Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery. Patent CN113045489B introduces a groundbreaking methodology for the preparation of 3-arylquinolin-2(1H)one derivatives, a privileged structure found in numerous bioactive molecules ranging from MAP Kinase inhibitors to HBV inhibitors. This innovation addresses the longstanding challenges associated with constructing the quinolinone core by leveraging a palladium-catalyzed aminocarbonylation strategy. Unlike traditional approaches that often require hazardous gases or multi-step sequences, this novel process utilizes benzisoxazole as a unique dual-purpose reagent, acting simultaneously as the nitrogen source and the formyl donor. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process efficiency and safety, enabling the rapid generation of diverse chemical libraries for biological evaluation while maintaining strict control over impurity profiles.
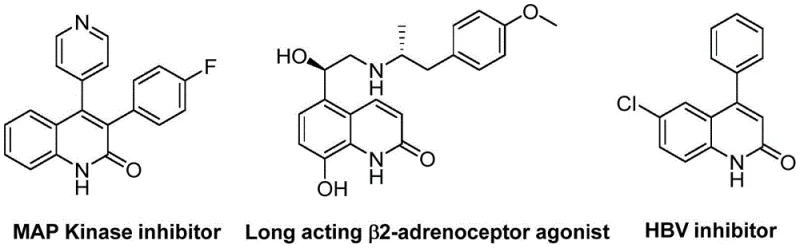
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied upon classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods are well-documented in academic literature, they frequently suffer from significant drawbacks when applied to industrial-scale manufacturing. Conventional routes often necessitate the use of corrosive reagents, harsh acidic or basic conditions, and elevated temperatures that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purge. Furthermore, transition metal-catalyzed improvements to these classical methods have historically struggled with limited substrate scope or the requirement for expensive and air-sensitive ligands. The reliance on external carbon monoxide gas for carbonylation reactions poses severe safety hazards and requires specialized high-pressure equipment, creating substantial barriers to entry for contract development and manufacturing organizations (CDMOs) aiming for cost reduction in API manufacturing. These operational complexities often result in prolonged lead times and inconsistent batch-to-batch quality, which are unacceptable for modern supply chain requirements.
The Novel Approach
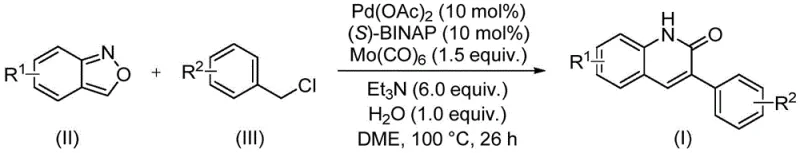
In stark contrast to these legacy techniques, the method disclosed in patent CN113045489B offers a streamlined, one-pot solution that fundamentally reimagines the construction of the quinolinone ring system. By employing benzisoxazole derivatives alongside benzyl chloride compounds, this process achieves the formation of the target heterocycle through an elegant aminocarbonylation cascade. The key innovation lies in the ability of the benzisoxazole moiety to undergo N-O bond cleavage under palladium catalysis, effectively releasing the necessary nitrogen atom and a carbonyl equivalent in situ. This eliminates the need for handling toxic carbon monoxide gas, significantly enhancing workplace safety and reducing the capital expenditure required for reactor infrastructure. As illustrated in the reaction scheme below, the transformation proceeds smoothly in ethylene glycol dimethyl ether (DME) at a moderate temperature of 100°C, demonstrating exceptional compatibility with a wide array of electronic and steric environments on the aromatic rings.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
The mechanistic pathway of this transformation is a sophisticated interplay of organometallic steps that ensure high regioselectivity and yield. The cycle initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step is crucial for activating the benzylic carbon-halogen bond, setting the stage for subsequent insertion events. Following oxidative addition, the coordination and insertion of the carbonyl species—derived from the decomposition of molybdenum hexacarbonyl or the benzisoxazole fragmentation—into the palladium-carbon bond occurs. The unique role of benzisoxazole cannot be overstated; it acts as an internal reservoir for the carbonyl group, undergoing ring opening to provide the necessary atoms for the lactam formation. This intramolecular delivery system minimizes side reactions such as homocoupling of the benzyl chloride, which is a common pitfall in standard carbonylation protocols. The presence of triethylamine serves to neutralize the hydrochloric acid byproduct, driving the equilibrium forward and preventing catalyst deactivation through protonation of the phosphine ligands.
