Scalable Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Drug Manufacturing
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for more efficient and sustainable pathways to access biologically active scaffolds. Among these, quinolin-2(1H)one derivatives stand out as privileged structures in medicinal chemistry, featuring prominently in the development of antibiotics, antitumor agents, and receptor antagonists. A significant technological advancement in this domain is documented in patent CN113045489B, which discloses a novel preparation method for 3-arylquinoline-2(1H) ketone derivatives. This innovation leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally alters the synthetic approach by utilizing benzisoxazole as a dual-purpose reagent. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this technology is critical. The method not only streamlines the synthetic sequence but also enhances the economic viability of producing these high-value compounds by relying on inexpensive and readily available starting materials. 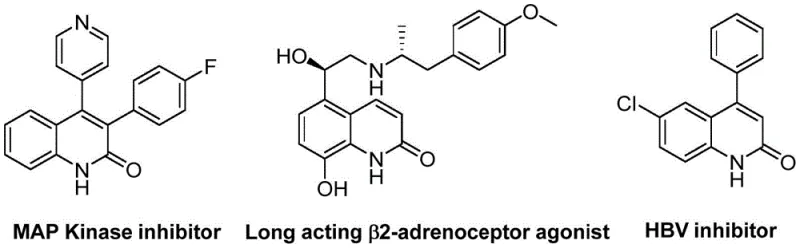
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolinone core has relied on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these established protocols have served the industry for decades, they often suffer from significant drawbacks that impact both operational safety and environmental compliance. Traditional routes frequently necessitate the use of harsh reagents, corrosive acids, or high-energy conditions that can limit the scope of compatible functional groups. Furthermore, many conventional strategies require multi-step sequences to install the carbonyl functionality, leading to cumulative yield losses and increased waste generation. For a procurement manager focused on cost reduction in pharmaceutical intermediates manufacturing, these inefficiencies translate directly into higher raw material consumption and more complex waste treatment protocols. The reliance on unstable or hazardous reagents also introduces supply chain risks, as the availability of such specialized chemicals can be volatile. Consequently, there is a pressing industrial need for a more robust, single-pot methodology that can deliver high purity without compromising on safety or scalability.
The Novel Approach
The methodology outlined in the referenced patent represents a paradigm shift by employing a transition metal-catalyzed carbonylation coupling reaction. This novel approach utilizes benzisoxazole and benzyl chloride compounds as the primary building blocks, reacting them in the presence of a palladium catalyst and a molybdenum carbonyl source. Unlike traditional methods that might require external carbon monoxide gas cylinders, this system generates the necessary carbonyl species in situ, significantly enhancing operational safety. The reaction proceeds at a moderate temperature of 100°C, which is conducive to energy-efficient manufacturing processes. By merging the nitrogen and formyl sources into a single benzisoxazole molecule, the synthetic route is drastically simplified, reducing the number of unit operations required. This simplification is a key driver for commercial scale-up of complex pharmaceutical intermediates, as it minimizes the potential for human error and equipment failure during production. The ability to tolerate a wide range of functional groups further underscores the versatility of this method, allowing for the rapid synthesis of diverse analog libraries essential for drug discovery programs.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
To fully appreciate the technical merit of this synthesis, one must delve into the mechanistic intricacies of the palladium-catalyzed cycle. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. Concurrently, the molybdenum hexacarbonyl acts as a solid surrogate for carbon monoxide, releasing CO slowly into the reaction medium to facilitate the carbonylation step without the need for high-pressure gas equipment. The benzisoxazole molecule then undergoes a ring-opening process, serving as a unique donor for both the nitrogen atom and the carbonyl carbon required to close the quinolinone ring. This dual functionality is chemically elegant, as it ensures perfect stoichiometric alignment between the nitrogen and carbonyl inputs, thereby minimizing side reactions associated with reagent imbalance. 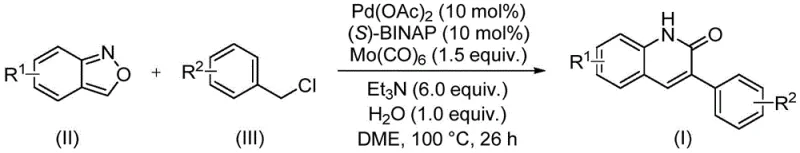
From an impurity control perspective, this mechanism offers distinct advantages over acid-mediated cyclizations. The mild basic conditions provided by triethylamine prevent the degradation of acid-sensitive functional groups that might be present on the substrate. Furthermore, the use of water as a co-reagent in specific molar ratios helps to modulate the reaction kinetics and assists in the hydrolysis steps necessary for the final product formation. The careful balance of the catalyst system, specifically the 0.1:0.1 molar ratio of palladium to ligand, ensures high turnover numbers while maintaining selectivity for the desired 3-aryl substitution pattern. For quality assurance teams, understanding that the reaction avoids harsh acidic workups means that the resulting crude material typically possesses a cleaner impurity profile. This reduces the burden on downstream purification processes, such as column chromatography or recrystallization, ultimately leading to higher overall recovery rates of the high-purity pharmaceutical intermediates required for clinical applications.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the intellectual property. The process begins with the precise weighing of palladium acetate, (S)-1,1'-binaphthyl-2,2'-bis-diphenylphosphine, and carbonyl molybdenum, which are combined with triethylamine and water in a sealed reaction vessel. The choice of solvent, ethylene glycol dimethyl ether (DME), is critical for solubilizing the organic substrates while maintaining thermal stability at the reaction temperature of 100°C. Operators must ensure that the reaction mixture is stirred thoroughly to maintain homogeneity, particularly given the presence of solid catalyst precursors and the inorganic carbonyl source. The detailed standardized synthesis steps see the guide below, which outlines the specific molar equivalents and timing required to achieve optimal conversion. Following the reaction period of approximately 26 hours, the workup involves filtration to remove metal residues, followed by silica gel treatment and purification.
