuring that the final material meets the stringent purity specifications required for clinical-grade intermediates. This level of control is essential for maintaining batch-to-batch reproducibility in a GMP environment.
How to Synthesize 2-Aminoquinoline Dihydrofuran Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and reaction parameters to maximize yield and minimize side reactions. The process begins by dissolving the o-aminophenyl homopropargyl alcohol substrate along with the palladium catalyst and ligand in a mixed solvent system of acetonitrile and toluene, which provides the optimal polarity for solubilizing both organic and inorganic components. The addition of copper acetate as a stoichiometric oxidant is critical for driving the catalytic turnover, while the introduction of the isonitrile triggers the cascade sequence. Maintaining the reaction temperature between 80 and 100°C for a duration of 6 to 9 hours ensures complete conversion while preventing thermal decomposition of the sensitive heterocyclic product. Detailed standardized synthesis steps follow below for technical reference.
- Dissolve o-aminophenyl homopropargyl alcohol, palladium tetratriphenylphosphine catalyst, 1,3-bis(diphenylphosphino)propane ligand, and copper acetate oxidant in a mixed solvent of acetonitrile and toluene.
- Add the isonitrile substrate to the reaction mixture and stir vigorously at a temperature range of 80-100°C for 6 to 9 hours to facilitate the tandem cyclization.
- Upon completion, extract the reaction liquid with ethyl acetate, dry the organic phase over anhydrous magnesium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The reliance on cheap and commercially abundant starting materials, such as o-iodoaniline derivatives and 3-butyne-1-alcohol, ensures a stable and cost-effective supply of raw inputs, mitigating the risk of shortages that often plague specialty chemical markets. Moreover, the elimination of hazardous azide reagents drastically reduces the regulatory burden and insurance costs associated with manufacturing, while the simplified workup procedure lowers the consumption of solvents and consumables. These factors collectively contribute to substantial cost savings in fine chemical manufacturing, allowing companies to allocate resources more efficiently towards R&D and market expansion initiatives.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of earth-abundant copper salts as oxidants and standard palladium catalysts that can potentially be recovered and recycled. By avoiding the multi-step synthesis of pre-functionalized quinoline cores, the overall material throughput is increased, and the labor hours required per kilogram of product are significantly decreased. This streamlined approach eliminates the need for expensive cryogenic equipment and specialized safety infrastructure, resulting in a lower capital intensity for production facilities. Consequently, the total cost of ownership for producing these complex heterocycles is markedly reduced, enhancing the competitiveness of the final pharmaceutical or agrochemical products in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output regardless of minor fluctuations in environmental parameters, which is crucial for maintaining uninterrupted supply to downstream clients. The broad substrate scope means that a single manufacturing line can be adapted to produce a variety of analogues by simply swapping the isonitrile or the substituted aniline precursor, providing flexibility to meet changing market demands. This adaptability reduces the lead time for high-purity pharmaceutical intermediates, enabling faster response to clinical trial requirements or commercial launch schedules. Furthermore, the stability of the reagents allows for long-term storage and bulk purchasing, securing the supply chain against volatility in raw material pricing.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by the homogeneous nature of the catalytic system and the use of common industrial solvents like toluene and acetonitrile. The high atom economy of the tandem cyclization minimizes the generation of chemical waste, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. Waste streams are simpler to treat due to the absence of heavy metal contaminants from exotic catalysts or toxic byproducts from azide decomposition. This environmental compliance not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, offering clarity on reaction parameters and substrate compatibility. Understanding these details is essential for process engineers aiming to transfer this technology from the laboratory to pilot or commercial scale operations.
Q: What are the key advantages of this palladium-catalyzed method over traditional quinofuran synthesis?
A: This method utilizes a one-pot tandem cyclization strategy that avoids the need for pre-synthesized quinoline intermediates or hazardous azide compounds. It operates under mild thermal conditions (80-100°C) with high atom economy, significantly simplifying the operational workflow compared to multi-step conventional routes.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
A: The reaction demonstrates excellent functional group tolerance. The R1 substituent on the benzene ring can accommodate electron-withdrawing groups like chloro, bromo, and trifluoromethyl, as well as electron-donating groups like methyl and methoxy. The R3 group on the isonitrile allows for diverse alkyl chains including tert-butyl, cyclohexyl, and adamantyl.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to the use of common organic solvents like acetonitrile and toluene, commercially available palladium catalysts, and stable oxidants like copper acetate. The absence of cryogenic conditions or explosive reagents makes it safe and cost-effective for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminoquinoline Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies in accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative chemistries like this Pd-catalyzed cyclization are translated into reliable supply solutions. Our state-of-the-art facilities are equipped with rigorous QC labs and analytical capabilities to guarantee stringent purity specifications for every batch of 2-aminoquinoline dihydrofuran compounds we produce. We are committed to delivering high-quality intermediates that empower our clients to advance their therapeutic candidates with confidence and speed.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for complex heterocyclic intermediates.
