Advanced Palladium-Catalyzed Synthesis of 2-Aminoquinoline Dihydrofuran Compounds for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN108409747B introduces a groundbreaking synthetic strategy for producing 2-aminoquinoline dihydrofuran compounds, a class of molecules renowned for their unique biological activities and presence in natural products. This innovation leverages a palladium-catalyzed tandem cyclization reaction between o-aminophenyl homopropargyl alcohol and isonitriles, operating under remarkably mild conditions ranging from 80 to 100°C. By utilizing a synergistic catalytic system comprising palladium tetratriphenylphosphine and a copper acetate oxidant, this method achieves high atom economy while avoiding the safety hazards associated with traditional azide chemistry. For R&D directors and procurement specialists, this represents a significant leap forward in accessing high-purity pharmaceutical intermediates with reduced operational complexity and enhanced safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinofuran skeletons has been plagued by significant operational hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often necessitate the pre-synthesis of quinoline precursors, adding multiple steps to the overall process and drastically reducing overall yield due to cumulative losses at each stage. Furthermore, many established protocols rely on the use of explosive azide compounds as key reagents, introducing severe safety risks that require specialized containment facilities and rigorous hazard management protocols, thereby inflating production costs. The reliance on harsh reaction conditions in older methods also limits the tolerance for sensitive functional groups, restricting the diversity of derivatives that can be synthesized without extensive protection and deprotection strategies. These factors collectively create a bottleneck in the supply chain, leading to longer lead times and higher prices for final API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108409747B offers a streamlined, one-pot solution that directly constructs the polycyclic framework from simple, readily available starting materials. The core of this innovation lies in the tandem cyclization of o-aminophenyl homopropargyl alcohol with isonitriles, a reaction that proceeds smoothly in a mixed solvent system of acetonitrile and toluene. This approach not only simplifies the workflow by eliminating intermediate isolation steps but also utilizes cheap and safe reagents, such as o-iodoaniline-derived alcohols, which are easily sourced from bulk chemical suppliers. The reaction conditions are exceptionally mild, typically requiring temperatures between 80 and 100°C, which significantly reduces energy consumption compared to high-temperature pyrolysis methods. This novel pathway effectively bypasses the safety and efficiency pitfalls of prior art, establishing a new standard for cost reduction in pharmaceutical intermediate manufacturing.
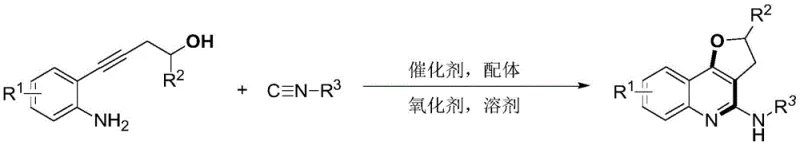
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The success of this synthetic route is underpinned by a sophisticated catalytic cycle initiated by intramolecular palladium oxide oxidation. The process begins with the activation of the o-aminophenyl homopropargyl alcohol substrate by the palladium catalyst, facilitating the formation of a key organopalladium intermediate. Subsequently, the isonitrile molecule undergoes migratory insertion into the carbon-palladium bond, a critical step that builds the carbon-nitrogen framework essential for the quinoline core. This insertion is followed by an intramolecular nucleophilic attack by the amino group present within the same molecule, which captures the generated alkyl carbon-palladium species. The cycle concludes with a reductive elimination step that releases the final 2-aminoquinoline dihydrofuran product and regenerates the active palladium catalyst for the next turnover. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters and minimize side reactions during scale-up.
From an impurity control perspective, the choice of copper acetate as a mild oxidant plays a pivotal role in maintaining product purity. Unlike aggressive oxidants that might degrade sensitive functional groups or generate toxic byproducts, copper acetate facilitates the necessary redox transitions without compromising the integrity of the substrate. The use of 1,3-bis(diphenylphosphino)propane (dppp) as a ligand further stabilizes the palladium center, ensuring consistent catalytic activity throughout the 3 to 9-hour reaction window. This stability is crucial for preventing the formation of palladium black or other inactive species that could contaminate the final product. Consequently, the resulting crude material requires less intensive purification, often needing only standard column chromatography with petroleum ether and ethyl acetate to achieve high-purity specifications required for downstream pharmaceutical applications.