From an impurity control perspective, this mechanism offers distinct advantages for process chemists. The use of a well-defined catalytic system involving Pd(OAc)2 and (S)-BINAP ensures that the reaction kinetics are predictable and reproducible. The moderate reaction temperature of 100°C is sufficient to drive the reaction to completion within 26 hours without promoting thermal degradation of the product or the starting materials. Experimental data from the patent indicates that yields can reach as high as 97% for certain substrates, such as those bearing electron-withdrawing cyano groups, while electron-donating groups like tert-butyl also perform exceptionally well with yields around 95%. This broad functional group tolerance implies that downstream purification is simplified, as fewer side products are generated compared to harsher acidic cyclizations. For quality assurance teams, this translates to a cleaner crude profile, reducing the burden on chromatographic purification steps and improving the overall mass balance of the manufacturing process.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize efficiency. The protocol dictates a specific molar ratio of catalyst to substrate, typically utilizing 10 mol% of palladium acetate and 10 mol% of the (S)-BINAP ligand relative to the limiting reagent. Molybdenum hexacarbonyl is employed in a slight excess (1.5 equivalents) to ensure a sufficient concentration of the carbonyl source throughout the reaction duration. The reaction is conducted in DME solvent, which provides excellent solubility for both the organic substrates and the inorganic bases. Triethylamine is added in a significant excess (6.0 equivalents) to act as both a base and a potential ligand stabilizer, while a small amount of water (1.0 equivalent) is surprisingly beneficial, likely facilitating the hydrolysis steps involved in the final aromatization of the quinolinone ring. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are provided in the technical guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary driver for cost optimization lies in the accessibility and price point of the starting materials. Benzisoxazoles and substituted benzyl chlorides are commodity chemicals available from multiple global vendors, ensuring a competitive bidding environment and mitigating the risk of single-source dependency. This availability directly contributes to cost reduction in pharmaceutical intermediate manufacturing by stabilizing raw material costs and preventing supply bottlenecks. Furthermore, the elimination of gaseous carbon monoxide removes the need for specialized gas cylinders, flow meters, and leak detection systems, thereby lowering the operational overhead and safety compliance costs associated with the production facility. The simplicity of the work-up procedure, involving filtration followed by standard silica gel chromatography, allows for rapid turnover of batches, enhancing the overall throughput of the manufacturing suite.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of earth-abundant catalysts and inexpensive reagents. By avoiding the use of exotic or proprietary reagents that command premium pricing, the overall cost of goods sold (COGS) is significantly lowered. The high atom economy of the reaction, where the majority of the starting material mass is incorporated into the final product, minimizes waste disposal costs. Additionally, the high yields reported across a diverse range of substrates mean that less starting material is required to produce a given amount of API intermediate, further driving down the unit cost. The robustness of the catalyst system also suggests that catalyst loading could potentially be optimized further in large-scale runs, offering additional avenues for financial savings without compromising reaction performance.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical for maintaining continuous drug production, and this method supports that goal through its reliance on stable, shelf-stable reagents. Unlike processes that require cryogenic conditions or strictly anhydrous environments, this reaction tolerates the presence of stoichiometric water, relaxing the stringent drying requirements for solvents and reagents. This tolerance simplifies logistics and storage, as raw materials do not require specialized desiccants or inert atmosphere packaging during transport. The broad substrate scope means that a single manufacturing platform can be adapted to produce a wide variety of analogues simply by swapping the benzyl chloride or benzisoxazole component. This flexibility allows manufacturers to respond rapidly to changing demand patterns or clinical trial requirements without the need for extensive process re-validation or equipment modification.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden challenges, but this protocol appears inherently scalable due to its homogeneous nature and moderate thermal profile. The reaction temperature of 100°C is easily achievable with standard steam heating or oil baths, avoiding the need for high-energy electrical heating or complex cooling loops. From an environmental standpoint, the use of DME as a solvent is favorable compared to chlorinated solvents often used in heterocycle synthesis, aligning with green chemistry principles. The reduction in hazardous waste generation, particularly the avoidance of heavy metal waste streams associated with stoichiometric oxidants used in other methods, simplifies wastewater treatment and regulatory compliance. This alignment with sustainability goals is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet corporate social responsibility targets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is essential for assessing the feasibility of integrating this chemistry into existing production workflows.
Q: What is the primary advantage of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for separate carbon monoxide gas or complex formylating agents, thereby simplifying the operational safety and equipment requirements.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds at 100°C for 26 hours in ethylene glycol dimethyl ether (DME) using a palladium catalyst system with triethylamine as the base.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, accommodating substituents such as halogens, alkoxy groups, cyano, and trifluoromethyl groups on both the benzisoxazole and benzyl chloride substrates without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
As the demand for complex heterocyclic intermediates continues to grow in the oncology and antiviral sectors, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands ready to support your drug development programs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest standards of safety and efficiency, ensuring that your supply of high-purity 3-arylquinolin-2(1H)one derivatives remains uninterrupted. We understand that stringent purity specifications are non-negotiable in the pharmaceutical industry, which is why our rigorous QC labs employ advanced analytical techniques to verify the identity and purity of every batch before release. Whether you require custom synthesis of novel analogues or metric-ton production of established intermediates, our team is dedicated to delivering excellence.
We invite you to collaborate with us to explore how this innovative synthetic route can accelerate your project timelines and optimize your budget. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your strategic partner in transforming cutting-edge chemical research into commercial reality.