- Combine palladium acetate, (S)-BINAP, carbonyl molybdenum, triethylamine, water, benzisoxazole, and benzyl chloride in a sealed vessel.
- Heat the reaction mixture to 100°C in ethylene glycol dimethyl ether (DME) solvent for approximately 26 hours.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and supply continuity, the economic implications of this patented technology are substantial. The shift towards using benzisoxazole and benzyl chloride as starting materials leverages commodity chemicals that are produced on a massive global scale, ensuring consistent availability and pricing stability. This stands in stark contrast to specialized heterocyclic building blocks that may have limited suppliers and long lead times. By adopting this route, manufacturers can achieve significant cost savings through the reduction of raw material expenses and the simplification of the supply chain network. The elimination of high-pressure carbon monoxide infrastructure also reduces capital expenditure requirements for reactor setup and safety monitoring systems. Furthermore, the operational simplicity translates into reduced labor costs and shorter training cycles for production staff, enhancing overall manufacturing efficiency.
- Cost Reduction in Manufacturing: The economic benefits are primarily driven by the use of inexpensive, commercially available reagents such as benzyl chloride and benzisoxazole, which are far more cost-effective than specialized acylating agents. The catalytic system, while utilizing palladium, operates at low loading levels, and the use of molybdenum carbonyl as a solid CO source eliminates the need for expensive gas handling infrastructure. Additionally, the streamlined one-pot nature of the reaction reduces solvent consumption and energy usage associated with multiple isolation steps. These factors collectively contribute to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved because the key starting materials are bulk chemicals with established production networks worldwide. Unlike custom-synthesized intermediates that rely on single-source vendors, benzyl chloride and its derivatives are produced by numerous chemical manufacturers, mitigating the risk of supply disruption. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing production schedules. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates and a more resilient inventory management strategy that can withstand market fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, avoiding the engineering challenges associated with high-pressure gas reactions. The use of a closed system with solid carbonyl sources minimizes the risk of toxic gas leaks, aligning with stringent environmental health and safety (EHS) regulations. Moreover, the high atom economy of using benzisoxazole as a dual source reduces the generation of chemical waste, simplifying effluent treatment processes. This environmental compatibility is increasingly important for maintaining regulatory approval and corporate sustainability goals, making the technology attractive for long-term commercial partnerships and green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on feasibility and performance. Understanding these details is essential for technical teams evaluating the integration of this method into existing production workflows.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, eliminating the need for separate toxic formylating agents and simplifying the overall reaction stoichiometry.
Q: Is this process suitable for large-scale pharmaceutical production?
A: Yes, the method utilizes commercially available starting materials like benzyl chloride and operates under relatively mild thermal conditions, facilitating safer commercial scale-up compared to traditional high-pressure carbonylation.
Q: What is the functional group tolerance of this catalytic system?
A: The palladium-catalyzed system demonstrates wide substrate tolerance, accommodating various substituents such as halogens, alkoxy groups, and cyano groups on the aromatic rings without significant yield degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into robust commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to plant. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-arylquinolin-2(1H)one derivatives meets the exacting standards required by the global pharmaceutical industry. We understand that consistency and quality are non-negotiable in drug manufacturing, and our technical team is dedicated to optimizing every parameter of the synthesis to maximize yield and minimize impurities.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency. Let us be your partner in delivering high-quality chemical solutions that drive your business forward.