How to Synthesize 2-Aminoquinoline Dihydrofuran Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction conditions is essential to maximize yield and reproducibility. The protocol dictates dissolving the o-aminophenyl homopropargyl alcohol substrate along with the palladium catalyst, ligand, and oxidant in a 1:1 volume ratio of acetonitrile and toluene before introducing the isonitrile. Maintaining the reaction temperature strictly within the 80-100°C range ensures optimal kinetics without triggering decomposition pathways. Following the reaction period, a standard aqueous workup involving ethyl acetate extraction and drying over anhydrous magnesium sulfate effectively isolates the organic product. For detailed operational parameters and specific molar ratios tailored to different substrates, please refer to the standardized synthesis guide below.
- Dissolve o-aminophenyl homopropargyl alcohol, palladium tetratriphenylphosphine catalyst, 1,3-bis(diphenylphosphino)propane ligand, and copper acetate oxidant in a mixed solvent of acetonitrile and toluene.
- Add the isonitrile substrate to the reaction mixture and stir vigorously at a temperature range of 80-100°C for 3 to 9 hours to facilitate the tandem cyclization.
- Upon completion, extract the reaction liquid with ethyl acetate, dry the organic phase over anhydrous magnesium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology translates into tangible strategic benefits regarding cost stability and supply continuity. The reliance on commodity chemicals such as o-iodoaniline, 3-butyne-1-ol, and common isonitriles means that raw material sourcing is not dependent on niche or volatile markets. This abundance of starting materials mitigates the risk of supply disruptions that often plague specialized reagent-dependent processes. Furthermore, the elimination of explosive azides removes the need for costly safety infrastructure upgrades, allowing existing manufacturing facilities to adopt this technology with minimal capital expenditure. The simplified one-pot nature of the reaction also reduces labor hours and solvent usage per kilogram of product, contributing to a leaner and more cost-effective production model.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive palladium salts and copper oxidants which are recoverable and reusable in many contexts, significantly lowering the cost of goods sold. By avoiding multi-step sequences and protecting group manipulations, the overall material throughput is improved, leading to substantial cost savings in raw material consumption. The high atom economy ensures that a greater proportion of the input mass is converted into the desired product, minimizing waste disposal costs which are a significant factor in modern chemical manufacturing. Additionally, the mild reaction conditions reduce energy overheads associated with heating and cooling, further enhancing the financial attractiveness of this route for large-scale operations.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against various functional groups ensures that supply chains remain resilient even when specific substrate batches vary slightly in quality. Since the starting materials are widely produced industrial chemicals, procurement teams can secure long-term contracts with multiple vendors, preventing single-source bottlenecks. The scalability of the reaction from gram to kilogram scales has been demonstrated through diverse examples in the patent, confirming that the process does not suffer from significant efficiency drops upon expansion. This reliability allows for accurate forecasting and inventory planning, crucial for meeting the Just-In-Time delivery requirements of global pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns well with increasingly stringent regulatory standards, as it avoids the generation of heavy metal waste streams associated with stoichiometric oxidants. The use of standard solvents like toluene and acetonitrile facilitates straightforward recycling and recovery processes, supporting green chemistry initiatives within the organization. The absence of hazardous azide intermediates simplifies the permitting process for new production lines, accelerating the time-to-market for new drug candidates. Moreover, the high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, reducing the load on wastewater treatment facilities and ensuring compliance with environmental discharge limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on substrate compatibility and process safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this tandem cyclization method over traditional quinofuran synthesis?
A: This method eliminates the need for pre-prepared quinoline intermediates or hazardous explosive azide compounds, offering a safer, one-pot synthetic route with high atom economy and mild reaction conditions suitable for industrial scaling.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
A: The process demonstrates excellent functional group tolerance, accommodating various substituents on the benzene ring such as halogens (Cl, Br), alkyl groups (methyl), electron-donating groups (methoxy), and electron-withdrawing groups (ester, trifluoromethyl), alongside diverse isonitriles like tert-butyl and cyclohexyl variants.
Q: Is this synthetic route scalable for commercial production of pharmaceutical intermediates?
A: Yes, the use of common palladium catalysts, inexpensive copper oxidants, and standard organic solvents like acetonitrile and toluene, combined with moderate temperatures (80-100°C), makes this protocol highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminoquinoline Dihydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our expertise in transition metal catalysis allows us to optimize this specific palladium-mediated route for maximum efficiency, guaranteeing a steady supply of high-quality 2-aminoquinoline dihydrofuran derivatives for your drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your commercial goals. Let us be your partner in transforming innovative chemistry into reliable commercial reality.
